Key Insights
The European Cardiac Rhythm Management (CRM) Devices Market is poised for robust growth, projected to reach approximately USD 30.05 billion in 2025 and expand at a Compound Annual Growth Rate (CAGR) of 6.55% through 2033. This significant market expansion is fueled by a confluence of escalating cardiovascular disease prevalence, a burgeoning elderly population susceptible to arrhythmias, and continuous technological advancements in CRM device capabilities. The increasing adoption of sophisticated implantable devices like Implantable Cardioverter Defibrillators (ICDs) and Cardiac Resynchronization Therapy (CRT) devices, designed to prevent sudden cardiac death and improve heart failure outcomes, is a primary driver. Furthermore, a growing awareness among healthcare providers and patients regarding the benefits of early diagnosis and intervention for cardiac rhythm disorders, coupled with favorable reimbursement policies across several European nations, are propelling market dynamism. The demand for minimally invasive procedures and remote monitoring capabilities integrated into these devices is also on the rise, reflecting a shift towards patient-centric and efficient cardiac care solutions.
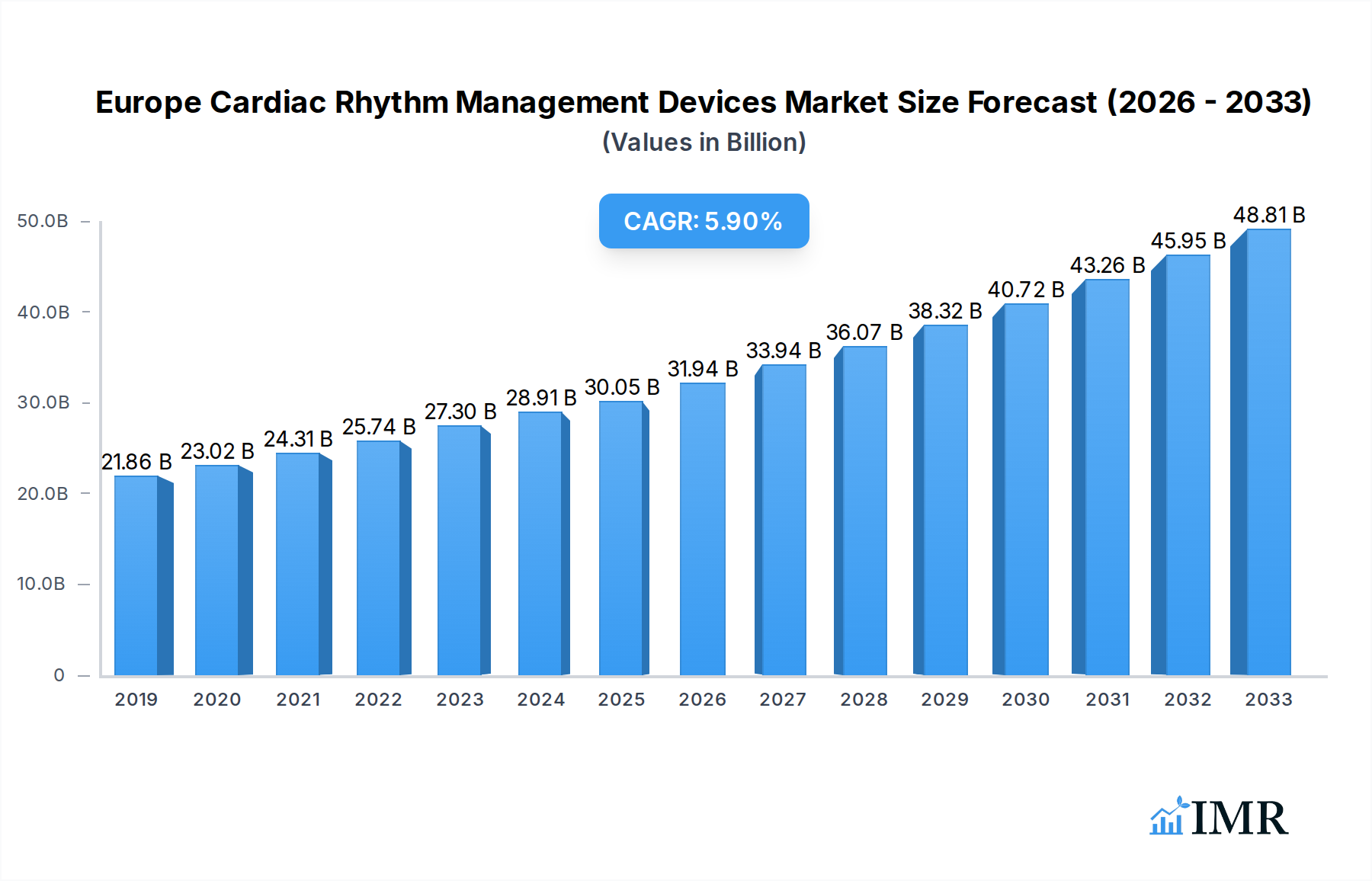
Europe Cardiac Rhythm Management Devices Market Market Size (In Billion)

The market's trajectory is further shaped by ongoing research and development efforts, leading to smaller, more advanced, and longer-lasting CRM devices with enhanced diagnostic features. Companies are focusing on developing wireless connectivity and artificial intelligence-powered analytics to optimize patient management and treatment efficacy. While the market exhibits strong growth potential, certain restraints exist, including the high cost of advanced CRM devices, which can limit accessibility in some regions or for certain patient demographics, and stringent regulatory approvals for novel technologies. However, the persistent burden of cardiovascular diseases, coupled with proactive healthcare initiatives and increasing investment in cardiovascular health research, is expected to outweigh these challenges. The market segments, including defibrillators and pacemakers, are all anticipated to witness substantial growth as healthcare systems prioritize the management of irregular heartbeats and related conditions.
Europe Cardiac Rhythm Management Devices Market Company Market Share

Here is a compelling, SEO-optimized report description for the Europe Cardiac Rhythm Management Devices Market, designed for maximum visibility and engagement with industry professionals:
Europe Cardiac Rhythm Management Devices Market: Comprehensive Growth Analysis & Forecast (2019-2033)
This in-depth report provides an unparalleled analysis of the Europe Cardiac Rhythm Management (CRM) Devices Market, dissecting intricate growth dynamics, technological advancements, and emerging opportunities. Covering the period from 2019 to 2033, with a base year of 2025 and a forecast period from 2025–2033, this research equips stakeholders with critical insights into market size, segmentation, competitive landscape, and future trajectory. Our analysis delves into parent and child markets, offering a holistic view of the CRM ecosystem, from advanced implantable devices to external solutions.
Key Market Segments Covered:
Key Players Analyzed: Schiller AG, Biotronik SE & Co KG, Shenzhen Mindray Biomedical Electronics Co., Ltd., Medtronic, Zoll Medical Corporation, LivaNova PLC, Cardinal Health, Abbott, Boston Scientific Corporation. (List is not exhaustive).
- Product:
- Defibrillators
- Implantable Cardioverter Defibrillators (ICDs)
- External Defibrillators (EDs)
- Pacemakers
- Implantable Pacemakers (ICPs)
- External Pacemakers
- Cardiac Resynchronization Therapy (CRT) Devices
- Cardiac Resynchronization Therapy Defibrillators (CRT-Ds)
- Cardiac Resynchronization Therapy Pacemakers (CRT-Ps)
- Defibrillators
Europe Cardiac Rhythm Management Devices Market Dynamics & Structure
The Europe Cardiac Rhythm Management (CRM) Devices Market is characterized by a moderately concentrated structure, with a few dominant players holding significant market share. This concentration is driven by high barriers to entry, including stringent regulatory approvals, substantial R&D investments, and established brand loyalty. Technological innovation remains a primary driver, with continuous advancements in miniaturization, battery life, wireless connectivity, and AI-driven diagnostics. The regulatory framework, particularly from the European Medicines Agency (EMA) and individual national health authorities, plays a crucial role in shaping market access and product development, often requiring extensive clinical trials. The market grapples with competitive product substitutes, such as advancements in drug therapies for certain arrhythmias and the potential for non-invasive treatments, though CRM devices remain essential for severe conditions. End-user demographics are influenced by an aging population, increasing prevalence of cardiovascular diseases, and rising awareness of cardiac health, leading to a growing demand for CRM solutions. Mergers and acquisitions (M&A) are a notable trend, as larger companies seek to acquire innovative technologies or expand their product portfolios to maintain competitive advantage. For instance, M&A activity in the CRM sector has seen a consistent volume of deals valued in the hundreds of millions to billions of dollars annually over the historical period, reflecting strategic consolidation.
- Market Concentration: Dominated by key players with a significant share.
- Technological Innovation: Driven by miniaturization, connectivity, and AI.
- Regulatory Framework: Influential, with stringent approval processes.
- Competitive Substitutes: Drug therapies and emerging non-invasive options.
- End-User Demographics: Aging population and rising CVD prevalence are key drivers.
- M&A Trends: Strategic consolidation to enhance market position and innovation.
Europe Cardiac Rhythm Management Devices Market Growth Trends & Insights
The Europe Cardiac Rhythm Management (CRM) Devices Market is poised for robust growth, projecting a Compound Annual Growth Rate (CAGR) of approximately 6.5% during the forecast period (2025–2033). This expansion is underpinned by an increasing market size, which is estimated to reach over $12 billion in 2025, with substantial growth anticipated over the next decade. Adoption rates for advanced CRM devices, particularly leadless pacemakers and subcutaneous implantable cardioverter-defibrillators (S-ICDs), are accelerating due to improved patient outcomes and minimally invasive implantation procedures. Technological disruptions, such as the integration of remote monitoring capabilities and sophisticated diagnostic algorithms, are transforming patient care and device efficacy. Consumer behavior is shifting towards proactive health management, with increased patient engagement in their cardiac health and a growing preference for less invasive and more convenient treatment options. The market penetration of CRM devices, though already significant, continues to rise as awareness of treatment options and their benefits expands across the European population. The aging demographic profile of Europe, coupled with the rising incidence of cardiovascular diseases like atrial fibrillation and heart failure, provides a sustained demand for CRM solutions. Furthermore, advancements in device longevity and remote patient management systems are contributing to better long-term patient management, thereby driving market expansion and enhancing the overall patient experience. The market size evolution is a direct reflection of these intertwined factors, demonstrating a dynamic and expanding landscape.
Dominant Regions, Countries, or Segments in Europe Cardiac Rhythm Management Devices Market
The Implantable Cardioverter Defibrillators (ICDs) segment is projected to be a dominant force within the Europe Cardiac Rhythm Management Devices Market, driven by the persistent high prevalence of sudden cardiac arrest (SCA) and an increasing adoption rate of advanced ICD technologies. Countries like Germany and France are expected to lead this dominance, owing to their well-established healthcare infrastructure, high healthcare expenditure, and proactive adoption of innovative medical technologies. Germany, with its robust clinical research ecosystem and a large, aging population, consistently leads in the implementation of advanced cardiac devices. France, on the other hand, benefits from comprehensive national health insurance coverage that supports the uptake of high-cost, high-benefit CRM devices.
Key drivers for the dominance of ICDs and these specific countries include:
- Economic Policies: Favorable reimbursement policies and government initiatives aimed at reducing the burden of cardiovascular diseases play a significant role. For instance, Germany's diagnosis-related groups (DRGs) system adequately compensates for the implantation of complex ICD devices, encouraging their use.
- Infrastructure: Advanced hospital facilities equipped with electrophysiology labs and skilled cardiologists are crucial for performing ICD implantations and complex cardiac procedures.
- Market Share: The ICD segment alone is estimated to capture over 35% of the total Europe CRM devices market share by 2025, with a projected growth of over 7% annually.
- Growth Potential: The continuous pipeline of next-generation ICDs, including those with enhanced sensing capabilities and smaller footprints, promises sustained growth and improved patient outcomes, further solidifying their market position.
- Technological Advancements: The development of subcutaneous ICDs (S-ICDs) and leadless ICDs is expanding the patient population eligible for defibrillation therapy, thus boosting market penetration.
- Awareness and Screening: Increased public awareness campaigns and improved cardiac screening programs lead to earlier diagnosis and intervention, thereby increasing the demand for life-saving devices like ICDs.
- Regulatory Support: Favorable regulatory pathways for novel ICD technologies in key European markets expedite their availability to patients.
The geographical dominance is concentrated in Western European nations due to their advanced healthcare systems and higher disposable incomes, allowing for greater investment in advanced medical devices.
Europe Cardiac Rhythm Management Devices Market Product Landscape
The Europe Cardiac Rhythm Management Devices Market product landscape is marked by significant innovation, particularly in implantable defibrillators and pacemakers. Implantable Cardioverter Defibrillators (ICDs) are continuously evolving with advancements in miniaturization, improved battery longevity, and enhanced diagnostic algorithms, offering greater efficacy and reduced patient burden. Implantable Pacemakers (ICPs) are witnessing a revolution with the advent of leadless pacing technology, offering minimally invasive implantation and reduced complications. Cardiac Resynchronization Therapy (CRT) devices, including CRT-Ds and CRT-Ps, are also seeing enhancements in their ability to effectively manage heart failure patients by synchronizing ventricular contractions. These product innovations are crucial for improving patient outcomes, increasing device longevity, and reducing the need for device replacements.
Key Drivers, Barriers & Challenges in Europe Cardiac Rhythm Management Devices Market
Key Drivers:
- Aging Population and Rising Cardiovascular Disease Prevalence: A growing elderly population and the increasing incidence of arrhythmias like atrial fibrillation and heart failure are the primary drivers for CRM devices. This demographic trend fuels a consistent demand for pacemakers and defibrillators.
- Technological Advancements: Continuous innovation in device miniaturization, wireless connectivity, remote monitoring, and AI-driven diagnostics enhances device efficacy and patient compliance, propelling market growth. The introduction of leadless pacemakers and subcutaneous ICDs represents significant technological leaps.
- Increasing Healthcare Expenditure and Reimbursement Policies: Higher healthcare spending across Europe and favorable reimbursement policies for advanced CRM devices in key countries encourage their adoption by healthcare providers and patients.
Barriers & Challenges:
- High Cost of Devices: The advanced nature of CRM devices often comes with a significant price tag, posing a challenge for some healthcare systems and patient affordability, despite reimbursement.
- Regulatory Hurdles and Approval Timelines: Stringent regulatory processes for new device approvals can be lengthy and costly, delaying market entry for innovative products.
- Shortage of Skilled Electrophysiologists: A potential shortage of trained professionals to perform complex implantation procedures can limit the widespread adoption and accessibility of CRM devices.
- Supply Chain Disruptions: Global supply chain vulnerabilities, as highlighted during recent geopolitical events, can impact the availability and cost of essential components for CRM device manufacturing. The potential impact on market growth can be a reduction of 1-2% in specific product segments due to component shortages.
Emerging Opportunities in Europe Cardiac Rhythm Management Devices Market
Emerging opportunities within the Europe Cardiac Rhythm Management Devices Market lie in the expanding application of remote patient monitoring and the development of AI-powered predictive diagnostics. As connectivity improves across Europe, devices that offer seamless data transmission to healthcare providers can lead to earlier intervention and better management of chronic cardiac conditions. The untapped potential in less developed Eastern European markets, where healthcare infrastructure is improving, presents a significant growth avenue. Furthermore, the increasing focus on personalized medicine is driving the demand for CRM devices tailored to specific patient profiles and genetic predispositions, opening up niche market segments. The development of smaller, less invasive devices with extended battery life also caters to evolving consumer preferences for improved quality of life post-implantation.
Growth Accelerators in the Europe Cardiac Rhythm Management Devices Market Industry
The long-term growth of the Europe Cardiac Rhythm Management Devices Market is being significantly accelerated by breakthroughs in leadless and subcutaneous technology, which are making CRM interventions accessible to a wider patient population. Strategic partnerships between medical device manufacturers and technology companies are fostering the integration of advanced digital health solutions, including sophisticated remote monitoring platforms and data analytics. Furthermore, market expansion strategies, such as increased investment in emerging European economies and targeted outreach programs to educate both healthcare professionals and patients about the benefits of advanced CRM, are acting as potent growth accelerators. The focus on evidence-based medicine and the publication of positive clinical trial outcomes for novel devices also bolster confidence and drive adoption.
Key Players Shaping the Europe Cardiac Rhythm Management Devices Market Market
- Schiller AG
- Biotronik SE & Co KG
- Shenzhen Mindray Biomedical Electronics Co., Ltd.
- Medtronic
- Zoll Medical Corporation
- LivaNova PLC
- Cardinal Health
- Abbott
- Boston Scientific Corporation
Notable Milestones in Europe Cardiac Rhythm Management Devices Market Sector
- November 2022: Heart experts at University Hospital Southampton (UHS) successfully implanted a new leadless pacemaker defibrillator system as part of a global clinical trial, featuring the innovative EMPOWER leadless pacemaker and a subcutaneously implanted cardioverter-defibrillator (S-ICD). This milestone highlights progress in advanced, less invasive CRM solutions for patients at risk of sudden cardiac arrest.
- February 2022: Abbott reported successful patient implants of its dual-chamber leadless pacemaker system within its AVEIR DR i2i pivotal clinical study. This marked a significant technological advancement for leadless pacing, with the study enrolling up to 550 patients across approximately 80 sites, including in Europe, with 12-month monitoring post-implantation.
In-Depth Europe Cardiac Rhythm Management Devices Market Market Outlook
The future outlook for the Europe Cardiac Rhythm Management Devices Market is exceptionally promising, driven by a confluence of sustained demand, technological innovation, and evolving healthcare strategies. Growth accelerators such as the relentless advancement in leadless and subcutaneous device technologies are broadening patient eligibility and improving treatment paradigms. Strategic alliances and the integration of digital health platforms are enhancing patient monitoring and care management. Furthermore, a concerted effort to expand access in underpenetrated regions and a growing emphasis on value-based healthcare will continue to fuel market expansion. The market is expected to witness continued robust growth, driven by the increasing burden of cardiovascular diseases and the ongoing development of sophisticated, patient-centric CRM solutions, reaching an estimated market value of over $19 billion by 2033.
Europe Cardiac Rhythm Management Devices Market Segmentation
-
1. Product
-
1.1. Defibrillators
- 1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 1.1.2. External Defibrillators (ED)
-
1.2. Pacemakers
- 1.2.1. Implantable Pacemakers (ICPS)
- 1.2.2. External Pacemakers
-
1.3. Cardiac Resynchronization Therapy Devices
- 1.3.1. Cardiac
- 1.3.2. Cardiac
-
1.1. Defibrillators
Europe Cardiac Rhythm Management Devices Market Segmentation By Geography
- 1. Germany
- 2. United Kingdom
- 3. France
- 4. Italy
- 5. Spain
- 6. Rest of Europe
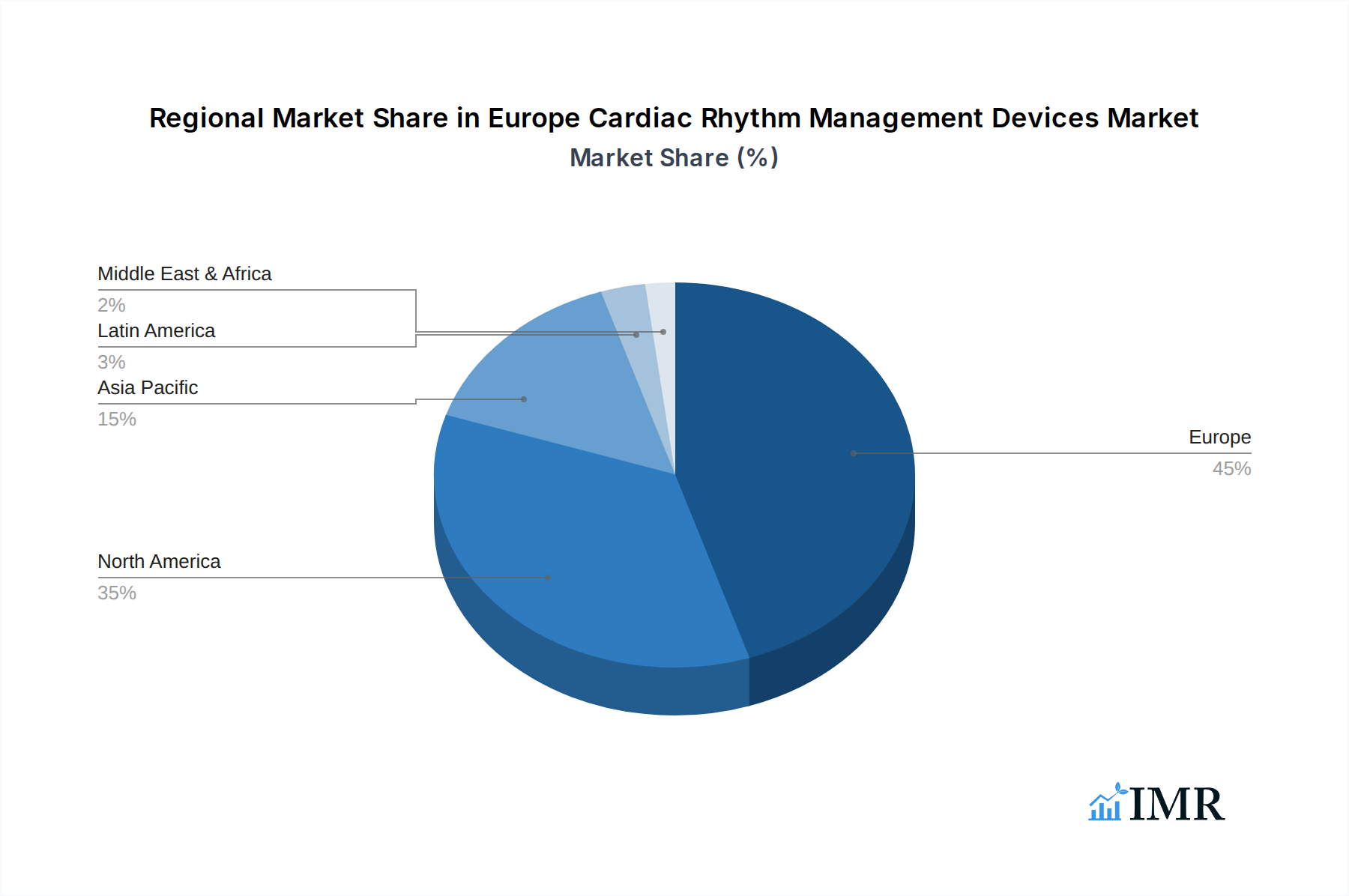
Europe Cardiac Rhythm Management Devices Market Regional Market Share

Geographic Coverage of Europe Cardiac Rhythm Management Devices Market
Europe Cardiac Rhythm Management Devices Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.55% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. IMR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product
- 5.1.1. Defibrillators
- 5.1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 5.1.1.2. External Defibrillators (ED)
- 5.1.2. Pacemakers
- 5.1.2.1. Implantable Pacemakers (ICPS)
- 5.1.2.2. External Pacemakers
- 5.1.3. Cardiac Resynchronization Therapy Devices
- 5.1.3.1. Cardiac
- 5.1.3.2. Cardiac
- 5.1.1. Defibrillators
- 5.2. Market Analysis, Insights and Forecast - by Region
- 5.2.1. Germany
- 5.2.2. United Kingdom
- 5.2.3. France
- 5.2.4. Italy
- 5.2.5. Spain
- 5.2.6. Rest of Europe
- 5.1. Market Analysis, Insights and Forecast - by Product
- 6. Europe Cardiac Rhythm Management Devices Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product
- 6.1.1. Defibrillators
- 6.1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 6.1.1.2. External Defibrillators (ED)
- 6.1.2. Pacemakers
- 6.1.2.1. Implantable Pacemakers (ICPS)
- 6.1.2.2. External Pacemakers
- 6.1.3. Cardiac Resynchronization Therapy Devices
- 6.1.3.1. Cardiac
- 6.1.3.2. Cardiac
- 6.1.1. Defibrillators
- 6.1. Market Analysis, Insights and Forecast - by Product
- 7. Germany Europe Cardiac Rhythm Management Devices Market Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Product
- 7.1.1. Defibrillators
- 7.1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 7.1.1.2. External Defibrillators (ED)
- 7.1.2. Pacemakers
- 7.1.2.1. Implantable Pacemakers (ICPS)
- 7.1.2.2. External Pacemakers
- 7.1.3. Cardiac Resynchronization Therapy Devices
- 7.1.3.1. Cardiac
- 7.1.3.2. Cardiac
- 7.1.1. Defibrillators
- 7.1. Market Analysis, Insights and Forecast - by Product
- 8. United Kingdom Europe Cardiac Rhythm Management Devices Market Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Product
- 8.1.1. Defibrillators
- 8.1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 8.1.1.2. External Defibrillators (ED)
- 8.1.2. Pacemakers
- 8.1.2.1. Implantable Pacemakers (ICPS)
- 8.1.2.2. External Pacemakers
- 8.1.3. Cardiac Resynchronization Therapy Devices
- 8.1.3.1. Cardiac
- 8.1.3.2. Cardiac
- 8.1.1. Defibrillators
- 8.1. Market Analysis, Insights and Forecast - by Product
- 9. France Europe Cardiac Rhythm Management Devices Market Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Product
- 9.1.1. Defibrillators
- 9.1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 9.1.1.2. External Defibrillators (ED)
- 9.1.2. Pacemakers
- 9.1.2.1. Implantable Pacemakers (ICPS)
- 9.1.2.2. External Pacemakers
- 9.1.3. Cardiac Resynchronization Therapy Devices
- 9.1.3.1. Cardiac
- 9.1.3.2. Cardiac
- 9.1.1. Defibrillators
- 9.1. Market Analysis, Insights and Forecast - by Product
- 10. Italy Europe Cardiac Rhythm Management Devices Market Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Product
- 10.1.1. Defibrillators
- 10.1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 10.1.1.2. External Defibrillators (ED)
- 10.1.2. Pacemakers
- 10.1.2.1. Implantable Pacemakers (ICPS)
- 10.1.2.2. External Pacemakers
- 10.1.3. Cardiac Resynchronization Therapy Devices
- 10.1.3.1. Cardiac
- 10.1.3.2. Cardiac
- 10.1.1. Defibrillators
- 10.1. Market Analysis, Insights and Forecast - by Product
- 11. Spain Europe Cardiac Rhythm Management Devices Market Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Product
- 11.1.1. Defibrillators
- 11.1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 11.1.1.2. External Defibrillators (ED)
- 11.1.2. Pacemakers
- 11.1.2.1. Implantable Pacemakers (ICPS)
- 11.1.2.2. External Pacemakers
- 11.1.3. Cardiac Resynchronization Therapy Devices
- 11.1.3.1. Cardiac
- 11.1.3.2. Cardiac
- 11.1.1. Defibrillators
- 11.1. Market Analysis, Insights and Forecast - by Product
- 12. Rest of Europe Europe Cardiac Rhythm Management Devices Market Analysis, Insights and Forecast, 2020-2032
- 12.1. Market Analysis, Insights and Forecast - by Product
- 12.1.1. Defibrillators
- 12.1.1.1. Implantable Cardioverter Defibrillators (ICDS)
- 12.1.1.2. External Defibrillators (ED)
- 12.1.2. Pacemakers
- 12.1.2.1. Implantable Pacemakers (ICPS)
- 12.1.2.2. External Pacemakers
- 12.1.3. Cardiac Resynchronization Therapy Devices
- 12.1.3.1. Cardiac
- 12.1.3.2. Cardiac
- 12.1.1. Defibrillators
- 12.1. Market Analysis, Insights and Forecast - by Product
- 13. Competitive Analysis
- 13.1. Company Profiles
- 13.1.1 SchillerAG
- 13.1.1.1. Company Overview
- 13.1.1.2. Products
- 13.1.1.3. Company Financials
- 13.1.1.4. SWOT Analysis
- 13.1.2 Biotronik SE & Co KG
- 13.1.2.1. Company Overview
- 13.1.2.2. Products
- 13.1.2.3. Company Financials
- 13.1.2.4. SWOT Analysis
- 13.1.3 Shenzhen Mindray Biomedical Electronics Co Ltd
- 13.1.3.1. Company Overview
- 13.1.3.2. Products
- 13.1.3.3. Company Financials
- 13.1.3.4. SWOT Analysis
- 13.1.4 Medtronic
- 13.1.4.1. Company Overview
- 13.1.4.2. Products
- 13.1.4.3. Company Financials
- 13.1.4.4. SWOT Analysis
- 13.1.5 Zoll Medical Corporation*List Not Exhaustive
- 13.1.5.1. Company Overview
- 13.1.5.2. Products
- 13.1.5.3. Company Financials
- 13.1.5.4. SWOT Analysis
- 13.1.6 LivaNova PLC
- 13.1.6.1. Company Overview
- 13.1.6.2. Products
- 13.1.6.3. Company Financials
- 13.1.6.4. SWOT Analysis
- 13.1.7 Cardinal Health
- 13.1.7.1. Company Overview
- 13.1.7.2. Products
- 13.1.7.3. Company Financials
- 13.1.7.4. SWOT Analysis
- 13.1.8 Abbott
- 13.1.8.1. Company Overview
- 13.1.8.2. Products
- 13.1.8.3. Company Financials
- 13.1.8.4. SWOT Analysis
- 13.1.9 Boston Scientific Corporation
- 13.1.9.1. Company Overview
- 13.1.9.2. Products
- 13.1.9.3. Company Financials
- 13.1.9.4. SWOT Analysis
- 13.1.1 SchillerAG
- 13.2. Market Entropy
- 13.2.1 Company's Key Areas Served
- 13.2.2 Recent Developments
- 13.3. Company Market Share Analysis 2025
- 13.3.1 Top 5 Companies Market Share Analysis
- 13.3.2 Top 3 Companies Market Share Analysis
- 13.4. List of Potential Customers
- 14. Research Methodology
List of Figures
- Figure 1: Europe Cardiac Rhythm Management Devices Market Revenue Breakdown (billion, %) by Product 2025 & 2033
- Figure 2: Europe Cardiac Rhythm Management Devices Market Share (%) by Company 2025
List of Tables
- Table 1: Europe Cardiac Rhythm Management Devices Market Revenue billion Forecast, by Product 2020 & 2033
- Table 2: Europe Cardiac Rhythm Management Devices Market Volume K Units Forecast, by Product 2020 & 2033
- Table 3: Europe Cardiac Rhythm Management Devices Market Revenue billion Forecast, by Region 2020 & 2033
- Table 4: Europe Cardiac Rhythm Management Devices Market Volume K Units Forecast, by Region 2020 & 2033
- Table 5: Europe Cardiac Rhythm Management Devices Market Revenue billion Forecast, by Product 2020 & 2033
- Table 6: Europe Cardiac Rhythm Management Devices Market Volume K Units Forecast, by Product 2020 & 2033
- Table 7: Europe Cardiac Rhythm Management Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 8: Europe Cardiac Rhythm Management Devices Market Volume K Units Forecast, by Country 2020 & 2033
- Table 9: Europe Cardiac Rhythm Management Devices Market Revenue billion Forecast, by Product 2020 & 2033
- Table 10: Europe Cardiac Rhythm Management Devices Market Volume K Units Forecast, by Product 2020 & 2033
- Table 11: Europe Cardiac Rhythm Management Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 12: Europe Cardiac Rhythm Management Devices Market Volume K Units Forecast, by Country 2020 & 2033
- Table 13: Europe Cardiac Rhythm Management Devices Market Revenue billion Forecast, by Product 2020 & 2033
- Table 14: Europe Cardiac Rhythm Management Devices Market Volume K Units Forecast, by Product 2020 & 2033
- Table 15: Europe Cardiac Rhythm Management Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 16: Europe Cardiac Rhythm Management Devices Market Volume K Units Forecast, by Country 2020 & 2033
- Table 17: Europe Cardiac Rhythm Management Devices Market Revenue billion Forecast, by Product 2020 & 2033
- Table 18: Europe Cardiac Rhythm Management Devices Market Volume K Units Forecast, by Product 2020 & 2033
- Table 19: Europe Cardiac Rhythm Management Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 20: Europe Cardiac Rhythm Management Devices Market Volume K Units Forecast, by Country 2020 & 2033
- Table 21: Europe Cardiac Rhythm Management Devices Market Revenue billion Forecast, by Product 2020 & 2033
- Table 22: Europe Cardiac Rhythm Management Devices Market Volume K Units Forecast, by Product 2020 & 2033
- Table 23: Europe Cardiac Rhythm Management Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 24: Europe Cardiac Rhythm Management Devices Market Volume K Units Forecast, by Country 2020 & 2033
- Table 25: Europe Cardiac Rhythm Management Devices Market Revenue billion Forecast, by Product 2020 & 2033
- Table 26: Europe Cardiac Rhythm Management Devices Market Volume K Units Forecast, by Product 2020 & 2033
- Table 27: Europe Cardiac Rhythm Management Devices Market Revenue billion Forecast, by Country 2020 & 2033
- Table 28: Europe Cardiac Rhythm Management Devices Market Volume K Units Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Europe Cardiac Rhythm Management Devices Market?
The projected CAGR is approximately 6.55%.
2. Which companies are prominent players in the Europe Cardiac Rhythm Management Devices Market?
Key companies in the market include SchillerAG, Biotronik SE & Co KG, Shenzhen Mindray Biomedical Electronics Co Ltd, Medtronic, Zoll Medical Corporation*List Not Exhaustive, LivaNova PLC, Cardinal Health, Abbott, Boston Scientific Corporation.
3. What are the main segments of the Europe Cardiac Rhythm Management Devices Market?
The market segments include Product.
4. Can you provide details about the market size?
The market size is estimated to be USD 30.05 billion as of 2022.
5. What are some drivers contributing to market growth?
Increasing Prevalence of Cardiovascular Disorders; Technological Advancements; Rise in the Use of Ambulatory and Home Services for Cardiac Monitoring.
6. What are the notable trends driving market growth?
External Defibrillator (ED) Segment is Expected to Hold a Significant Share During the Forecast Period.
7. Are there any restraints impacting market growth?
High Cost of Devices.
8. Can you provide examples of recent developments in the market?
November 2022: Heart experts at University Hospital Southampton (UHS) implanted a new leadless pacemaker defibrillator system to treat patients at risk of sudden cardiac arrest. The surgery is a component of a global clinical trial involving the implantation of the innovative leadless pacemaker EMPOWER and the subcutaneously implanted cardioverter-defibrillator (S-ICD).
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion and volume, measured in K Units.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Europe Cardiac Rhythm Management Devices Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Europe Cardiac Rhythm Management Devices Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Europe Cardiac Rhythm Management Devices Market?
To stay informed about further developments, trends, and reports in the Europe Cardiac Rhythm Management Devices Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
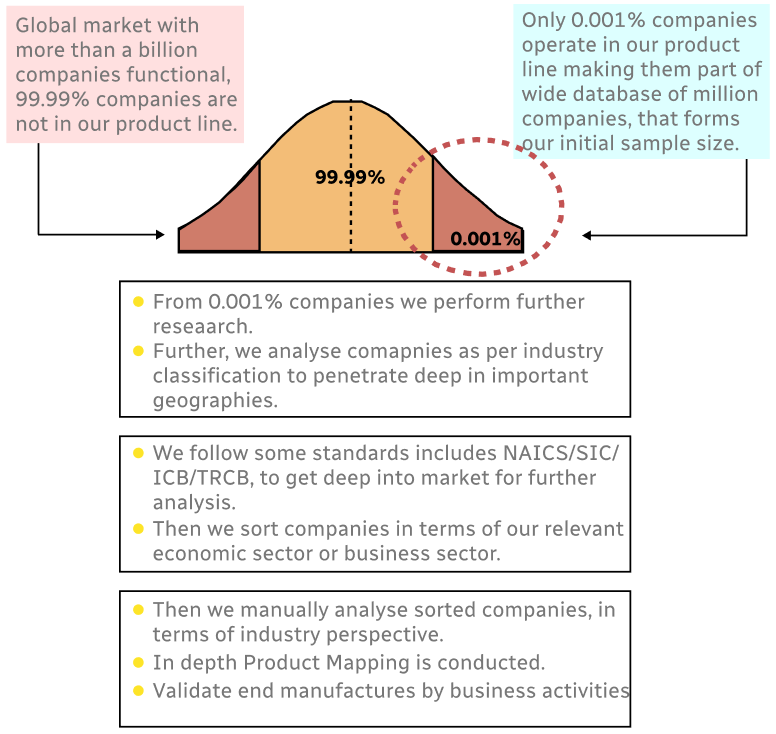
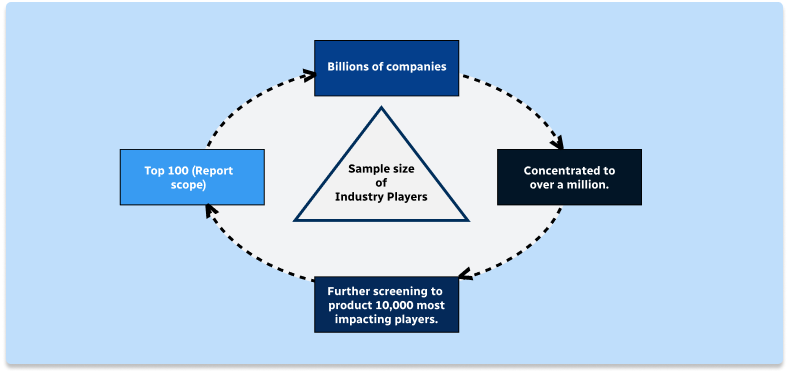
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
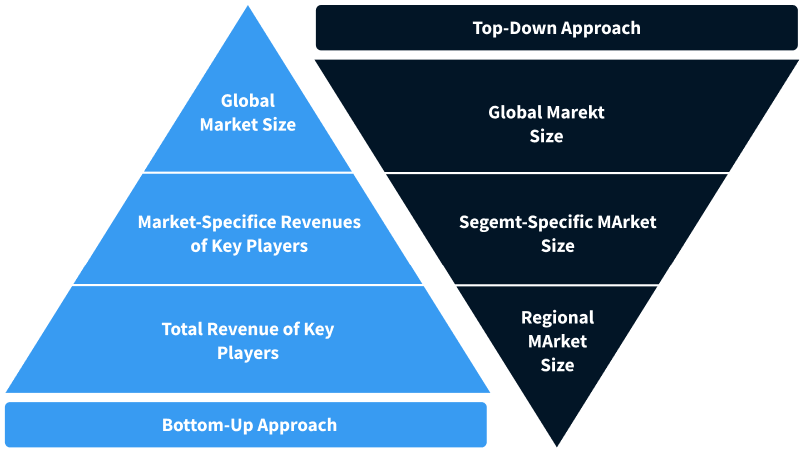
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
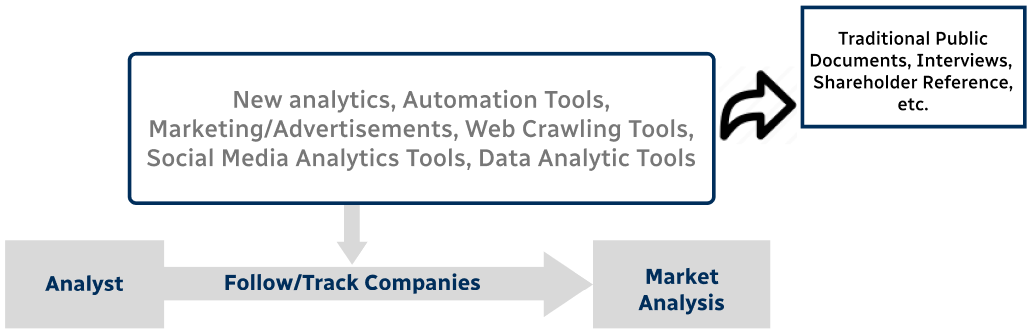
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


