Key Insights
The global Infant Care Equipment Market is poised for significant expansion, projected to reach an estimated $1.88 Billion by 2025, and is expected to witness a robust Compound Annual Growth Rate (CAGR) of 6.69% during the forecast period of 2025-2033. This growth is propelled by a confluence of factors, including rising global birth rates, increasing awareness regarding neonatal health and safety, and the continuous technological advancements in medical devices designed for infant care. The market is segmented into Fetal Care Equipment and Neonatal Care Equipment. Within Neonatal Care, Phototherapy Equipment stands out as a critical segment due to its effectiveness in treating neonatal jaundice, a common condition affecting newborns. The increasing prevalence of hyperbilirubinemia necessitates the widespread adoption of advanced phototherapy solutions, driving demand for innovative and efficient devices. Furthermore, a growing emphasis on specialized care for premature infants and those with critical health conditions contributes to the overall market expansion.
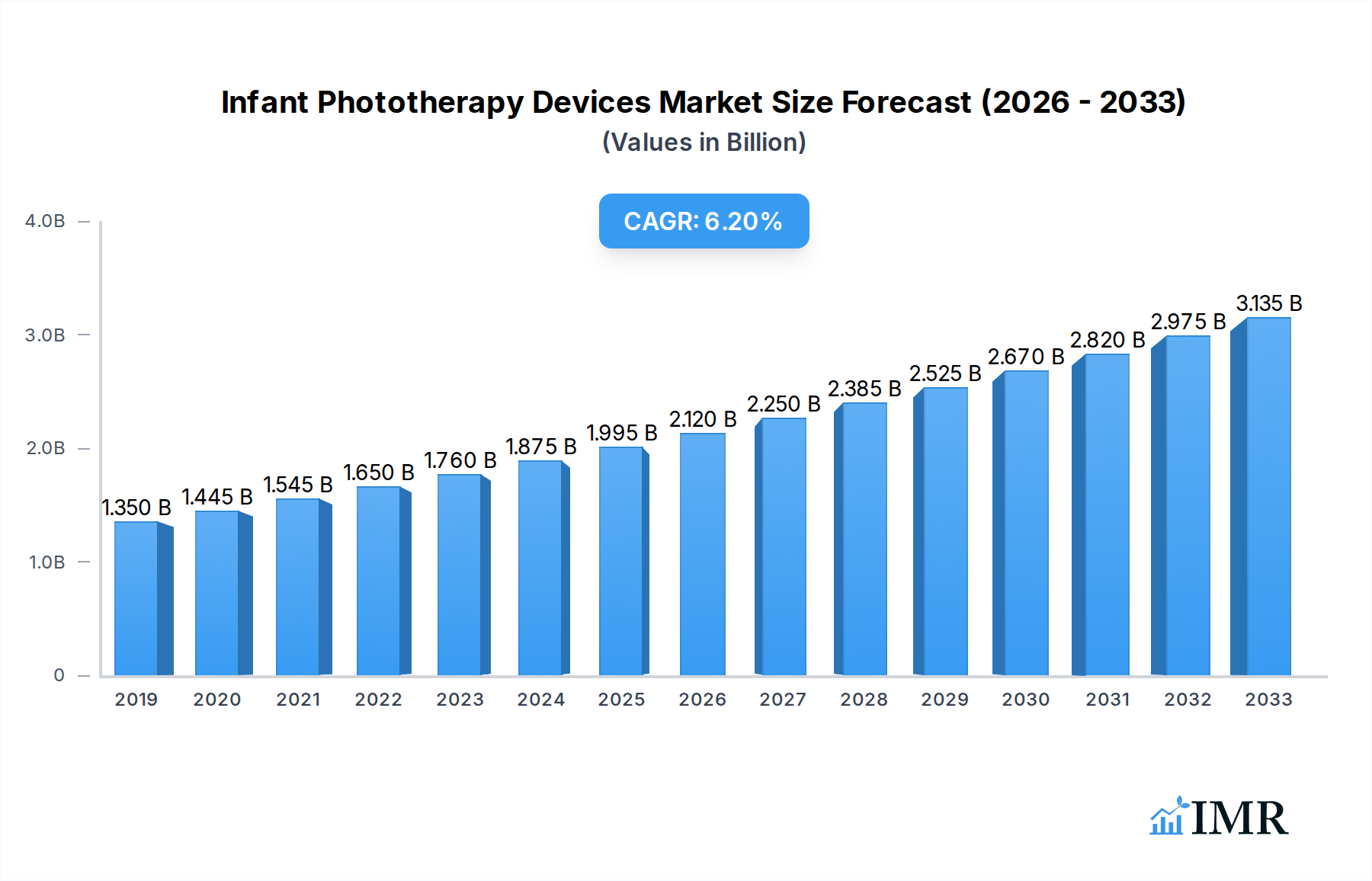
Infant Phototherapy Devices Market Market Size (In Billion)

The market is characterized by intense competition among leading global players such as GE Healthcare, Koninklijke Philips N.V., Medtronic PLC, and Siemens Healthineers AG, who are actively engaged in research and development to introduce next-generation infant care solutions. Key trends include the development of portable and integrated phototherapy devices, improved monitoring capabilities, and the adoption of energy-efficient technologies. However, the market also faces certain restraints, including the high cost of advanced infant care equipment and reimbursement challenges in certain regions, which can impede widespread adoption. Despite these challenges, the expanding healthcare infrastructure, particularly in emerging economies, and the increasing focus on improving neonatal outcomes are expected to sustain the positive growth trajectory of the infant care equipment market. The European region, with its well-established healthcare systems and high adoption rates of advanced medical technologies, is anticipated to hold a significant market share, driven by countries like Germany, the United Kingdom, and France.
Infant Phototherapy Devices Market Company Market Share

Unlock the potential of the global Infant Phototherapy Devices Market with our in-depth analysis. This report provides crucial insights into market dynamics, growth trends, key players, and emerging opportunities within the neonatal and fetal care sectors. Essential for manufacturers, distributors, healthcare providers, and investors seeking to navigate this critical segment of the medical device industry.
Infant Phototherapy Devices Market Dynamics & Structure
The infant phototherapy devices market exhibits a moderate concentration, with key players like GE Company (GE Healthcare), Koninklijke Philips N V, and Medtronic PLC holding significant market shares. Technological innovation is a primary driver, with continuous advancements in LED technology, wavelength accuracy, and device portability enhancing treatment efficacy and patient comfort. Regulatory frameworks, including FDA approvals and CE marking, play a crucial role in market entry and product development, ensuring patient safety and device performance. Competitive product substitutes, such as exchange transfusions, are less common for mild to moderate jaundice but represent a competitive consideration. End-user demographics are shifting towards a greater demand for home-use phototherapy devices, driven by parental convenience and reduced healthcare costs. Mergers and acquisitions (M&A) activity, while not overtly high, indicates strategic consolidation and portfolio expansion by larger entities. For instance, the acquisition of smaller specialized device manufacturers by broader medical technology companies contributes to market restructuring. The market's structure is influenced by both the fetal care equipment and neonatal care equipment segments, with advancements in one often influencing the other.
- Market Concentration: Moderate, with established players dominating.
- Technological Innovation Drivers: LED advancements, portability, user-friendliness.
- Regulatory Frameworks: FDA, CE marking, stringent quality control.
- Competitive Product Substitutes: Exchange transfusions (limited scope).
- End-User Demographics: Growing demand for home-use devices.
- M&A Trends: Strategic consolidation and portfolio expansion.
Infant Phototherapy Devices Market Growth Trends & Insights
The infant phototherapy devices market is poised for robust expansion, driven by an increasing global birth rate and a rising incidence of neonatal jaundice. Market size evolution is characterized by consistent year-over-year growth, with the global market size projected to reach $XXX million units by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of approximately XX% during the forecast period (2025–2033). Adoption rates for advanced phototherapy solutions, particularly those utilizing blue LED light, are steadily increasing due to their superior efficacy, reduced heat emission, and lower energy consumption compared to traditional fluorescent lamps. Technological disruptions, such as the integration of smart features for real-time monitoring and data logging, are enhancing treatment precision and clinical outcomes. Consumer behavior shifts are evident, with a growing preference among parents for convenient, non-invasive treatment options that can be administered at home, leading to a surge in demand for portable and user-friendly phototherapy units. This trend is further supported by healthcare provider recommendations and evolving reimbursement policies favoring outpatient care. The market penetration of advanced phototherapy devices is expected to rise significantly as awareness about neonatal jaundice management grows and healthcare infrastructure in emerging economies improves. The interplay between the fetal care equipment and neonatal care equipment segments also contributes to overall market growth, as early detection and management during fetal development can preempt the need for more intensive neonatal interventions. The historical period (2019–2024) has laid the groundwork for this accelerated growth, with increased R&D investments and a growing understanding of the long-term implications of untreated neonatal jaundice. The base year (2025) provides a snapshot of the current market landscape, serving as a crucial reference point for projected growth trajectory.
Dominant Regions, Countries, or Segments in Infant Phototherapy Devices Market
The North America region is a dominant force in the infant phototherapy devices market, primarily driven by high per capita healthcare spending, advanced healthcare infrastructure, and a strong emphasis on neonatal intensive care units (NICUs). The United States, in particular, contributes a substantial share due to its well-established medical device industry and continuous innovation in neonatal care technologies. Within the Product Type segmentation, the Neonatal Care Equipment segment, especially Phototherapy Equipment, is the primary growth engine, accounting for a significant market share. This segment benefits from the widespread prevalence of neonatal jaundice and the established protocols for its management. The fetal care equipment segment, while crucial for early diagnostics, directly influences the demand for subsequent neonatal care.
Key Drivers in North America:
- High Healthcare Expenditure: Enabling access to advanced and specialized neonatal equipment.
- Advanced Healthcare Infrastructure: Extensive network of hospitals and NICUs equipped with state-of-the-art technology.
- Technological Adoption: Early and rapid adoption of innovative phototherapy devices.
- Awareness and Research: High levels of awareness regarding neonatal health and continuous research driving product development.
- Favorable Reimbursement Policies: Robust insurance coverage for neonatal treatments.
Within the Neonatal Care Equipment segment, Phototherapy Equipment leads in market share and growth potential. This is directly linked to the prevalence of hyperbilirubinemia in newborns. Innovations in light-emitting diode (LED) phototherapy, offering targeted wavelengths, increased efficiency, and reduced heat, are further fueling adoption. The Neonatal Monitoring Devices and Respiratory Assistance and Monitoring Devices also contribute significantly, as these often work in conjunction with phototherapy to provide comprehensive neonatal care. The Fetal Care Equipment segment, including Ultrasound Devices and Fetal Dopplers, plays a vital indirect role by enabling early detection of potential complications that might necessitate neonatal interventions, including phototherapy. The growth potential in Asia-Pacific is also substantial, driven by an increasing birth rate and improving healthcare access, making it a key region to watch for future market expansion.
Infant Phototherapy Devices Market Product Landscape
The infant phototherapy devices market is characterized by continuous product innovation focused on enhancing treatment efficacy, patient safety, and user convenience. Advancements in LED technology have led to the development of devices emitting specific blue light wavelengths (460-480 nm), which are highly effective in degrading bilirubin. Manufacturers are increasingly integrating features such as adjustable intensity, timers, and integrated temperature control to optimize treatment. Portable phototherapy units, designed for homecare settings, are gaining traction, offering parents a convenient option for managing mild to moderate jaundice. Performance metrics are continually being refined, with a focus on faster treatment times, reduced energy consumption, and improved patient comfort, minimizing the risk of overheating and dehydration. Unique selling propositions often revolve around the compactness and portability of devices, the precision of light delivery, and enhanced safety features.
Key Drivers, Barriers & Challenges in Infant Phototherapy Devices Market
Key Drivers:
The infant phototherapy devices market is propelled by several key drivers. Firstly, the persistent global rise in neonatal jaundice incidence, particularly in premature infants, necessitates effective treatment solutions. Secondly, technological advancements, such as the transition to energy-efficient and effective LED phototherapy, are driving market growth by offering superior treatment outcomes and lower operational costs. The increasing global birth rate, especially in developing economies, further fuels demand. Furthermore, growing parental awareness regarding neonatal health and the availability of non-invasive treatment options like phototherapy encourages adoption. Favorable government initiatives and healthcare policies promoting neonatal care also play a significant role.
Key Barriers & Challenges:
Despite the growth, the market faces certain barriers and challenges. The high initial cost of advanced phototherapy devices can be a restraint, particularly in resource-limited settings. Stringent regulatory approvals and the need for extensive clinical validation can prolong product development timelines and increase costs. Competition from established players and the emergence of generic alternatives can also exert pricing pressure. Supply chain disruptions, as witnessed in recent global events, can impact manufacturing and distribution, leading to potential shortages. Moreover, the availability of alternative treatments, though less common for widespread jaundice, remains a competitive factor. The need for continuous training for healthcare professionals on the effective use of newer technologies also presents an ongoing challenge.
Emerging Opportunities in Infant Phototherapy Devices Market
Emerging opportunities within the infant phototherapy devices market lie in the expansion of home-use phototherapy solutions, driven by the increasing preference for at-home care and telehealth integration. Untapped markets in developing economies, where the incidence of neonatal jaundice is high but access to advanced equipment is limited, present significant growth potential. Innovative applications, such as the development of wearable or integrated phototherapy solutions, could redefine treatment paradigms. Evolving consumer preferences for user-friendly and smart devices, equipped with data tracking capabilities for remote monitoring by healthcare professionals, also represent a lucrative avenue. The development of specialized phototherapy devices tailored for specific neonatal conditions or genetic predispositions could further diversify the market.
Growth Accelerators in the Infant Phototherapy Devices Market Industry
Growth accelerators for the infant phototherapy devices market are primarily centered around continuous technological breakthroughs that enhance efficacy and patient safety. The transition to advanced LED technology, offering precise wavelength delivery and reduced heat output, is a significant accelerator. Strategic partnerships between medical device manufacturers and research institutions are fostering rapid innovation and product development. Market expansion strategies, particularly focusing on emerging economies with growing healthcare expenditures and increasing birth rates, are crucial growth catalysts. The development of user-friendly, portable devices that facilitate home-based care and reduce hospitalizations is also a key accelerator, aligning with global healthcare trends towards cost-effectiveness and patient convenience.
Key Players Shaping the Infant Phototherapy Devices Market Market
- F Hoffmann-La Roche Ltd
- GE Company (GE Healthcare)
- Atom Medical Corporation
- Koninklijke Philips N V
- Medtronic PLC
- Siemens Healthineers AG
- Gentinge AB
- Becton Dickinson and Company
- Dragerwerk AG & Co KGaA
- Fisher & Paykel Healthcare Ltd
Notable Milestones in Infant Phototherapy Devices Market Sector
- 2019: Increased adoption of blue LED phototherapy devices due to improved efficacy and reduced side effects.
- 2020: Introduction of portable and compact phototherapy units for home-use applications, driven by parental convenience.
- 2021: Focus on developing smart phototherapy devices with integrated monitoring and data logging capabilities for enhanced clinical management.
- 2022: Emergence of hybrid phototherapy solutions combining light therapy with other treatment modalities.
- 2023: Greater emphasis on energy-efficient and sustainable phototherapy device designs.
- 2024: Advancements in wavelength customization for phototherapy devices to target specific bilirubin degradation pathways.
In-Depth Infant Phototherapy Devices Market Market Outlook
The infant phototherapy devices market is set for sustained growth, fueled by ongoing technological advancements in LED-based treatments and an increasing global demand for effective neonatal jaundice management. Growth accelerators will continue to include innovations in device portability and smart functionalities, enabling enhanced home-care and remote monitoring. Strategic collaborations and market expansion into underserved regions, particularly in emerging economies, will further bolster market penetration. The evolving landscape of neonatal care, with a growing emphasis on non-invasive and efficient treatment options, positions the phototherapy devices market for significant future potential and strategic opportunities.
Infant Phototherapy Devices Market Segmentation
-
1. Product Type
-
1.1. Fetal Care Equipment
- 1.1.1. Fetal Dopplers
- 1.1.2. Fetal Magnetic Resonance Imaging (MRI) Devices
- 1.1.3. Ultrasound Devices
- 1.1.4. Fetal Pulse Oximeters
- 1.1.5. Other Fetal Care Equipment
-
1.2. Neonatal Care Equipment
- 1.2.1. Incubators
- 1.2.2. Neonatal Monitoring Devices
- 1.2.3. Phototherapy Equipment
- 1.2.4. Respiratory Assistance and Monitoring Devices
- 1.2.5. Other Neonatal Care Equipment
-
1.1. Fetal Care Equipment
Infant Phototherapy Devices Market Segmentation By Geography
- 1. Germany
- 2. United Kingdom
- 3. France
- 4. Italy
- 5. Spain
- 6. Rest of Europe
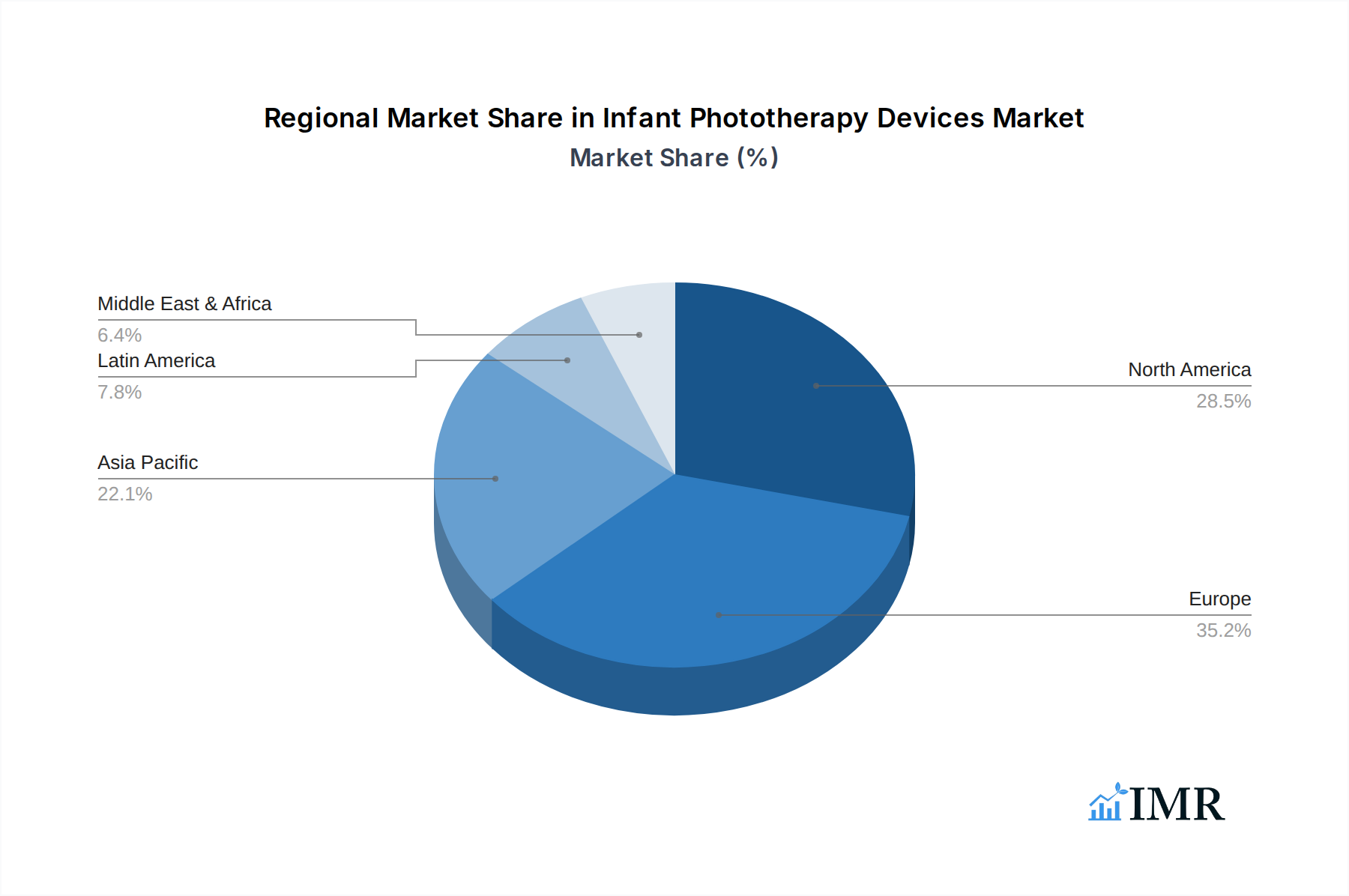
Infant Phototherapy Devices Market Regional Market Share

Geographic Coverage of Infant Phototherapy Devices Market
Infant Phototherapy Devices Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.69% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. IMR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product Type
- 5.1.1. Fetal Care Equipment
- 5.1.1.1. Fetal Dopplers
- 5.1.1.2. Fetal Magnetic Resonance Imaging (MRI) Devices
- 5.1.1.3. Ultrasound Devices
- 5.1.1.4. Fetal Pulse Oximeters
- 5.1.1.5. Other Fetal Care Equipment
- 5.1.2. Neonatal Care Equipment
- 5.1.2.1. Incubators
- 5.1.2.2. Neonatal Monitoring Devices
- 5.1.2.3. Phototherapy Equipment
- 5.1.2.4. Respiratory Assistance and Monitoring Devices
- 5.1.2.5. Other Neonatal Care Equipment
- 5.1.1. Fetal Care Equipment
- 5.2. Market Analysis, Insights and Forecast - by Region
- 5.2.1. Germany
- 5.2.2. United Kingdom
- 5.2.3. France
- 5.2.4. Italy
- 5.2.5. Spain
- 5.2.6. Rest of Europe
- 5.1. Market Analysis, Insights and Forecast - by Product Type
- 6. Global Infant Phototherapy Devices Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product Type
- 6.1.1. Fetal Care Equipment
- 6.1.1.1. Fetal Dopplers
- 6.1.1.2. Fetal Magnetic Resonance Imaging (MRI) Devices
- 6.1.1.3. Ultrasound Devices
- 6.1.1.4. Fetal Pulse Oximeters
- 6.1.1.5. Other Fetal Care Equipment
- 6.1.2. Neonatal Care Equipment
- 6.1.2.1. Incubators
- 6.1.2.2. Neonatal Monitoring Devices
- 6.1.2.3. Phototherapy Equipment
- 6.1.2.4. Respiratory Assistance and Monitoring Devices
- 6.1.2.5. Other Neonatal Care Equipment
- 6.1.1. Fetal Care Equipment
- 6.1. Market Analysis, Insights and Forecast - by Product Type
- 7. Germany Infant Phototherapy Devices Market Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Product Type
- 7.1.1. Fetal Care Equipment
- 7.1.1.1. Fetal Dopplers
- 7.1.1.2. Fetal Magnetic Resonance Imaging (MRI) Devices
- 7.1.1.3. Ultrasound Devices
- 7.1.1.4. Fetal Pulse Oximeters
- 7.1.1.5. Other Fetal Care Equipment
- 7.1.2. Neonatal Care Equipment
- 7.1.2.1. Incubators
- 7.1.2.2. Neonatal Monitoring Devices
- 7.1.2.3. Phototherapy Equipment
- 7.1.2.4. Respiratory Assistance and Monitoring Devices
- 7.1.2.5. Other Neonatal Care Equipment
- 7.1.1. Fetal Care Equipment
- 7.1. Market Analysis, Insights and Forecast - by Product Type
- 8. United Kingdom Infant Phototherapy Devices Market Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Product Type
- 8.1.1. Fetal Care Equipment
- 8.1.1.1. Fetal Dopplers
- 8.1.1.2. Fetal Magnetic Resonance Imaging (MRI) Devices
- 8.1.1.3. Ultrasound Devices
- 8.1.1.4. Fetal Pulse Oximeters
- 8.1.1.5. Other Fetal Care Equipment
- 8.1.2. Neonatal Care Equipment
- 8.1.2.1. Incubators
- 8.1.2.2. Neonatal Monitoring Devices
- 8.1.2.3. Phototherapy Equipment
- 8.1.2.4. Respiratory Assistance and Monitoring Devices
- 8.1.2.5. Other Neonatal Care Equipment
- 8.1.1. Fetal Care Equipment
- 8.1. Market Analysis, Insights and Forecast - by Product Type
- 9. France Infant Phototherapy Devices Market Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Product Type
- 9.1.1. Fetal Care Equipment
- 9.1.1.1. Fetal Dopplers
- 9.1.1.2. Fetal Magnetic Resonance Imaging (MRI) Devices
- 9.1.1.3. Ultrasound Devices
- 9.1.1.4. Fetal Pulse Oximeters
- 9.1.1.5. Other Fetal Care Equipment
- 9.1.2. Neonatal Care Equipment
- 9.1.2.1. Incubators
- 9.1.2.2. Neonatal Monitoring Devices
- 9.1.2.3. Phototherapy Equipment
- 9.1.2.4. Respiratory Assistance and Monitoring Devices
- 9.1.2.5. Other Neonatal Care Equipment
- 9.1.1. Fetal Care Equipment
- 9.1. Market Analysis, Insights and Forecast - by Product Type
- 10. Italy Infant Phototherapy Devices Market Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Product Type
- 10.1.1. Fetal Care Equipment
- 10.1.1.1. Fetal Dopplers
- 10.1.1.2. Fetal Magnetic Resonance Imaging (MRI) Devices
- 10.1.1.3. Ultrasound Devices
- 10.1.1.4. Fetal Pulse Oximeters
- 10.1.1.5. Other Fetal Care Equipment
- 10.1.2. Neonatal Care Equipment
- 10.1.2.1. Incubators
- 10.1.2.2. Neonatal Monitoring Devices
- 10.1.2.3. Phototherapy Equipment
- 10.1.2.4. Respiratory Assistance and Monitoring Devices
- 10.1.2.5. Other Neonatal Care Equipment
- 10.1.1. Fetal Care Equipment
- 10.1. Market Analysis, Insights and Forecast - by Product Type
- 11. Spain Infant Phototherapy Devices Market Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Product Type
- 11.1.1. Fetal Care Equipment
- 11.1.1.1. Fetal Dopplers
- 11.1.1.2. Fetal Magnetic Resonance Imaging (MRI) Devices
- 11.1.1.3. Ultrasound Devices
- 11.1.1.4. Fetal Pulse Oximeters
- 11.1.1.5. Other Fetal Care Equipment
- 11.1.2. Neonatal Care Equipment
- 11.1.2.1. Incubators
- 11.1.2.2. Neonatal Monitoring Devices
- 11.1.2.3. Phototherapy Equipment
- 11.1.2.4. Respiratory Assistance and Monitoring Devices
- 11.1.2.5. Other Neonatal Care Equipment
- 11.1.1. Fetal Care Equipment
- 11.1. Market Analysis, Insights and Forecast - by Product Type
- 12. Rest of Europe Infant Phototherapy Devices Market Analysis, Insights and Forecast, 2020-2032
- 12.1. Market Analysis, Insights and Forecast - by Product Type
- 12.1.1. Fetal Care Equipment
- 12.1.1.1. Fetal Dopplers
- 12.1.1.2. Fetal Magnetic Resonance Imaging (MRI) Devices
- 12.1.1.3. Ultrasound Devices
- 12.1.1.4. Fetal Pulse Oximeters
- 12.1.1.5. Other Fetal Care Equipment
- 12.1.2. Neonatal Care Equipment
- 12.1.2.1. Incubators
- 12.1.2.2. Neonatal Monitoring Devices
- 12.1.2.3. Phototherapy Equipment
- 12.1.2.4. Respiratory Assistance and Monitoring Devices
- 12.1.2.5. Other Neonatal Care Equipment
- 12.1.1. Fetal Care Equipment
- 12.1. Market Analysis, Insights and Forecast - by Product Type
- 13. Competitive Analysis
- 13.1. Company Profiles
- 13.1.1 F Hoffmann-La Roche Ltd
- 13.1.1.1. Company Overview
- 13.1.1.2. Products
- 13.1.1.3. Company Financials
- 13.1.1.4. SWOT Analysis
- 13.1.2 GE Company (GE Healthcare)
- 13.1.2.1. Company Overview
- 13.1.2.2. Products
- 13.1.2.3. Company Financials
- 13.1.2.4. SWOT Analysis
- 13.1.3 Atom Medical Corporation
- 13.1.3.1. Company Overview
- 13.1.3.2. Products
- 13.1.3.3. Company Financials
- 13.1.3.4. SWOT Analysis
- 13.1.4 Koninklijke Philips N V
- 13.1.4.1. Company Overview
- 13.1.4.2. Products
- 13.1.4.3. Company Financials
- 13.1.4.4. SWOT Analysis
- 13.1.5 Medtronic PLC
- 13.1.5.1. Company Overview
- 13.1.5.2. Products
- 13.1.5.3. Company Financials
- 13.1.5.4. SWOT Analysis
- 13.1.6 Siemens Healthineers AG
- 13.1.6.1. Company Overview
- 13.1.6.2. Products
- 13.1.6.3. Company Financials
- 13.1.6.4. SWOT Analysis
- 13.1.7 Gentinge AB
- 13.1.7.1. Company Overview
- 13.1.7.2. Products
- 13.1.7.3. Company Financials
- 13.1.7.4. SWOT Analysis
- 13.1.8 Becton Dickinson and Company
- 13.1.8.1. Company Overview
- 13.1.8.2. Products
- 13.1.8.3. Company Financials
- 13.1.8.4. SWOT Analysis
- 13.1.9 Dragerwerk AG & Co KGaA
- 13.1.9.1. Company Overview
- 13.1.9.2. Products
- 13.1.9.3. Company Financials
- 13.1.9.4. SWOT Analysis
- 13.1.10 Fisher & Paykel Healthcare Ltd
- 13.1.10.1. Company Overview
- 13.1.10.2. Products
- 13.1.10.3. Company Financials
- 13.1.10.4. SWOT Analysis
- 13.1.1 F Hoffmann-La Roche Ltd
- 13.2. Market Entropy
- 13.2.1 Company's Key Areas Served
- 13.2.2 Recent Developments
- 13.3. Company Market Share Analysis 2025
- 13.3.1 Top 5 Companies Market Share Analysis
- 13.3.2 Top 3 Companies Market Share Analysis
- 13.4. List of Potential Customers
- 14. Research Methodology
List of Figures
- Figure 1: Global Infant Phototherapy Devices Market Revenue Breakdown (Million, %) by Region 2025 & 2033
- Figure 2: Global Infant Phototherapy Devices Market Volume Breakdown (K Unit, %) by Region 2025 & 2033
- Figure 3: Germany Infant Phototherapy Devices Market Revenue (Million), by Product Type 2025 & 2033
- Figure 4: Germany Infant Phototherapy Devices Market Volume (K Unit), by Product Type 2025 & 2033
- Figure 5: Germany Infant Phototherapy Devices Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 6: Germany Infant Phototherapy Devices Market Volume Share (%), by Product Type 2025 & 2033
- Figure 7: Germany Infant Phototherapy Devices Market Revenue (Million), by Country 2025 & 2033
- Figure 8: Germany Infant Phototherapy Devices Market Volume (K Unit), by Country 2025 & 2033
- Figure 9: Germany Infant Phototherapy Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 10: Germany Infant Phototherapy Devices Market Volume Share (%), by Country 2025 & 2033
- Figure 11: United Kingdom Infant Phototherapy Devices Market Revenue (Million), by Product Type 2025 & 2033
- Figure 12: United Kingdom Infant Phototherapy Devices Market Volume (K Unit), by Product Type 2025 & 2033
- Figure 13: United Kingdom Infant Phototherapy Devices Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 14: United Kingdom Infant Phototherapy Devices Market Volume Share (%), by Product Type 2025 & 2033
- Figure 15: United Kingdom Infant Phototherapy Devices Market Revenue (Million), by Country 2025 & 2033
- Figure 16: United Kingdom Infant Phototherapy Devices Market Volume (K Unit), by Country 2025 & 2033
- Figure 17: United Kingdom Infant Phototherapy Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 18: United Kingdom Infant Phototherapy Devices Market Volume Share (%), by Country 2025 & 2033
- Figure 19: France Infant Phototherapy Devices Market Revenue (Million), by Product Type 2025 & 2033
- Figure 20: France Infant Phototherapy Devices Market Volume (K Unit), by Product Type 2025 & 2033
- Figure 21: France Infant Phototherapy Devices Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 22: France Infant Phototherapy Devices Market Volume Share (%), by Product Type 2025 & 2033
- Figure 23: France Infant Phototherapy Devices Market Revenue (Million), by Country 2025 & 2033
- Figure 24: France Infant Phototherapy Devices Market Volume (K Unit), by Country 2025 & 2033
- Figure 25: France Infant Phototherapy Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 26: France Infant Phototherapy Devices Market Volume Share (%), by Country 2025 & 2033
- Figure 27: Italy Infant Phototherapy Devices Market Revenue (Million), by Product Type 2025 & 2033
- Figure 28: Italy Infant Phototherapy Devices Market Volume (K Unit), by Product Type 2025 & 2033
- Figure 29: Italy Infant Phototherapy Devices Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 30: Italy Infant Phototherapy Devices Market Volume Share (%), by Product Type 2025 & 2033
- Figure 31: Italy Infant Phototherapy Devices Market Revenue (Million), by Country 2025 & 2033
- Figure 32: Italy Infant Phototherapy Devices Market Volume (K Unit), by Country 2025 & 2033
- Figure 33: Italy Infant Phototherapy Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 34: Italy Infant Phototherapy Devices Market Volume Share (%), by Country 2025 & 2033
- Figure 35: Spain Infant Phototherapy Devices Market Revenue (Million), by Product Type 2025 & 2033
- Figure 36: Spain Infant Phototherapy Devices Market Volume (K Unit), by Product Type 2025 & 2033
- Figure 37: Spain Infant Phototherapy Devices Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 38: Spain Infant Phototherapy Devices Market Volume Share (%), by Product Type 2025 & 2033
- Figure 39: Spain Infant Phototherapy Devices Market Revenue (Million), by Country 2025 & 2033
- Figure 40: Spain Infant Phototherapy Devices Market Volume (K Unit), by Country 2025 & 2033
- Figure 41: Spain Infant Phototherapy Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 42: Spain Infant Phototherapy Devices Market Volume Share (%), by Country 2025 & 2033
- Figure 43: Rest of Europe Infant Phototherapy Devices Market Revenue (Million), by Product Type 2025 & 2033
- Figure 44: Rest of Europe Infant Phototherapy Devices Market Volume (K Unit), by Product Type 2025 & 2033
- Figure 45: Rest of Europe Infant Phototherapy Devices Market Revenue Share (%), by Product Type 2025 & 2033
- Figure 46: Rest of Europe Infant Phototherapy Devices Market Volume Share (%), by Product Type 2025 & 2033
- Figure 47: Rest of Europe Infant Phototherapy Devices Market Revenue (Million), by Country 2025 & 2033
- Figure 48: Rest of Europe Infant Phototherapy Devices Market Volume (K Unit), by Country 2025 & 2033
- Figure 49: Rest of Europe Infant Phototherapy Devices Market Revenue Share (%), by Country 2025 & 2033
- Figure 50: Rest of Europe Infant Phototherapy Devices Market Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Infant Phototherapy Devices Market Revenue Million Forecast, by Product Type 2020 & 2033
- Table 2: Global Infant Phototherapy Devices Market Volume K Unit Forecast, by Product Type 2020 & 2033
- Table 3: Global Infant Phototherapy Devices Market Revenue Million Forecast, by Region 2020 & 2033
- Table 4: Global Infant Phototherapy Devices Market Volume K Unit Forecast, by Region 2020 & 2033
- Table 5: Global Infant Phototherapy Devices Market Revenue Million Forecast, by Product Type 2020 & 2033
- Table 6: Global Infant Phototherapy Devices Market Volume K Unit Forecast, by Product Type 2020 & 2033
- Table 7: Global Infant Phototherapy Devices Market Revenue Million Forecast, by Country 2020 & 2033
- Table 8: Global Infant Phototherapy Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 9: Global Infant Phototherapy Devices Market Revenue Million Forecast, by Product Type 2020 & 2033
- Table 10: Global Infant Phototherapy Devices Market Volume K Unit Forecast, by Product Type 2020 & 2033
- Table 11: Global Infant Phototherapy Devices Market Revenue Million Forecast, by Country 2020 & 2033
- Table 12: Global Infant Phototherapy Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 13: Global Infant Phototherapy Devices Market Revenue Million Forecast, by Product Type 2020 & 2033
- Table 14: Global Infant Phototherapy Devices Market Volume K Unit Forecast, by Product Type 2020 & 2033
- Table 15: Global Infant Phototherapy Devices Market Revenue Million Forecast, by Country 2020 & 2033
- Table 16: Global Infant Phototherapy Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 17: Global Infant Phototherapy Devices Market Revenue Million Forecast, by Product Type 2020 & 2033
- Table 18: Global Infant Phototherapy Devices Market Volume K Unit Forecast, by Product Type 2020 & 2033
- Table 19: Global Infant Phototherapy Devices Market Revenue Million Forecast, by Country 2020 & 2033
- Table 20: Global Infant Phototherapy Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 21: Global Infant Phototherapy Devices Market Revenue Million Forecast, by Product Type 2020 & 2033
- Table 22: Global Infant Phototherapy Devices Market Volume K Unit Forecast, by Product Type 2020 & 2033
- Table 23: Global Infant Phototherapy Devices Market Revenue Million Forecast, by Country 2020 & 2033
- Table 24: Global Infant Phototherapy Devices Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 25: Global Infant Phototherapy Devices Market Revenue Million Forecast, by Product Type 2020 & 2033
- Table 26: Global Infant Phototherapy Devices Market Volume K Unit Forecast, by Product Type 2020 & 2033
- Table 27: Global Infant Phototherapy Devices Market Revenue Million Forecast, by Country 2020 & 2033
- Table 28: Global Infant Phototherapy Devices Market Volume K Unit Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Infant Phototherapy Devices Market?
The projected CAGR is approximately 6.69%.
2. Which companies are prominent players in the Infant Phototherapy Devices Market?
Key companies in the market include F Hoffmann-La Roche Ltd, GE Company (GE Healthcare), Atom Medical Corporation, Koninklijke Philips N V, Medtronic PLC, Siemens Healthineers AG, Gentinge AB, Becton Dickinson and Company, Dragerwerk AG & Co KGaA, Fisher & Paykel Healthcare Ltd.
3. What are the main segments of the Infant Phototherapy Devices Market?
The market segments include Product Type.
4. Can you provide details about the market size?
The market size is estimated to be USD 1.88 Million as of 2022.
5. What are some drivers contributing to market growth?
Increasing Number of Preterm and Low-weight Births; Rise in Demand and Technological Advancement in Infant and Maternal Care Products.
6. What are the notable trends driving market growth?
Neonatal Monitoring Devices Sub-segment in Neonatal Care Equipment is Expected to Witness the Highest CAGR over the Forecast Period.
7. Are there any restraints impacting market growth?
High Cost Associated with Neonatal Care; Stringent Regulatory Policies for New Devices Approval.
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Infant Phototherapy Devices Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Infant Phototherapy Devices Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Infant Phototherapy Devices Market?
To stay informed about further developments, trends, and reports in the Infant Phototherapy Devices Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
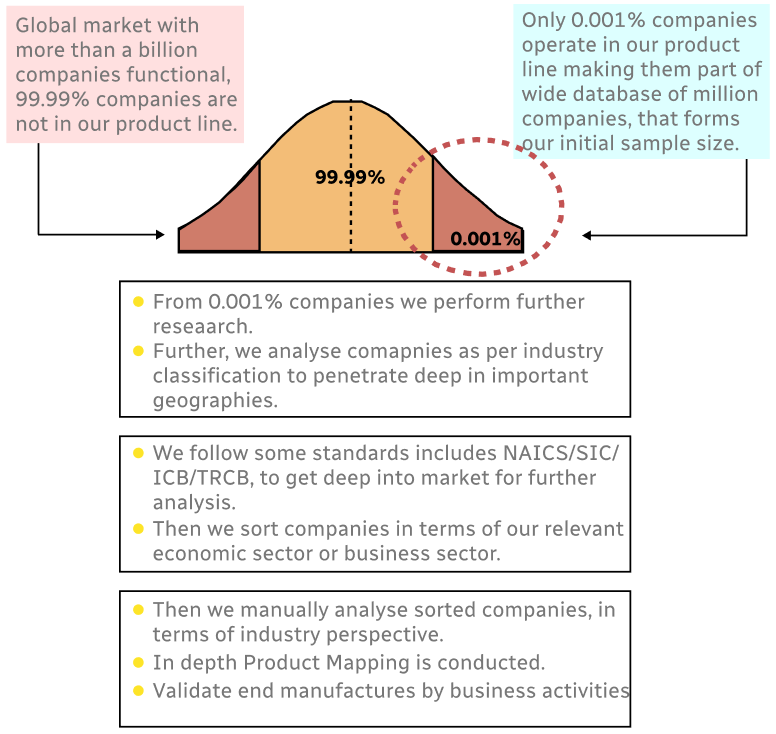
Step 1 - Identification of Relevant Samples Size from Population Database
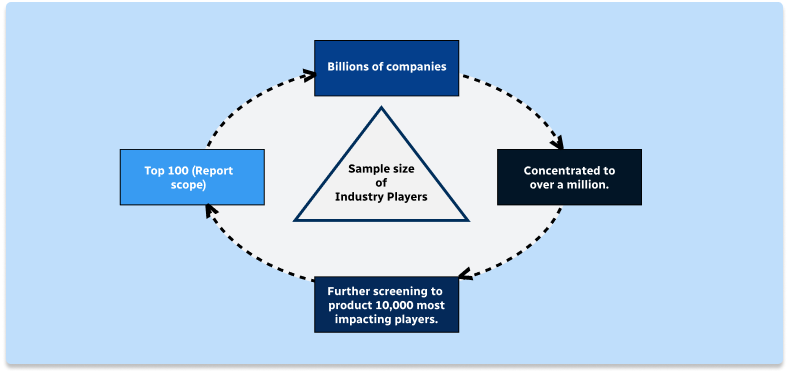
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
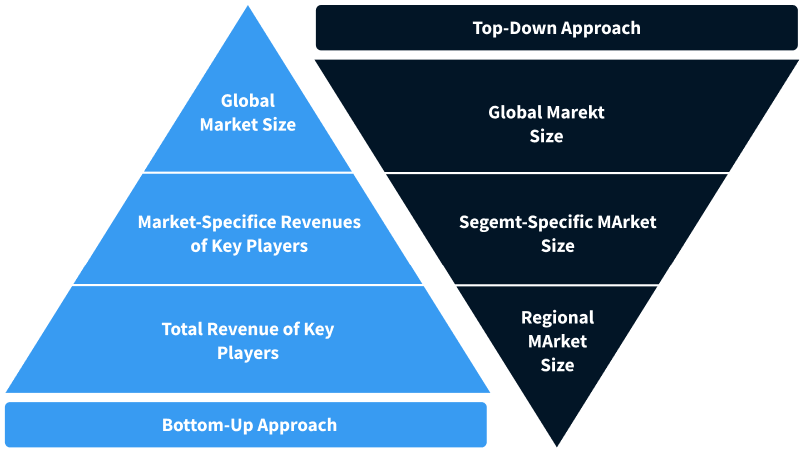
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
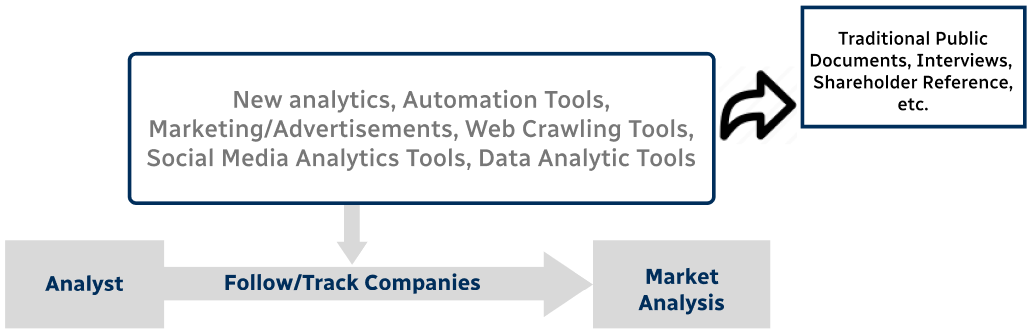
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


