Key Insights
The global Endoluminal Suturing Devices Market is poised for significant expansion, projected to reach USD 98.4 million in 2024 and experience a robust CAGR of 12.32% during the forecast period of 2025-2033. This growth is underpinned by an increasing prevalence of chronic diseases requiring minimally invasive interventions, particularly in the cardiovascular and gastrointestinal sectors. Technological advancements, such as the development of more sophisticated robotic-assisted suturing systems and single-use, highly maneuverable devices, are key drivers fueling market adoption. The demand for faster patient recovery times and reduced hospital stays further accentuates the preference for endoluminal suturing over traditional open surgical methods. Leading companies are actively investing in research and development to introduce innovative solutions that enhance precision and safety in these complex procedures, thereby expanding the market's potential.
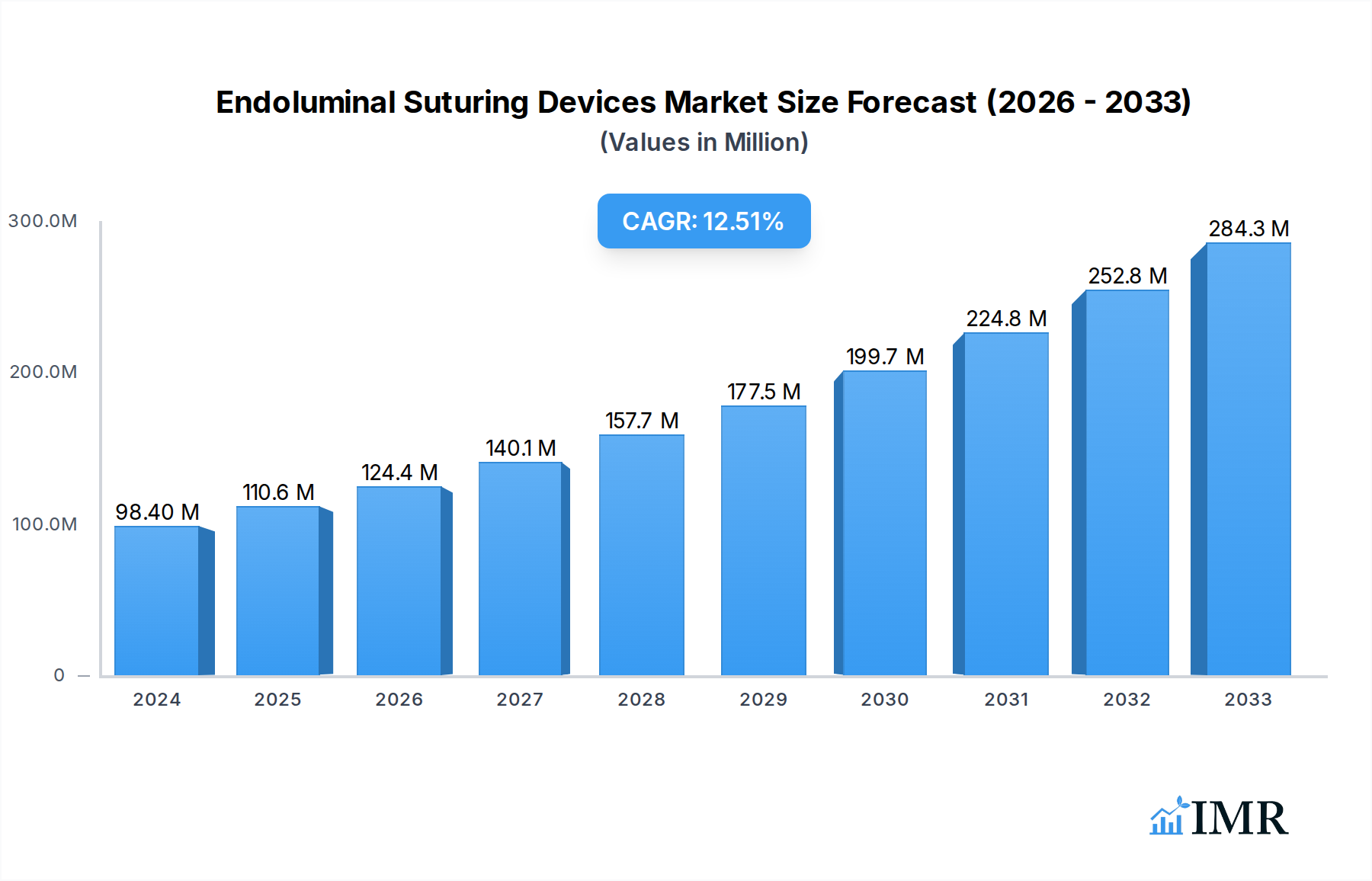
Endoluminal Suturing Devices Market Market Size (In Million)

The market's trajectory is also influenced by the growing emphasis on preventative healthcare and early diagnosis, which often necessitates less invasive treatment options. The rising geriatric population, more susceptible to conditions requiring surgical intervention, contributes significantly to market demand. While the market benefits from these strong growth drivers, potential restraints include the high cost associated with advanced endoluminal suturing devices and the need for specialized training for healthcare professionals. However, the continuous innovation in product design, leading to more cost-effective disposable options and enhanced procedural efficiency, is expected to mitigate these restraints. Geographically, North America and Europe currently dominate the market due to advanced healthcare infrastructure and high patient awareness, but the Asia Pacific region is expected to witness substantial growth driven by increasing healthcare expenditure and the expanding medical device industry.
Endoluminal Suturing Devices Market Company Market Share

Endoluminal Suturing Devices Market: Comprehensive Analysis and Future Outlook (2019–2033)
This in-depth report provides a comprehensive analysis of the global Endoluminal Suturing Devices Market, exploring market dynamics, growth trends, regional dominance, product landscape, key drivers, emerging opportunities, and a detailed player analysis. With a focus on minimally invasive surgery and advanced gastrointestinal procedures, this report offers critical insights for stakeholders navigating this rapidly evolving sector. The study encompasses a historical period from 2019 to 2024, a base and estimated year of 2025, and a robust forecast period from 2025 to 2033.
Endoluminal Suturing Devices Market Dynamics & Structure
The Endoluminal Suturing Devices Market is characterized by moderate concentration, with a few key players holding significant market share, yet fostering innovation through a dynamic competitive landscape. Technological innovation is a primary driver, fueled by the increasing demand for less invasive surgical techniques and improved patient outcomes in procedures such as gastrointestinal surgeries and cardiovascular interventions. Stringent regulatory frameworks, while ensuring patient safety, also influence product development cycles and market entry. Competitive product substitutes, including staplers and advanced adhesives, exist but endoluminal sutures offer unique advantages in certain applications. End-user demographics, particularly the aging global population and rising prevalence of chronic diseases, contribute to the growing demand for surgical solutions. Merger and acquisition (M&A) activities are observed as companies seek to expand their product portfolios and market reach within the surgical device market.
- Market Concentration: Moderate, with key players like Medtronic and Johnson & Johnson (Ethicon Inc.) leading.
- Technological Innovation Drivers: Demand for minimally invasive surgery, improved patient outcomes, and advancements in robotics.
- Regulatory Frameworks: FDA and EMA approvals are crucial, impacting time-to-market for new medical devices.
- Competitive Product Substitutes: Surgical staplers, endoscopic clips, and bio-adhesives.
- End-User Demographics: Growing elderly population and increasing incidence of gastrointestinal and cardiovascular diseases.
- M&A Trends: Strategic acquisitions to enhance product offerings and expand geographic presence.
Endoluminal Suturing Devices Market Growth Trends & Insights
The Endoluminal Suturing Devices Market is poised for substantial growth, driven by a confluence of factors including increasing adoption rates of minimally invasive surgical techniques, technological disruptions, and evolving consumer behavior towards less invasive procedures. The market size is projected to expand significantly, with an estimated Compound Annual Growth Rate (CAGR) of xx% from 2025 to 2033, reaching an estimated market value of USD XXX million units in 2025. This growth trajectory is underpinned by the relentless pursuit of improved patient outcomes, reduced hospital stays, and faster recovery times, which are paramount considerations for both patients and healthcare providers. The rising prevalence of gastrointestinal disorders, cardiovascular diseases, and gynecological conditions globally further fuels the demand for advanced surgical solutions like endoluminal suturing devices. Furthermore, advancements in endoscopic technology, including improved visualization and maneuverability, are enabling surgeons to perform increasingly complex procedures with greater precision and efficacy, thereby accelerating the adoption of these innovative devices.
The shift in consumer preference towards less invasive alternatives is a significant behavioral shift impacting the market. Patients are increasingly seeking procedures that minimize scarring, pain, and the risk of complications. Endoluminal suturing, by allowing for suture placement and knot tying through natural orifices or small incisions, directly addresses these patient desires. Healthcare systems worldwide are also recognizing the economic benefits associated with shorter hospital stays and reduced post-operative care, making endoluminal suturing a cost-effective solution in the long run. The growing penetration of these devices in various surgical specialties, from bariatric surgery to cardiology, indicates a broadening acceptance and integration into standard surgical protocols. Continuous research and development efforts are focused on enhancing the usability, safety, and efficacy of these devices, leading to the introduction of novel functionalities and materials. These disruptions are not only expanding the existing market but also creating new application areas for endoluminal suturing technologies, ensuring sustained growth and market expansion throughout the forecast period. The integration of robotics with endoluminal suturing platforms also presents a significant growth accelerator, promising enhanced precision and remote surgical capabilities.
Dominant Regions, Countries, or Segments in Endoluminal Suturing Devices Market
The Endoluminal Suturing Devices Market witnesses significant growth across various regions and segments, with a notable dominance observed in the Gastrointestinal application segment, driven by its widespread use in treating a plethora of digestive system disorders. Within product types, Disposable endoluminal suturing devices are currently leading the market due to their inherent advantages of sterility, reduced risk of cross-contamination, and convenience for single-use procedures, which are increasingly favored in healthcare settings to mitigate infection risks. This segment is projected to maintain its leading position throughout the forecast period, with an estimated xx million units sold in 2025.
North America, particularly the United States, stands as the dominant region in the global Endoluminal Suturing Devices Market. This dominance is attributed to several key factors:
- Advanced Healthcare Infrastructure: High adoption rates of advanced medical technologies and a well-established reimbursement system for minimally invasive procedures.
- High Prevalence of Target Diseases: A significant burden of gastrointestinal, cardiovascular, and obesity-related diseases necessitates advanced surgical interventions.
- Strong R&D Investment: Substantial investment in research and development by leading medical device manufacturers based in the region, fostering rapid innovation.
- Favorable Regulatory Environment: A relatively streamlined regulatory process for new medical device approvals, encouraging market entry for innovative products.
Europe follows closely, driven by a strong healthcare network and increasing awareness and acceptance of minimally invasive surgeries. The region benefits from government initiatives promoting technological advancements in healthcare and a growing demand for sophisticated surgical solutions. The Asia-Pacific region is emerging as a high-growth market, fueled by rapid economic development, expanding healthcare access, and a growing middle class with increasing disposable income to spend on advanced medical treatments. Countries like China and India are witnessing substantial investments in their healthcare sectors, creating a fertile ground for the adoption of endoluminal suturing devices.
The dominance of the Gastrointestinal application segment is further amplified by the rising incidence of conditions such as gastroesophageal reflux disease (GERD), inflammatory bowel disease (IBD), peptic ulcers, and bariatric surgery needs. These conditions often require endoscopic interventions for diagnosis and treatment, where endoluminal suturing plays a crucial role in lesion closure, tissue approximation, and securing devices. The segment is estimated to account for xx% of the total market in 2025. The ongoing advancements in endoscopic procedures, including ESD (Endoscopic Submucosal Dissection) and EMR (Endoscopic Mucosal Resection), directly boost the demand for sophisticated suturing devices.
Endoluminal Suturing Devices Market Product Landscape
The product landscape of the Endoluminal Suturing Devices Market is characterized by continuous innovation aimed at enhancing precision, safety, and ease of use for surgeons. Key product developments include advanced articulating tips for improved maneuverability in complex anatomical structures, integrated imaging capabilities for better visualization during procedures, and the development of bioabsorbable suture materials that eliminate the need for removal. The performance metrics focus on suture strength, knot security, and tissue interaction to ensure optimal wound closure and healing. Unique selling propositions often revolve around the specific application benefits, such as the ability to perform complex closures in narrow lumens or the reduced tissue trauma compared to traditional suturing methods. Technological advancements are also leading to the integration of smart features, such as real-time feedback on suture tension, further optimizing surgical outcomes.
Key Drivers, Barriers & Challenges in Endoluminal Suturing Devices Market
Key Drivers:
- Technological Advancements: Continuous innovation in endoscopic technology and suturing mechanisms, enabling more complex and less invasive procedures.
- Growing Demand for Minimally Invasive Surgery: Patient preference and surgeon preference for reduced trauma, shorter recovery times, and improved aesthetics.
- Increasing Prevalence of Chronic Diseases: Rising incidence of gastrointestinal, cardiovascular, and gynecological conditions necessitating surgical intervention.
- Favorable Reimbursement Policies: Increasing coverage for minimally invasive procedures by healthcare payers.
- Aging Global Population: A demographic shift leading to higher demand for surgical interventions across various specialties.
Barriers & Challenges:
- High Cost of Devices: The advanced nature of endoluminal suturing devices can lead to higher acquisition costs for healthcare facilities.
- Steep Learning Curve for Surgeons: While designed for ease of use, some advanced techniques require specialized training, potentially limiting initial adoption.
- Regulatory Hurdles: Stringent approval processes and evolving regulatory requirements can delay product launches and market entry.
- Competition from Alternative Technologies: The presence of established alternatives like surgical staplers and clips can pose a competitive challenge in certain applications.
- Reimbursement Variability: Inconsistent reimbursement policies across different regions and healthcare systems can impact market penetration.
- Supply Chain Disruptions: Global supply chain vulnerabilities can affect the availability and cost of critical components, as seen with xx million units of potential supply chain impact.
Emerging Opportunities in Endoluminal Suturing Devices Market
Emerging opportunities in the Endoluminal Suturing Devices Market lie in the development of more sophisticated robotic-assisted suturing systems, which promise enhanced precision and remote surgical capabilities. Untapped markets in developing economies with growing healthcare expenditure and increasing adoption of advanced medical technologies represent significant growth potential. Innovative applications in fields like reconstructive surgery and tissue engineering are also emerging, where endoluminal suturing could play a pivotal role. Furthermore, the development of smart sutures with integrated sensing capabilities for monitoring wound healing and delivering targeted therapies presents a promising avenue for future innovation and market expansion.
Growth Accelerators in the Endoluminal Suturing Devices Market Industry
Growth accelerators in the Endoluminal Suturing Devices Market industry are primarily driven by technological breakthroughs, such as the integration of artificial intelligence for enhanced surgical guidance and automation. Strategic partnerships between medical device manufacturers and academic institutions are fostering collaborative research and development, leading to accelerated innovation. Market expansion strategies focused on emerging economies, coupled with targeted physician training programs, are crucial for increasing adoption rates and driving long-term growth. The development of novel bio-compatible and dissolvable suture materials further enhances the appeal and versatility of these devices.
Key Players Shaping the Endoluminal Suturing Devices Market Market
- Mellon Medical B V
- Medtronic
- Smith & Nephew Inc
- Johnson & Johnson (Ethicon Inc.)
- LSI Solutions Inc
- Sutrue Limited
- Boston Scientific Corporation
- B Braun SE
Notable Milestones in Endoluminal Suturing Devices Market Sector
- January 2023: Able Medical Devices launched valkyrie looped sternotomy sutures.
- May 2022: Paragon 28, Inc. launched the Grappler Suture Anchor System, designed to address the challenges of performing soft tissue tensioning and ligament reconstruction in acute foot and ankle procedures.
In-Depth Endoluminal Suturing Devices Market Market Outlook
The future outlook for the Endoluminal Suturing Devices Market is exceptionally bright, driven by sustained demand for minimally invasive solutions and ongoing technological advancements. Strategic investments in research and development, coupled with the expansion of product portfolios to cater to a wider range of clinical applications, will be key differentiators. Growth accelerators such as the increasing integration with robotic surgery platforms and the exploration of novel bio-materials will unlock new market segments and further solidify the industry's growth trajectory. Opportunities in emerging economies and the potential for advancements in interventional cardiology and advanced gastrointestinal procedures will provide significant avenues for market expansion and revenue generation, estimating a potential market size increase of xx% by 2033.
Endoluminal Suturing Devices Market Segmentation
-
1. Product Type
- 1.1. Disposable
- 1.2. Reusable
-
2. Application
- 2.1. Cardiovascular
- 2.2. Gynecological
- 2.3. Gastrointestinal
- 2.4. Orthopedic
- 2.5. Others
Endoluminal Suturing Devices Market Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
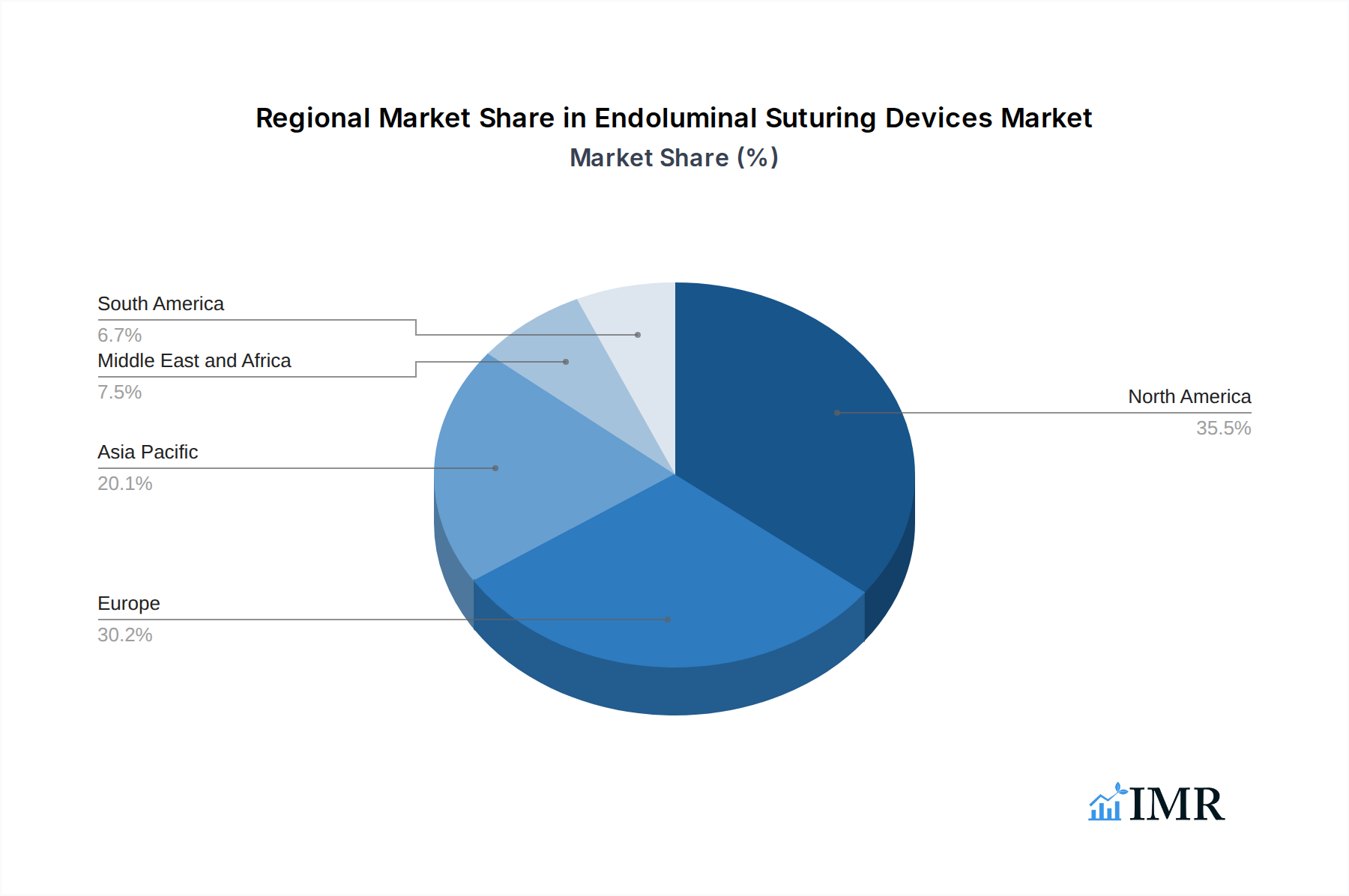
Endoluminal Suturing Devices Market Regional Market Share

Geographic Coverage of Endoluminal Suturing Devices Market
Endoluminal Suturing Devices Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 12.32% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. IMR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product Type
- 5.1.1. Disposable
- 5.1.2. Reusable
- 5.2. Market Analysis, Insights and Forecast - by Application
- 5.2.1. Cardiovascular
- 5.2.2. Gynecological
- 5.2.3. Gastrointestinal
- 5.2.4. Orthopedic
- 5.2.5. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. Europe
- 5.3.3. Asia Pacific
- 5.3.4. Middle East and Africa
- 5.3.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Product Type
- 6. Global Endoluminal Suturing Devices Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product Type
- 6.1.1. Disposable
- 6.1.2. Reusable
- 6.2. Market Analysis, Insights and Forecast - by Application
- 6.2.1. Cardiovascular
- 6.2.2. Gynecological
- 6.2.3. Gastrointestinal
- 6.2.4. Orthopedic
- 6.2.5. Others
- 6.1. Market Analysis, Insights and Forecast - by Product Type
- 7. North America Endoluminal Suturing Devices Market Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Product Type
- 7.1.1. Disposable
- 7.1.2. Reusable
- 7.2. Market Analysis, Insights and Forecast - by Application
- 7.2.1. Cardiovascular
- 7.2.2. Gynecological
- 7.2.3. Gastrointestinal
- 7.2.4. Orthopedic
- 7.2.5. Others
- 7.1. Market Analysis, Insights and Forecast - by Product Type
- 8. Europe Endoluminal Suturing Devices Market Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Product Type
- 8.1.1. Disposable
- 8.1.2. Reusable
- 8.2. Market Analysis, Insights and Forecast - by Application
- 8.2.1. Cardiovascular
- 8.2.2. Gynecological
- 8.2.3. Gastrointestinal
- 8.2.4. Orthopedic
- 8.2.5. Others
- 8.1. Market Analysis, Insights and Forecast - by Product Type
- 9. Asia Pacific Endoluminal Suturing Devices Market Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Product Type
- 9.1.1. Disposable
- 9.1.2. Reusable
- 9.2. Market Analysis, Insights and Forecast - by Application
- 9.2.1. Cardiovascular
- 9.2.2. Gynecological
- 9.2.3. Gastrointestinal
- 9.2.4. Orthopedic
- 9.2.5. Others
- 9.1. Market Analysis, Insights and Forecast - by Product Type
- 10. Middle East and Africa Endoluminal Suturing Devices Market Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Product Type
- 10.1.1. Disposable
- 10.1.2. Reusable
- 10.2. Market Analysis, Insights and Forecast - by Application
- 10.2.1. Cardiovascular
- 10.2.2. Gynecological
- 10.2.3. Gastrointestinal
- 10.2.4. Orthopedic
- 10.2.5. Others
- 10.1. Market Analysis, Insights and Forecast - by Product Type
- 11. South America Endoluminal Suturing Devices Market Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Product Type
- 11.1.1. Disposable
- 11.1.2. Reusable
- 11.2. Market Analysis, Insights and Forecast - by Application
- 11.2.1. Cardiovascular
- 11.2.2. Gynecological
- 11.2.3. Gastrointestinal
- 11.2.4. Orthopedic
- 11.2.5. Others
- 11.1. Market Analysis, Insights and Forecast - by Product Type
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Mellon Medical B V
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Medtronic
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Smith & Nephew Inc
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Johnson & Johnson (Ethicon Inc )
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 LSI Solutions Inc
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Sutrue Limited
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Boston Scientific Corporation
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 B Braun SE
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.1 Mellon Medical B V
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Endoluminal Suturing Devices Market Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: Global Endoluminal Suturing Devices Market Volume Breakdown (K Unit, %) by Region 2025 & 2033
- Figure 3: North America Endoluminal Suturing Devices Market Revenue (undefined), by Product Type 2025 & 2033
- Figure 4: North America Endoluminal Suturing Devices Market Volume (K Unit), by Product Type 2025 & 2033
- Figure 5: North America Endoluminal Suturing Devices Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 6: North America Endoluminal Suturing Devices Market Volume Share (%), by Product Type 2025 & 2033
- Figure 7: North America Endoluminal Suturing Devices Market Revenue (undefined), by Application 2025 & 2033
- Figure 8: North America Endoluminal Suturing Devices Market Volume (K Unit), by Application 2025 & 2033
- Figure 9: North America Endoluminal Suturing Devices Market Revenue Share (%), by Application 2025 & 2033
- Figure 10: North America Endoluminal Suturing Devices Market Volume Share (%), by Application 2025 & 2033
- Figure 11: North America Endoluminal Suturing Devices Market Revenue (undefined), by Country 2025 & 2033
- Figure 12: North America Endoluminal Suturing Devices Market Volume (K Unit), by Country 2025 & 2033
- Figure 13: North America Endoluminal Suturing Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 14: North America Endoluminal Suturing Devices Market Volume Share (%), by Country 2025 & 2033
- Figure 15: Europe Endoluminal Suturing Devices Market Revenue (undefined), by Product Type 2025 & 2033
- Figure 16: Europe Endoluminal Suturing Devices Market Volume (K Unit), by Product Type 2025 & 2033
- Figure 17: Europe Endoluminal Suturing Devices Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 18: Europe Endoluminal Suturing Devices Market Volume Share (%), by Product Type 2025 & 2033
- Figure 19: Europe Endoluminal Suturing Devices Market Revenue (undefined), by Application 2025 & 2033
- Figure 20: Europe Endoluminal Suturing Devices Market Volume (K Unit), by Application 2025 & 2033
- Figure 21: Europe Endoluminal Suturing Devices Market Revenue Share (%), by Application 2025 & 2033
- Figure 22: Europe Endoluminal Suturing Devices Market Volume Share (%), by Application 2025 & 2033
- Figure 23: Europe Endoluminal Suturing Devices Market Revenue (undefined), by Country 2025 & 2033
- Figure 24: Europe Endoluminal Suturing Devices Market Volume (K Unit), by Country 2025 & 2033
- Figure 25: Europe Endoluminal Suturing Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 26: Europe Endoluminal Suturing Devices Market Volume Share (%), by Country 2025 & 2033
- Figure 27: Asia Pacific Endoluminal Suturing Devices Market Revenue (undefined), by Product Type 2025 & 2033
- Figure 28: Asia Pacific Endoluminal Suturing Devices Market Volume (K Unit), by Product Type 2025 & 2033
- Figure 29: Asia Pacific Endoluminal Suturing Devices Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 30: Asia Pacific Endoluminal Suturing Devices Market Volume Share (%), by Product Type 2025 & 2033
- Figure 31: Asia Pacific Endoluminal Suturing Devices Market Revenue (undefined), by Application 2025 & 2033
- Figure 32: Asia Pacific Endoluminal Suturing Devices Market Volume (K Unit), by Application 2025 & 2033
- Figure 33: Asia Pacific Endoluminal Suturing Devices Market Revenue Share (%), by Application 2025 & 2033
- Figure 34: Asia Pacific Endoluminal Suturing Devices Market Volume Share (%), by Application 2025 & 2033
- Figure 35: Asia Pacific Endoluminal Suturing Devices Market Revenue (undefined), by Country 2025 & 2033
- Figure 36: Asia Pacific Endoluminal Suturing Devices Market Volume (K Unit), by Country 2025 & 2033
- Figure 37: Asia Pacific Endoluminal Suturing Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 38: Asia Pacific Endoluminal Suturing Devices Market Volume Share (%), by Country 2025 & 2033
- Figure 39: Middle East and Africa Endoluminal Suturing Devices Market Revenue (undefined), by Product Type 2025 & 2033
- Figure 40: Middle East and Africa Endoluminal Suturing Devices Market Volume (K Unit), by Product Type 2025 & 2033
- Figure 41: Middle East and Africa Endoluminal Suturing Devices Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 42: Middle East and Africa Endoluminal Suturing Devices Market Volume Share (%), by Product Type 2025 & 2033
- Figure 43: Middle East and Africa Endoluminal Suturing Devices Market Revenue (undefined), by Application 2025 & 2033
- Figure 44: Middle East and Africa Endoluminal Suturing Devices Market Volume (K Unit), by Application 2025 & 2033
- Figure 45: Middle East and Africa Endoluminal Suturing Devices Market Revenue Share (%), by Application 2025 & 2033
- Figure 46: Middle East and Africa Endoluminal Suturing Devices Market Volume Share (%), by Application 2025 & 2033
- Figure 47: Middle East and Africa Endoluminal Suturing Devices Market Revenue (undefined), by Country 2025 & 2033
- Figure 48: Middle East and Africa Endoluminal Suturing Devices Market Volume (K Unit), by Country 2025 & 2033
- Figure 49: Middle East and Africa Endoluminal Suturing Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 50: Middle East and Africa Endoluminal Suturing Devices Market Volume Share (%), by Country 2025 & 2033
- Figure 51: South America Endoluminal Suturing Devices Market Revenue (undefined), by Product Type 2025 & 2033
- Figure 52: South America Endoluminal Suturing Devices Market Volume (K Unit), by Product Type 2025 & 2033
- Figure 53: South America Endoluminal Suturing Devices Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 54: South America Endoluminal Suturing Devices Market Volume Share (%), by Product Type 2025 & 2033
- Figure 55: South America Endoluminal Suturing Devices Market Revenue (undefined), by Application 2025 & 2033
- Figure 56: South America Endoluminal Suturing Devices Market Volume (K Unit), by Application 2025 & 2033
- Figure 57: South America Endoluminal Suturing Devices Market Revenue Share (%), by Application 2025 & 2033
- Figure 58: South America Endoluminal Suturing Devices Market Volume Share (%), by Application 2025 & 2033
- Figure 59: South America Endoluminal Suturing Devices Market Revenue (undefined), by Country 2025 & 2033
- Figure 60: South America Endoluminal Suturing Devices Market Volume (K Unit), by Country 2025 & 2033
- Figure 61: South America Endoluminal Suturing Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 62: South America Endoluminal Suturing Devices Market Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Product Type 2020 & 2033
- Table 2: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Product Type 2020 & 2033
- Table 3: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Application 2020 & 2033
- Table 4: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Application 2020 & 2033
- Table 5: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Region 2020 & 2033
- Table 6: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Region 2020 & 2033
- Table 7: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Product Type 2020 & 2033
- Table 8: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Product Type 2020 & 2033
- Table 9: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Application 2020 & 2033
- Table 10: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Application 2020 & 2033
- Table 11: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Country 2020 & 2033
- Table 12: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 13: United States Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: United States Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 15: Canada Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Canada Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 17: Mexico Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 18: Mexico Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 19: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Product Type 2020 & 2033
- Table 20: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Product Type 2020 & 2033
- Table 21: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Application 2020 & 2033
- Table 22: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Application 2020 & 2033
- Table 23: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Country 2020 & 2033
- Table 24: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 25: Germany Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Germany Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 27: United Kingdom Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: United Kingdom Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 29: France Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 30: France Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 31: Italy Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Italy Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 33: Spain Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: Spain Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 35: Rest of Europe Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Europe Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 37: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Product Type 2020 & 2033
- Table 38: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Product Type 2020 & 2033
- Table 39: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Application 2020 & 2033
- Table 40: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Application 2020 & 2033
- Table 41: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Country 2020 & 2033
- Table 42: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 43: China Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: China Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 45: Japan Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Japan Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 47: India Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 48: India Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 49: Australia Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 50: Australia Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 51: South Korea Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 52: South Korea Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 53: Rest of Asia Pacific Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 54: Rest of Asia Pacific Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 55: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Product Type 2020 & 2033
- Table 56: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Product Type 2020 & 2033
- Table 57: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Application 2020 & 2033
- Table 58: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Application 2020 & 2033
- Table 59: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Country 2020 & 2033
- Table 60: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 61: GCC Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 62: GCC Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 63: South Africa Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 64: South Africa Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 65: Rest of Middle East and Africa Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 66: Rest of Middle East and Africa Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 67: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Product Type 2020 & 2033
- Table 68: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Product Type 2020 & 2033
- Table 69: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Application 2020 & 2033
- Table 70: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Application 2020 & 2033
- Table 71: Global Endoluminal Suturing Devices Market Revenue undefined Forecast, by Country 2020 & 2033
- Table 72: Global Endoluminal Suturing Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 73: Brazil Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 74: Brazil Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 75: Argentina Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 76: Argentina Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
- Table 77: Rest of South America Endoluminal Suturing Devices Market Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 78: Rest of South America Endoluminal Suturing Devices Market Volume (K Unit) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Endoluminal Suturing Devices Market?
The projected CAGR is approximately 12.32%.
2. Which companies are prominent players in the Endoluminal Suturing Devices Market?
Key companies in the market include Mellon Medical B V , Medtronic, Smith & Nephew Inc, Johnson & Johnson (Ethicon Inc ), LSI Solutions Inc, Sutrue Limited, Boston Scientific Corporation, B Braun SE.
3. What are the main segments of the Endoluminal Suturing Devices Market?
The market segments include Product Type, Application.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
Increased Number of Surgical Procedures; Increase in Accidents and Trauma Cases.
6. What are the notable trends driving market growth?
The Disposable Segment is Expected to Hold a Significant Share in the Automated Suturing Devices Market..
7. Are there any restraints impacting market growth?
Low Product Availability and High Cost of the Devices; Lack of Skilled Professionals.
8. Can you provide examples of recent developments in the market?
January 2023: Able Medical Devices launched valkyrie looped sternotomy sutures.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Endoluminal Suturing Devices Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Endoluminal Suturing Devices Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Endoluminal Suturing Devices Market?
To stay informed about further developments, trends, and reports in the Endoluminal Suturing Devices Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
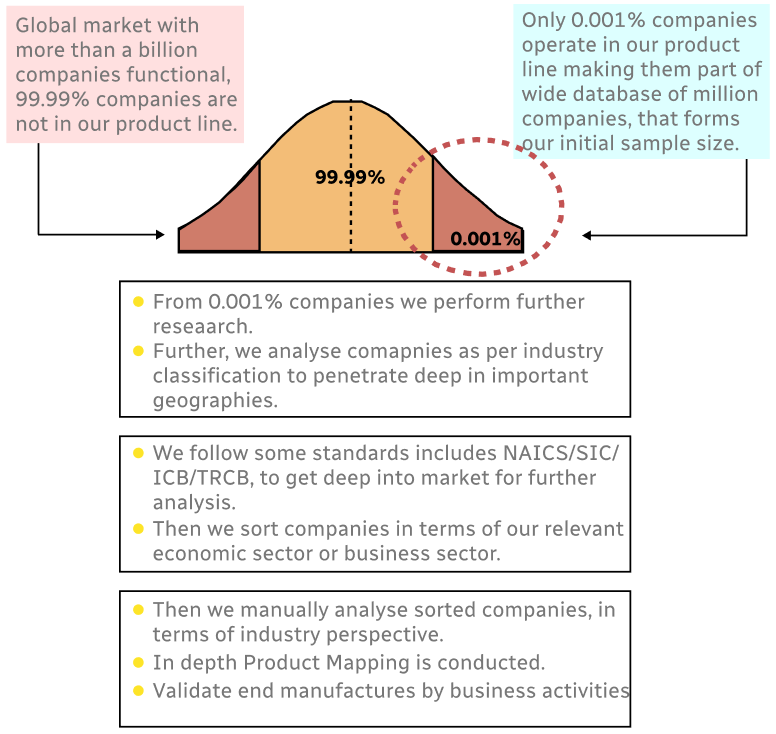
Step 1 - Identification of Relevant Samples Size from Population Database
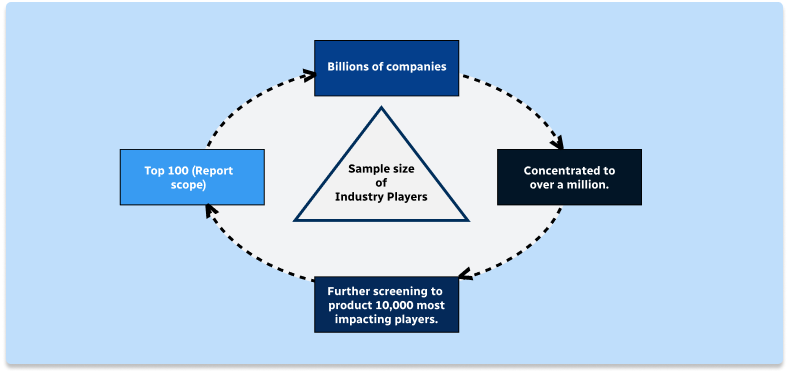
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
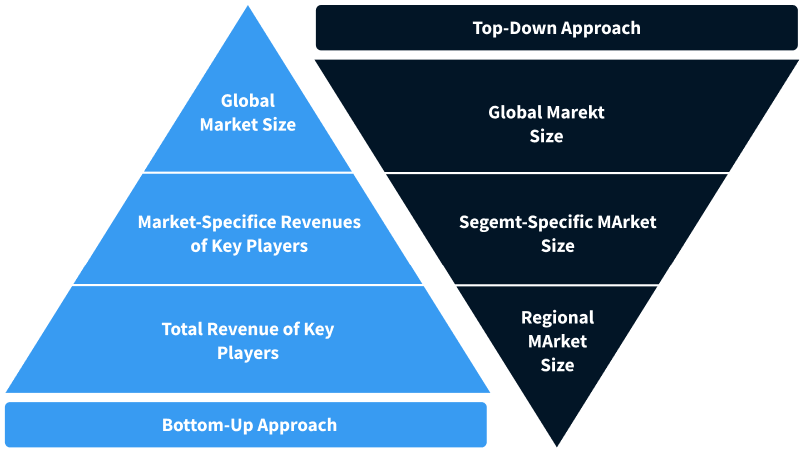
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
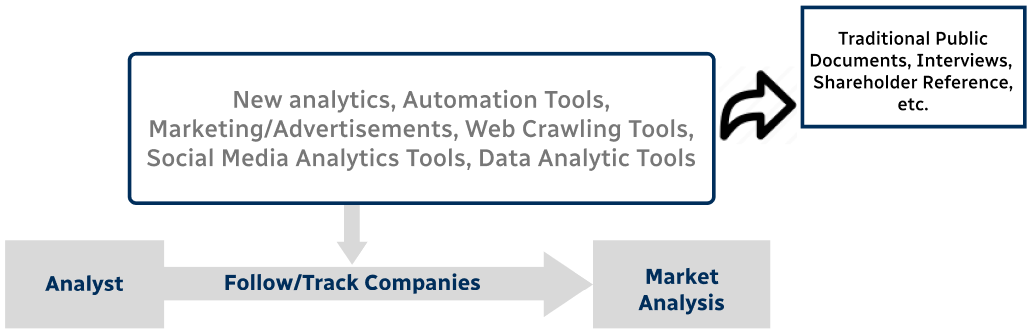
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


