Key Insights
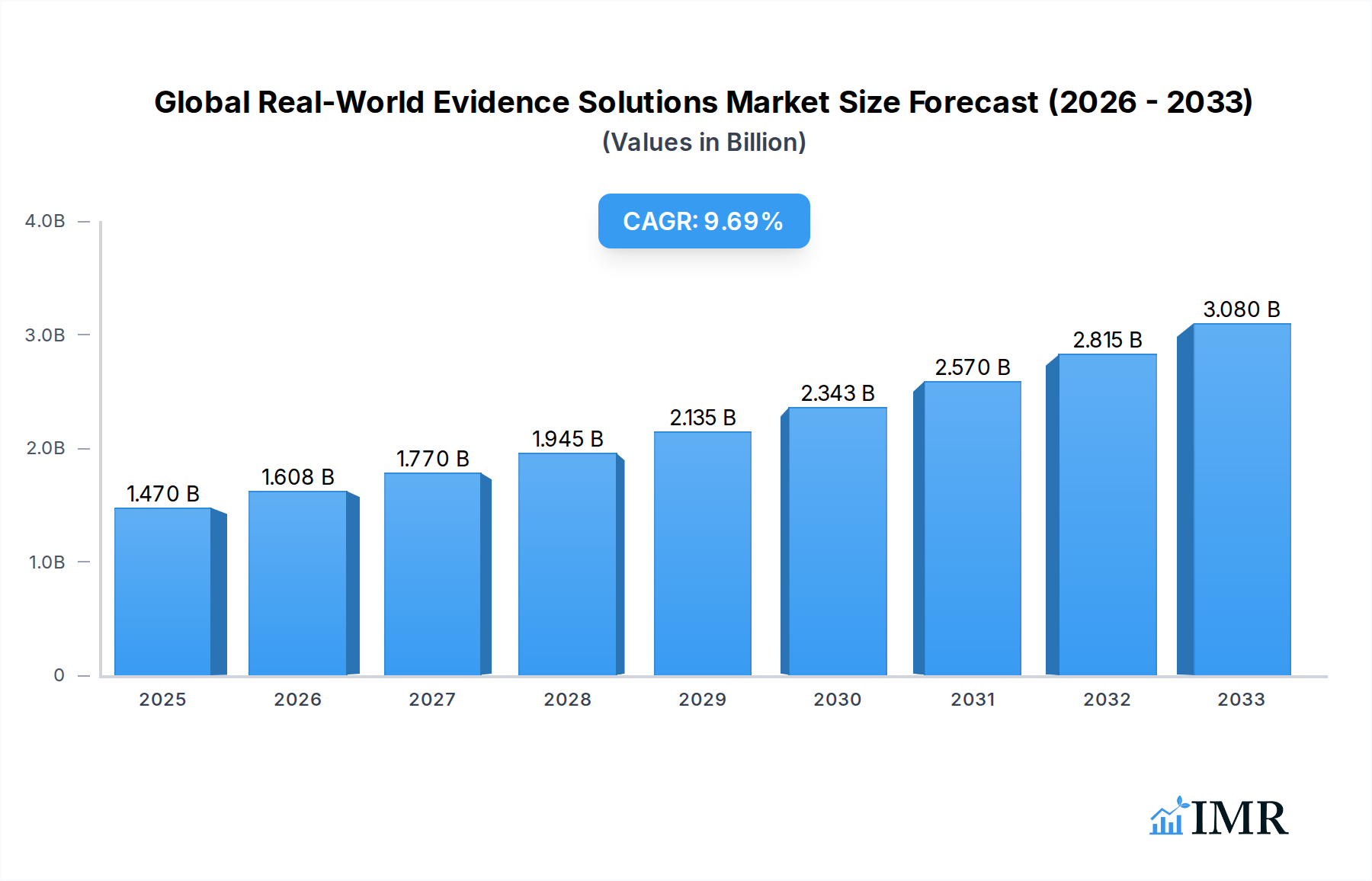
The Global Real-World Evidence (RWE) Solutions Market is poised for substantial expansion, projected to reach $1.47 Billion in 2025, with a robust Compound Annual Growth Rate (CAGR) of 9.40% throughout the forecast period of 2025-2033. This growth is fueled by a convergence of critical drivers, including the increasing demand for evidence-based decision-making in healthcare, the burgeoning volume of digital health data, and the imperative for pharmaceutical companies to demonstrate drug value and effectiveness in real-world settings. The shift towards value-based care models further incentivizes the adoption of RWE solutions, as payers and providers seek to optimize patient outcomes and control costs. Key market segments experiencing significant traction include the utilization of Claims Data and Clinical Settings Data, both vital for understanding patient journeys and treatment efficacy. Therapeutic areas like Oncology, Immunology, and Neurology are at the forefront, driving innovation and investment in RWE due to their complex disease patterns and the ongoing development of novel therapies.
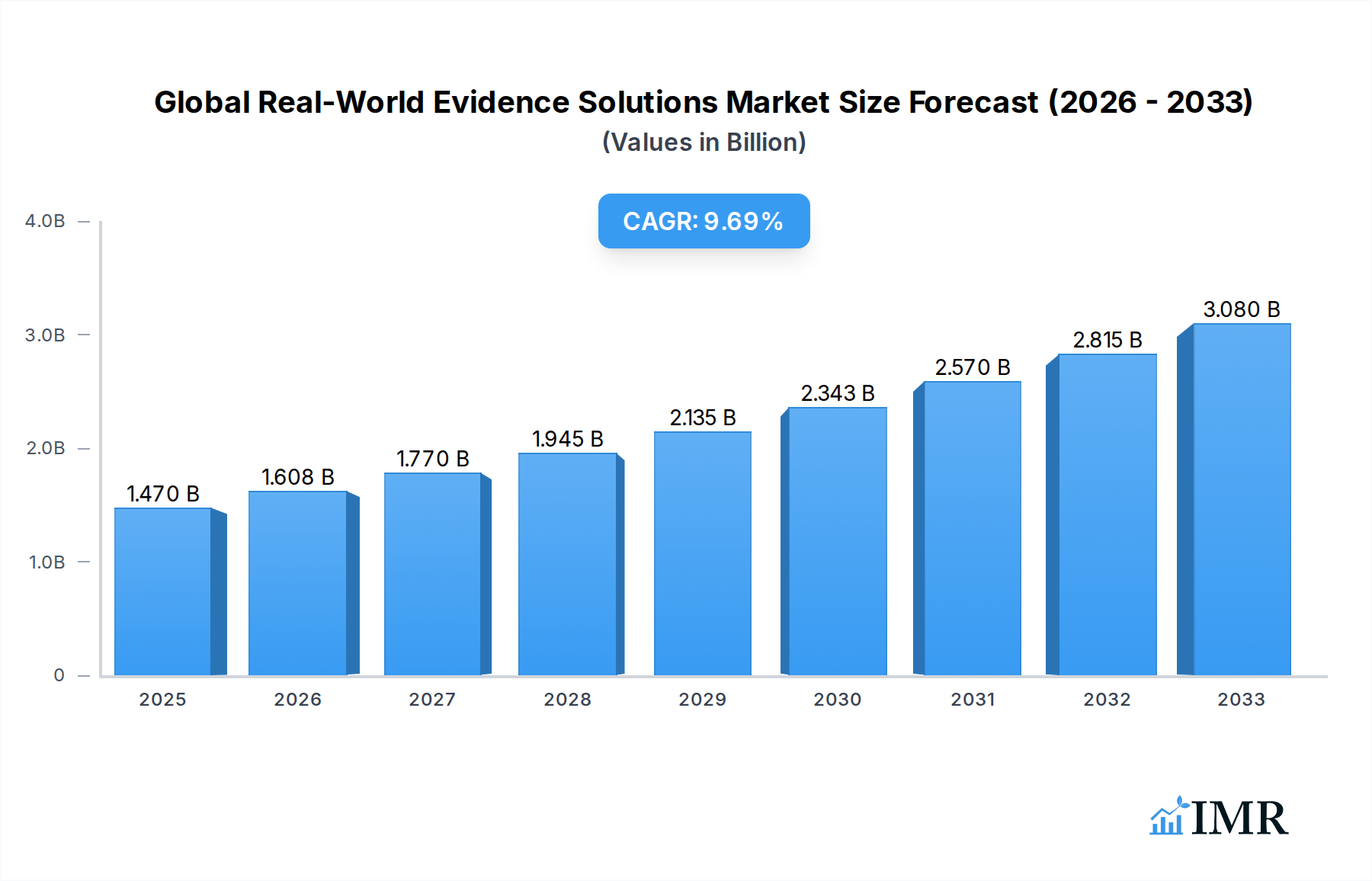
Global Real-World Evidence Solutions Market Market Size (In Billion)

The market is characterized by a dynamic competitive landscape with established players such as IQVIA, Oracle Corporation, and IBM alongside specialized RWE providers like Flatiron Health Inc. and Syneos Health. These companies are actively innovating and expanding their offerings to cater to the evolving needs of healthcare payers, providers, and pharmaceutical and medical device companies. While the RWE market demonstrates strong growth potential, certain restraints, such as data privacy concerns and the need for standardized data governance frameworks, require careful navigation. However, ongoing advancements in data analytics, artificial intelligence, and machine learning are continuously enhancing the capabilities and accessibility of RWE solutions, paving the way for more sophisticated and impactful applications in drug development, regulatory submissions, and post-market surveillance. The Asia Pacific region, particularly China and India, is emerging as a significant growth engine due to increasing healthcare expenditure and a growing focus on evidence generation.
Global Real-World Evidence Solutions Market Company Market Share

Global Real-World Evidence Solutions Market: Unlocking Insights for a Data-Driven Healthcare Future (2019-2033)
This comprehensive report provides an in-depth analysis of the Global Real-World Evidence Solutions Market, a critical sector enabling healthcare innovation and decision-making. Explore market dynamics, growth trajectories, dominant segments, and key players driving the adoption of real-world data (RWD) and real-world evidence (RWE) across the healthcare ecosystem. With a forecast period extending to 2033, this report offers unparalleled insights for stakeholders navigating the evolving landscape of evidence generation.
Key Market Segments Covered:
- Component: Claims Data, Clinical Settings Data, Patient-Powered Data, Pharmacy Data, Other Components
- Therapeutic Area: Oncology, Immunology, Neurology, Cardiovascular Disease, Other Therapeutic Areas
- End User: Healthcare Payers, Healthcare Providers, Pharmaceutical & Medical Device Companies, Other End Users
- Parent Markets: Real-World Data Market, Healthcare Analytics Market
- Child Markets: RWE Analytics Market, RWE Data Management Market, RWE Consulting Services Market
Global Real-World Evidence Solutions Market Market Dynamics & Structure
The Global Real-World Evidence Solutions Market is characterized by intense innovation and strategic consolidation, driven by the increasing demand for evidence-based healthcare decisions. Market concentration is moderate, with major players like IQVIA, Syneos Health, and Oracle Corporation holding significant shares, yet fostering an environment for specialized RWE solution providers and emerging tech companies. Technological innovation is a primary driver, fueled by advancements in AI, machine learning, and advanced analytics for processing vast datasets. Regulatory frameworks, such as those from the FDA and EMA, are increasingly encouraging the use of RWE in drug development and regulatory submissions, further stimulating market growth. Competitive product substitutes exist, primarily traditional clinical trial data, but RWE offers cost-effectiveness and broader applicability. End-user demographics are diverse, with pharmaceutical and medical device companies, healthcare payers, and providers all actively leveraging RWE for various applications, including treatment effectiveness, patient outcomes, and market access. Mergers and acquisitions (M&A) remain a significant trend, as larger entities acquire specialized RWE capabilities or data assets to expand their service portfolios. For instance, in the historical period (2019-2024), there were xx M&A deals valued at an estimated $XXX million focused on enhancing RWE data integration and analytical tools. Barriers to innovation include data privacy concerns, interoperability challenges across different data sources, and the need for robust data validation and governance.
Global Real-World Evidence Solutions Market Growth Trends & Insights
The Global Real-World Evidence Solutions Market is poised for substantial expansion, projected to grow at a Compound Annual Growth Rate (CAGR) of approximately 12.5% between 2025 and 2033, reaching an estimated $XXX billion by the end of the forecast period. This robust growth is underpinned by several key trends. The escalating demand for personalized medicine and precision therapies is a significant catalyst, as RWE provides the granular insights needed to tailor treatments to individual patient profiles and disease subtypes. Furthermore, the increasing pressure on healthcare systems to demonstrate value and cost-effectiveness is driving the adoption of RWE in health technology assessments (HTA) and reimbursement decisions.
Technological disruptions, particularly in artificial intelligence (AI) and machine learning (ML), are revolutionizing how RWE is collected, processed, and analyzed. These technologies enable more sophisticated pattern recognition, predictive modeling, and the identification of novel biomarkers and treatment pathways. The shift towards value-based care models globally further incentivizes the use of RWE to measure patient outcomes and provider performance, thereby optimizing resource allocation and improving overall healthcare quality.
Consumer behavior is also evolving, with patients becoming more engaged in their health journeys and actively seeking information about treatment options and outcomes. This growing patient empowerment, coupled with the increasing availability of patient-generated health data (PGHD) through wearables and mobile health applications, contributes to a richer and more comprehensive RWE landscape. The Real-World Evidence Solutions Market is therefore not just growing; it's becoming more sophisticated and integral to every facet of healthcare, from early-stage drug discovery to post-market surveillance and patient support. Adoption rates for RWE solutions are accelerating across all end-user segments, indicating a strong market penetration driven by tangible benefits like reduced R&D costs, faster time-to-market for new therapies, and improved clinical trial design.
Dominant Regions, Countries, or Segments in Global Real-World Evidence Solutions Market
The Global Real-World Evidence Solutions Market exhibits distinct regional dominance and segment leadership, driven by a confluence of factors including regulatory advancements, healthcare infrastructure, and the prevalence of specific therapeutic areas.
North America currently stands as the leading region, consistently driving market growth. This dominance is attributed to a mature healthcare ecosystem with high adoption rates of advanced technologies, robust data infrastructure, and a proactive stance from regulatory bodies like the U.S. Food and Drug Administration (FDA) in accepting RWE for decision-making. The significant investment in R&D by pharmaceutical and biotechnology companies in the United States further bolsters the demand for RWE solutions.
Within the Component segment, Claims Data has historically been a dominant source of RWE due to its widespread availability and extensive historical coverage across large patient populations. However, Clinical Settings Data, encompassing electronic health records (EHRs) and clinical registries, is rapidly gaining traction, offering richer, more granular clinical detail. Patient-Powered Data, though nascent, is experiencing exponential growth, driven by the proliferation of wearables and patient-reported outcome (PRO) platforms, offering unique insights into daily health and treatment adherence.
In terms of Therapeutic Areas, Oncology leads the market, owing to the complexity of cancer treatments, the need for evidence on treatment efficacy in diverse patient populations, and the rapid pace of innovation in cancer therapeutics. The high cost of oncology drugs and the imperative to demonstrate value for reimbursement further amplify the demand for RWE in this segment. Cardiovascular Disease and Neurology also represent significant markets due to the chronic nature of these conditions and the ongoing need for real-world effectiveness data.
The End User segment is heavily influenced by Pharmaceutical & Medical Device Companies, who utilize RWE extensively for drug development, post-market surveillance, comparative effectiveness research, and market access strategies. Healthcare Providers are increasingly adopting RWE for clinical decision support, quality improvement initiatives, and operational efficiency. Healthcare Payers leverage RWE for formulary decisions, risk stratification, and value-based contracting.
The growth potential in regions like Europe is significant, driven by initiatives from the European Medicines Agency (EMA) to integrate RWE into regulatory processes and the increasing focus on real-world effectiveness and safety across member states. Emerging markets in Asia-Pacific are also demonstrating rapid growth, fueled by expanding healthcare access, increasing healthcare expenditure, and a growing R&D focus, particularly in countries like China and India.
Global Real-World Evidence Solutions Market Product Landscape
The Global Real-World Evidence Solutions Market product landscape is defined by a sophisticated suite of offerings designed to capture, manage, analyze, and report on real-world data. These solutions range from advanced data analytics platforms leveraging AI and machine learning for predictive insights and causal inference, to comprehensive data management systems ensuring data integrity, privacy, and regulatory compliance. Specialized modules for patient cohort identification, synthetic control arm generation, and comparative effectiveness research are increasingly prevalent. Unique selling propositions often lie in the breadth and depth of integrated data sources, the speed and accuracy of insights delivery, and the ability to support diverse regulatory and research requirements. Technological advancements are focused on natural language processing (NLP) for unstructured data extraction from clinical notes, advanced visualization tools for intuitive data interpretation, and secure, cloud-based infrastructure for scalability and accessibility.
Key Drivers, Barriers & Challenges in Global Real-World Evidence Solutions Market
Key Drivers:
- Increasing Demand for Value-Based Healthcare: The global shift towards demonstrating treatment effectiveness and cost-efficiency mandates the use of RWE to inform decision-making for payers, providers, and manufacturers.
- Regulatory Acceptance: Growing recognition and acceptance of RWE by regulatory bodies like the FDA and EMA for drug approval, label expansion, and post-market surveillance is a significant market accelerant.
- Technological Advancements: Sophisticated AI, machine learning, and advanced analytics tools are enhancing the ability to extract meaningful insights from complex RWD.
- Cost and Time Efficiency: RWE solutions offer a more cost-effective and faster alternative or complement to traditional clinical trials for certain research objectives.
- Personalized Medicine & Precision Health: The drive for tailored treatments requires granular patient data and outcomes, which RWE effectively provides.
Barriers & Challenges:
- Data Quality and Standardization: Inconsistent data collection methods, data silos, and lack of interoperability across various data sources pose significant challenges to generating reliable RWE.
- Privacy and Security Concerns: Protecting patient privacy and ensuring the secure handling of sensitive health data in compliance with regulations like GDPR and HIPAA is paramount.
- Methodological Rigor and Bias: Ensuring the scientific validity and minimizing bias in RWE studies is crucial for regulatory acceptance and widespread adoption.
- Talent Gap: A shortage of skilled professionals with expertise in data science, biostatistics, epidemiology, and RWE methodology can hinder market growth.
- Regulatory Uncertainty: While regulatory acceptance is growing, evolving guidelines and the need for further clarity on specific RWE applications can create hurdles. The Global Real-World Evidence Solutions Market faces a substantial hurdle in achieving robust data standardization, with an estimated XX% of healthcare data still residing in unstructured formats, impacting the efficiency of data analysis and insight generation.
Emerging Opportunities in Global Real-World Evidence Solutions Market
Emerging opportunities in the Global Real-World Evidence Solutions Market are vast and transformative. The growing adoption of wearables and the Internet of Medical Things (IoMT) presents a significant avenue for capturing continuous, real-time patient-generated health data, offering unprecedented insights into disease progression and treatment response outside traditional clinical settings. The expansion of RWE applications into rare diseases is another burgeoning area, where traditional clinical trials are often challenging and expensive; RWE can help aggregate limited patient data for more robust understanding. Furthermore, the development of federated learning and privacy-preserving analytics techniques offers solutions to address data privacy concerns, enabling collaborative RWE research without direct data sharing. The increasing focus on health equity and understanding treatment outcomes across diverse patient populations also opens up new RWE applications.
Growth Accelerators in the Global Real-World Evidence Solutions Market Industry
Several catalysts are accelerating the growth of the Global Real-World Evidence Solutions Market. Technological breakthroughs, particularly in AI and natural language processing (NLP), are enabling more sophisticated analysis of unstructured clinical data, unlocking deeper insights. Strategic partnerships between technology providers, pharmaceutical companies, and academic institutions are fostering innovation and accelerating the development of robust RWE methodologies. Market expansion strategies, including the increasing penetration of RWE solutions in emerging economies and the development of tailored solutions for specific therapeutic areas, are also driving growth. The establishment of data consortia and collaborative research networks is further enhancing data accessibility and the reliability of RWE findings, creating a more fertile ground for evidence generation and application.
Key Players Shaping the Global Real-World Evidence Solutions Market Market
- Clinigen Group PLC
- Syneos Health
- Flatiron Health Inc
- Parexel International
- IQVIA
- SAS Institute
- Pharmaceutical Product Development (PPD Inc )
- Oracle Corporation
- PerkinElmer Inc
- IBM
- Icon PLC
Notable Milestones in Global Real-World Evidence Solutions Market Sector
- December 2021: EVERSANA signed an agreement with Janssen Research & Development LLC (Janssen) to drive evidence-based development of Janssen therapies, treatments, and patient support models, highlighting the growing strategic importance of RWE partnerships in drug development and patient care.
- October 2021: The Real-World Evidence Transparency Initiative launched the Real-World Evidence Registry to establish a culture of transparency for the analysis and reporting of Real-World Evidence in healthcare and health research. This initiative, a partnership between ISPOR, the International Society for Pharmacoepidemiology, the Duke-Margolis Center for Health Policy, and the National Pharmaceutical Council, signifies a crucial step towards enhancing the credibility and trustworthiness of RWE.
In-Depth Global Real-World Evidence Solutions Market Market Outlook
The Global Real-World Evidence Solutions Market is poised for sustained and significant growth, driven by an imperative for data-informed healthcare decisions across the entire value chain. Future market potential is anchored in the expanding applications of RWE in drug discovery, clinical trial optimization, regulatory submissions, and post-market surveillance. Strategic opportunities lie in the continued development of interoperable data platforms, advanced AI-driven analytics, and the integration of diverse data sources, including genomics and wearables. The increasing focus on personalized medicine and value-based care will further solidify RWE's role in demonstrating treatment efficacy and patient outcomes. Stakeholders can anticipate a dynamic market characterized by ongoing technological innovation, strategic collaborations, and a growing demand for evidence that accurately reflects the complexities of real-world healthcare.
Global Real-World Evidence Solutions Market Segmentation
-
1. Component
- 1.1. Claims Data
- 1.2. Clinical Settings Data
- 1.3. Patient-Powered Data
- 1.4. Pharmacy Data
- 1.5. Other Components
-
2. Therapeutic Area
- 2.1. Oncology
- 2.2. Immunology
- 2.3. Neurology
- 2.4. Cardiovascular Disease
- 2.5. Other Therapeutic Areas
-
3. End User
- 3.1. Healthcare Payers
- 3.2. Healthcare Providers
- 3.3. Pharmaceutical & Medical Device Companies
- 3.4. Other End Users
Global Real-World Evidence Solutions Market Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
- 4. Middle East
-
5. GCC
- 5.1. South Africa
- 5.2. Rest of Middle East
-
6. South America
- 6.1. Brazil
- 6.2. Argentina
- 6.3. Rest of South America
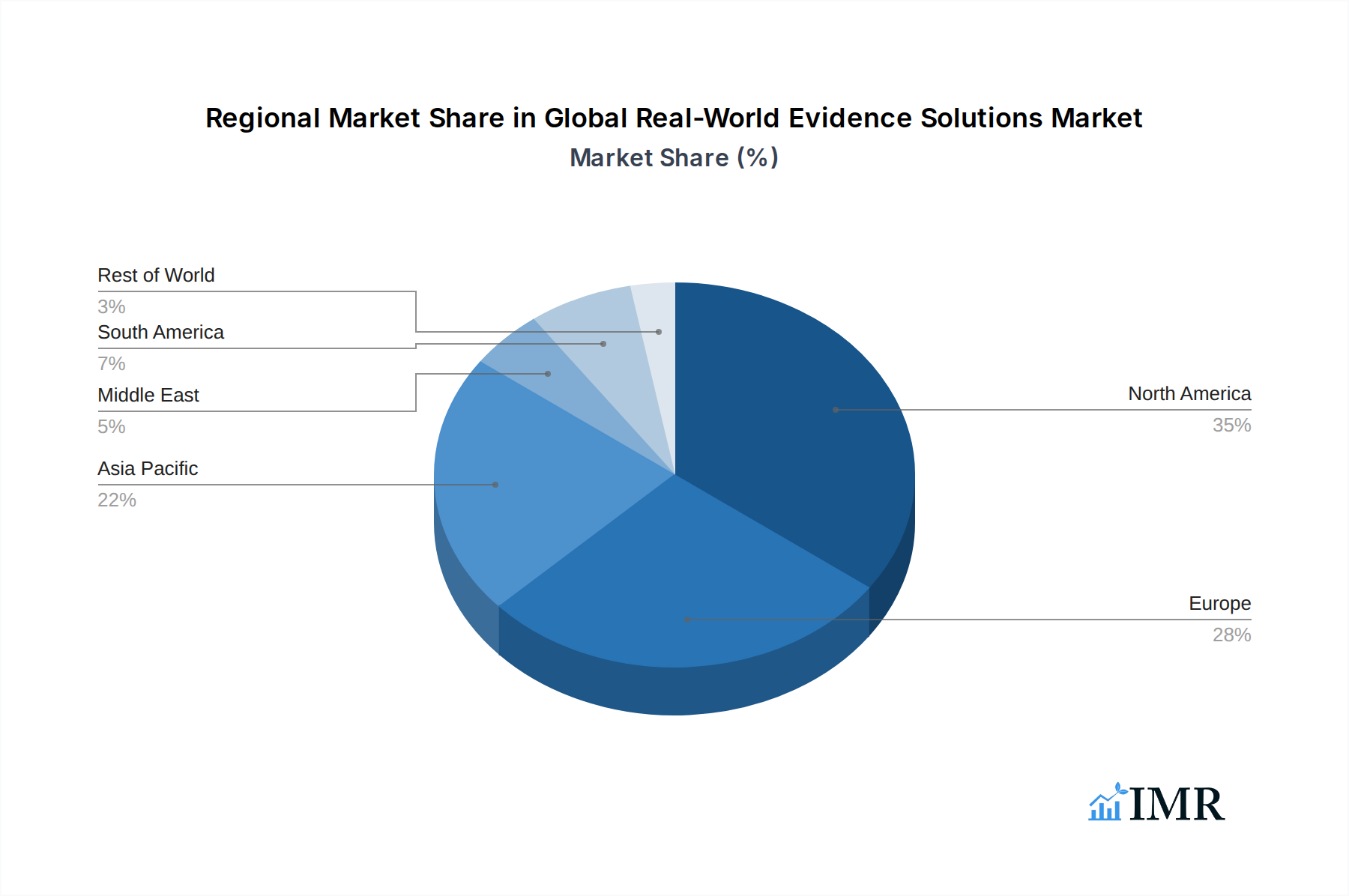
Global Real-World Evidence Solutions Market Regional Market Share

Geographic Coverage of Global Real-World Evidence Solutions Market
Global Real-World Evidence Solutions Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 9.40% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. IMR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Component
- 5.1.1. Claims Data
- 5.1.2. Clinical Settings Data
- 5.1.3. Patient-Powered Data
- 5.1.4. Pharmacy Data
- 5.1.5. Other Components
- 5.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 5.2.1. Oncology
- 5.2.2. Immunology
- 5.2.3. Neurology
- 5.2.4. Cardiovascular Disease
- 5.2.5. Other Therapeutic Areas
- 5.3. Market Analysis, Insights and Forecast - by End User
- 5.3.1. Healthcare Payers
- 5.3.2. Healthcare Providers
- 5.3.3. Pharmaceutical & Medical Device Companies
- 5.3.4. Other End Users
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. North America
- 5.4.2. Europe
- 5.4.3. Asia Pacific
- 5.4.4. Middle East
- 5.4.5. GCC
- 5.4.6. South America
- 5.1. Market Analysis, Insights and Forecast - by Component
- 6. Global Real-World Evidence Solutions Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Component
- 6.1.1. Claims Data
- 6.1.2. Clinical Settings Data
- 6.1.3. Patient-Powered Data
- 6.1.4. Pharmacy Data
- 6.1.5. Other Components
- 6.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 6.2.1. Oncology
- 6.2.2. Immunology
- 6.2.3. Neurology
- 6.2.4. Cardiovascular Disease
- 6.2.5. Other Therapeutic Areas
- 6.3. Market Analysis, Insights and Forecast - by End User
- 6.3.1. Healthcare Payers
- 6.3.2. Healthcare Providers
- 6.3.3. Pharmaceutical & Medical Device Companies
- 6.3.4. Other End Users
- 6.1. Market Analysis, Insights and Forecast - by Component
- 7. North America Global Real-World Evidence Solutions Market Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Component
- 7.1.1. Claims Data
- 7.1.2. Clinical Settings Data
- 7.1.3. Patient-Powered Data
- 7.1.4. Pharmacy Data
- 7.1.5. Other Components
- 7.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 7.2.1. Oncology
- 7.2.2. Immunology
- 7.2.3. Neurology
- 7.2.4. Cardiovascular Disease
- 7.2.5. Other Therapeutic Areas
- 7.3. Market Analysis, Insights and Forecast - by End User
- 7.3.1. Healthcare Payers
- 7.3.2. Healthcare Providers
- 7.3.3. Pharmaceutical & Medical Device Companies
- 7.3.4. Other End Users
- 7.1. Market Analysis, Insights and Forecast - by Component
- 8. Europe Global Real-World Evidence Solutions Market Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Component
- 8.1.1. Claims Data
- 8.1.2. Clinical Settings Data
- 8.1.3. Patient-Powered Data
- 8.1.4. Pharmacy Data
- 8.1.5. Other Components
- 8.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 8.2.1. Oncology
- 8.2.2. Immunology
- 8.2.3. Neurology
- 8.2.4. Cardiovascular Disease
- 8.2.5. Other Therapeutic Areas
- 8.3. Market Analysis, Insights and Forecast - by End User
- 8.3.1. Healthcare Payers
- 8.3.2. Healthcare Providers
- 8.3.3. Pharmaceutical & Medical Device Companies
- 8.3.4. Other End Users
- 8.1. Market Analysis, Insights and Forecast - by Component
- 9. Asia Pacific Global Real-World Evidence Solutions Market Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Component
- 9.1.1. Claims Data
- 9.1.2. Clinical Settings Data
- 9.1.3. Patient-Powered Data
- 9.1.4. Pharmacy Data
- 9.1.5. Other Components
- 9.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 9.2.1. Oncology
- 9.2.2. Immunology
- 9.2.3. Neurology
- 9.2.4. Cardiovascular Disease
- 9.2.5. Other Therapeutic Areas
- 9.3. Market Analysis, Insights and Forecast - by End User
- 9.3.1. Healthcare Payers
- 9.3.2. Healthcare Providers
- 9.3.3. Pharmaceutical & Medical Device Companies
- 9.3.4. Other End Users
- 9.1. Market Analysis, Insights and Forecast - by Component
- 10. Middle East Global Real-World Evidence Solutions Market Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Component
- 10.1.1. Claims Data
- 10.1.2. Clinical Settings Data
- 10.1.3. Patient-Powered Data
- 10.1.4. Pharmacy Data
- 10.1.5. Other Components
- 10.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 10.2.1. Oncology
- 10.2.2. Immunology
- 10.2.3. Neurology
- 10.2.4. Cardiovascular Disease
- 10.2.5. Other Therapeutic Areas
- 10.3. Market Analysis, Insights and Forecast - by End User
- 10.3.1. Healthcare Payers
- 10.3.2. Healthcare Providers
- 10.3.3. Pharmaceutical & Medical Device Companies
- 10.3.4. Other End Users
- 10.1. Market Analysis, Insights and Forecast - by Component
- 11. GCC Global Real-World Evidence Solutions Market Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Component
- 11.1.1. Claims Data
- 11.1.2. Clinical Settings Data
- 11.1.3. Patient-Powered Data
- 11.1.4. Pharmacy Data
- 11.1.5. Other Components
- 11.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 11.2.1. Oncology
- 11.2.2. Immunology
- 11.2.3. Neurology
- 11.2.4. Cardiovascular Disease
- 11.2.5. Other Therapeutic Areas
- 11.3. Market Analysis, Insights and Forecast - by End User
- 11.3.1. Healthcare Payers
- 11.3.2. Healthcare Providers
- 11.3.3. Pharmaceutical & Medical Device Companies
- 11.3.4. Other End Users
- 11.1. Market Analysis, Insights and Forecast - by Component
- 12. South America Global Real-World Evidence Solutions Market Analysis, Insights and Forecast, 2020-2032
- 12.1. Market Analysis, Insights and Forecast - by Component
- 12.1.1. Claims Data
- 12.1.2. Clinical Settings Data
- 12.1.3. Patient-Powered Data
- 12.1.4. Pharmacy Data
- 12.1.5. Other Components
- 12.2. Market Analysis, Insights and Forecast - by Therapeutic Area
- 12.2.1. Oncology
- 12.2.2. Immunology
- 12.2.3. Neurology
- 12.2.4. Cardiovascular Disease
- 12.2.5. Other Therapeutic Areas
- 12.3. Market Analysis, Insights and Forecast - by End User
- 12.3.1. Healthcare Payers
- 12.3.2. Healthcare Providers
- 12.3.3. Pharmaceutical & Medical Device Companies
- 12.3.4. Other End Users
- 12.1. Market Analysis, Insights and Forecast - by Component
- 13. Competitive Analysis
- 13.1. Company Profiles
- 13.1.1 Clinigen Group PLC
- 13.1.1.1. Company Overview
- 13.1.1.2. Products
- 13.1.1.3. Company Financials
- 13.1.1.4. SWOT Analysis
- 13.1.2 Syneos Health
- 13.1.2.1. Company Overview
- 13.1.2.2. Products
- 13.1.2.3. Company Financials
- 13.1.2.4. SWOT Analysis
- 13.1.3 Flatiron Health Inc
- 13.1.3.1. Company Overview
- 13.1.3.2. Products
- 13.1.3.3. Company Financials
- 13.1.3.4. SWOT Analysis
- 13.1.4 Parexel International
- 13.1.4.1. Company Overview
- 13.1.4.2. Products
- 13.1.4.3. Company Financials
- 13.1.4.4. SWOT Analysis
- 13.1.5 IQVIA
- 13.1.5.1. Company Overview
- 13.1.5.2. Products
- 13.1.5.3. Company Financials
- 13.1.5.4. SWOT Analysis
- 13.1.6 SAS Institute
- 13.1.6.1. Company Overview
- 13.1.6.2. Products
- 13.1.6.3. Company Financials
- 13.1.6.4. SWOT Analysis
- 13.1.7 Pharmaceutical Product Development (PPD Inc )
- 13.1.7.1. Company Overview
- 13.1.7.2. Products
- 13.1.7.3. Company Financials
- 13.1.7.4. SWOT Analysis
- 13.1.8 Oracle Corporation
- 13.1.8.1. Company Overview
- 13.1.8.2. Products
- 13.1.8.3. Company Financials
- 13.1.8.4. SWOT Analysis
- 13.1.9 PerkinElmer Inc
- 13.1.9.1. Company Overview
- 13.1.9.2. Products
- 13.1.9.3. Company Financials
- 13.1.9.4. SWOT Analysis
- 13.1.10 IBM
- 13.1.10.1. Company Overview
- 13.1.10.2. Products
- 13.1.10.3. Company Financials
- 13.1.10.4. SWOT Analysis
- 13.1.11 Icon PLC
- 13.1.11.1. Company Overview
- 13.1.11.2. Products
- 13.1.11.3. Company Financials
- 13.1.11.4. SWOT Analysis
- 13.1.1 Clinigen Group PLC
- 13.2. Market Entropy
- 13.2.1 Company's Key Areas Served
- 13.2.2 Recent Developments
- 13.3. Company Market Share Analysis 2025
- 13.3.1 Top 5 Companies Market Share Analysis
- 13.3.2 Top 3 Companies Market Share Analysis
- 13.4. List of Potential Customers
- 14. Research Methodology
List of Figures
- Figure 1: Global Global Real-World Evidence Solutions Market Revenue Breakdown (Million, %) by Region 2025 & 2033
- Figure 2: North America Global Real-World Evidence Solutions Market Revenue (Million), by Component 2025 & 2033
- Figure 3: North America Global Real-World Evidence Solutions Market Revenue Share (%), by Component 2025 & 2033
- Figure 4: North America Global Real-World Evidence Solutions Market Revenue (Million), by Therapeutic Area 2025 & 2033
- Figure 5: North America Global Real-World Evidence Solutions Market Revenue Share (%), by Therapeutic Area 2025 & 2033
- Figure 6: North America Global Real-World Evidence Solutions Market Revenue (Million), by End User 2025 & 2033
- Figure 7: North America Global Real-World Evidence Solutions Market Revenue Share (%), by End User 2025 & 2033
- Figure 8: North America Global Real-World Evidence Solutions Market Revenue (Million), by Country 2025 & 2033
- Figure 9: North America Global Real-World Evidence Solutions Market Revenue Share (%), by Country 2025 & 2033
- Figure 10: Europe Global Real-World Evidence Solutions Market Revenue (Million), by Component 2025 & 2033
- Figure 11: Europe Global Real-World Evidence Solutions Market Revenue Share (%), by Component 2025 & 2033
- Figure 12: Europe Global Real-World Evidence Solutions Market Revenue (Million), by Therapeutic Area 2025 & 2033
- Figure 13: Europe Global Real-World Evidence Solutions Market Revenue Share (%), by Therapeutic Area 2025 & 2033
- Figure 14: Europe Global Real-World Evidence Solutions Market Revenue (Million), by End User 2025 & 2033
- Figure 15: Europe Global Real-World Evidence Solutions Market Revenue Share (%), by End User 2025 & 2033
- Figure 16: Europe Global Real-World Evidence Solutions Market Revenue (Million), by Country 2025 & 2033
- Figure 17: Europe Global Real-World Evidence Solutions Market Revenue Share (%), by Country 2025 & 2033
- Figure 18: Asia Pacific Global Real-World Evidence Solutions Market Revenue (Million), by Component 2025 & 2033
- Figure 19: Asia Pacific Global Real-World Evidence Solutions Market Revenue Share (%), by Component 2025 & 2033
- Figure 20: Asia Pacific Global Real-World Evidence Solutions Market Revenue (Million), by Therapeutic Area 2025 & 2033
- Figure 21: Asia Pacific Global Real-World Evidence Solutions Market Revenue Share (%), by Therapeutic Area 2025 & 2033
- Figure 22: Asia Pacific Global Real-World Evidence Solutions Market Revenue (Million), by End User 2025 & 2033
- Figure 23: Asia Pacific Global Real-World Evidence Solutions Market Revenue Share (%), by End User 2025 & 2033
- Figure 24: Asia Pacific Global Real-World Evidence Solutions Market Revenue (Million), by Country 2025 & 2033
- Figure 25: Asia Pacific Global Real-World Evidence Solutions Market Revenue Share (%), by Country 2025 & 2033
- Figure 26: Middle East Global Real-World Evidence Solutions Market Revenue (Million), by Component 2025 & 2033
- Figure 27: Middle East Global Real-World Evidence Solutions Market Revenue Share (%), by Component 2025 & 2033
- Figure 28: Middle East Global Real-World Evidence Solutions Market Revenue (Million), by Therapeutic Area 2025 & 2033
- Figure 29: Middle East Global Real-World Evidence Solutions Market Revenue Share (%), by Therapeutic Area 2025 & 2033
- Figure 30: Middle East Global Real-World Evidence Solutions Market Revenue (Million), by End User 2025 & 2033
- Figure 31: Middle East Global Real-World Evidence Solutions Market Revenue Share (%), by End User 2025 & 2033
- Figure 32: Middle East Global Real-World Evidence Solutions Market Revenue (Million), by Country 2025 & 2033
- Figure 33: Middle East Global Real-World Evidence Solutions Market Revenue Share (%), by Country 2025 & 2033
- Figure 34: GCC Global Real-World Evidence Solutions Market Revenue (Million), by Component 2025 & 2033
- Figure 35: GCC Global Real-World Evidence Solutions Market Revenue Share (%), by Component 2025 & 2033
- Figure 36: GCC Global Real-World Evidence Solutions Market Revenue (Million), by Therapeutic Area 2025 & 2033
- Figure 37: GCC Global Real-World Evidence Solutions Market Revenue Share (%), by Therapeutic Area 2025 & 2033
- Figure 38: GCC Global Real-World Evidence Solutions Market Revenue (Million), by End User 2025 & 2033
- Figure 39: GCC Global Real-World Evidence Solutions Market Revenue Share (%), by End User 2025 & 2033
- Figure 40: GCC Global Real-World Evidence Solutions Market Revenue (Million), by Country 2025 & 2033
- Figure 41: GCC Global Real-World Evidence Solutions Market Revenue Share (%), by Country 2025 & 2033
- Figure 42: South America Global Real-World Evidence Solutions Market Revenue (Million), by Component 2025 & 2033
- Figure 43: South America Global Real-World Evidence Solutions Market Revenue Share (%), by Component 2025 & 2033
- Figure 44: South America Global Real-World Evidence Solutions Market Revenue (Million), by Therapeutic Area 2025 & 2033
- Figure 45: South America Global Real-World Evidence Solutions Market Revenue Share (%), by Therapeutic Area 2025 & 2033
- Figure 46: South America Global Real-World Evidence Solutions Market Revenue (Million), by End User 2025 & 2033
- Figure 47: South America Global Real-World Evidence Solutions Market Revenue Share (%), by End User 2025 & 2033
- Figure 48: South America Global Real-World Evidence Solutions Market Revenue (Million), by Country 2025 & 2033
- Figure 49: South America Global Real-World Evidence Solutions Market Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Component 2020 & 2033
- Table 2: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Therapeutic Area 2020 & 2033
- Table 3: Global Real-World Evidence Solutions Market Revenue Million Forecast, by End User 2020 & 2033
- Table 4: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Region 2020 & 2033
- Table 5: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Component 2020 & 2033
- Table 6: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Therapeutic Area 2020 & 2033
- Table 7: Global Real-World Evidence Solutions Market Revenue Million Forecast, by End User 2020 & 2033
- Table 8: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Country 2020 & 2033
- Table 9: United States Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 10: Canada Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 11: Mexico Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 12: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Component 2020 & 2033
- Table 13: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Therapeutic Area 2020 & 2033
- Table 14: Global Real-World Evidence Solutions Market Revenue Million Forecast, by End User 2020 & 2033
- Table 15: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Country 2020 & 2033
- Table 16: Germany Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 17: United Kingdom Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 18: France Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 19: Italy Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 20: Spain Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 21: Rest of Europe Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 22: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Component 2020 & 2033
- Table 23: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Therapeutic Area 2020 & 2033
- Table 24: Global Real-World Evidence Solutions Market Revenue Million Forecast, by End User 2020 & 2033
- Table 25: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Country 2020 & 2033
- Table 26: China Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 27: Japan Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 28: India Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 29: Australia Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 30: South Korea Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 31: Rest of Asia Pacific Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 32: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Component 2020 & 2033
- Table 33: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Therapeutic Area 2020 & 2033
- Table 34: Global Real-World Evidence Solutions Market Revenue Million Forecast, by End User 2020 & 2033
- Table 35: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Country 2020 & 2033
- Table 36: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Component 2020 & 2033
- Table 37: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Therapeutic Area 2020 & 2033
- Table 38: Global Real-World Evidence Solutions Market Revenue Million Forecast, by End User 2020 & 2033
- Table 39: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Country 2020 & 2033
- Table 40: South Africa Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 41: Rest of Middle East Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 42: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Component 2020 & 2033
- Table 43: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Therapeutic Area 2020 & 2033
- Table 44: Global Real-World Evidence Solutions Market Revenue Million Forecast, by End User 2020 & 2033
- Table 45: Global Real-World Evidence Solutions Market Revenue Million Forecast, by Country 2020 & 2033
- Table 46: Brazil Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 47: Argentina Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
- Table 48: Rest of South America Global Real-World Evidence Solutions Market Revenue (Million) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Global Real-World Evidence Solutions Market?
The projected CAGR is approximately 9.40%.
2. Which companies are prominent players in the Global Real-World Evidence Solutions Market?
Key companies in the market include Clinigen Group PLC, Syneos Health, Flatiron Health Inc, Parexel International, IQVIA, SAS Institute, Pharmaceutical Product Development (PPD Inc ), Oracle Corporation, PerkinElmer Inc, IBM, Icon PLC.
3. What are the main segments of the Global Real-World Evidence Solutions Market?
The market segments include Component, Therapeutic Area, End User.
4. Can you provide details about the market size?
The market size is estimated to be USD 1.47 Million as of 2022.
5. What are some drivers contributing to market growth?
Shift From Volume- to Value-based Care; Increasing Aging Population and Prevalence of Chronic Diseases.
6. What are the notable trends driving market growth?
Oncology is Anticipated to be the Dominant Segment During the Forecast Period.
7. Are there any restraints impacting market growth?
Unwillingness to Rely on Real-world Studies; Lack of Standards.
8. Can you provide examples of recent developments in the market?
In December 2021, EVERSANA signed an agreement with Janssen Research & Development LLC (Janssen) to drive evidence-based development of Janssen therapies, treatments, and patient support models.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Global Real-World Evidence Solutions Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Global Real-World Evidence Solutions Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Global Real-World Evidence Solutions Market?
To stay informed about further developments, trends, and reports in the Global Real-World Evidence Solutions Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
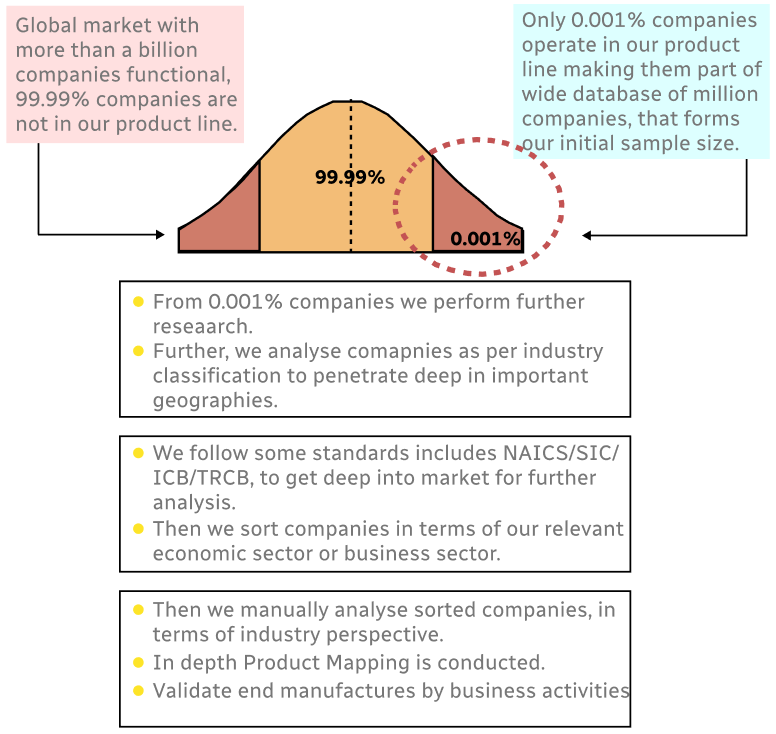
Step 1 - Identification of Relevant Samples Size from Population Database
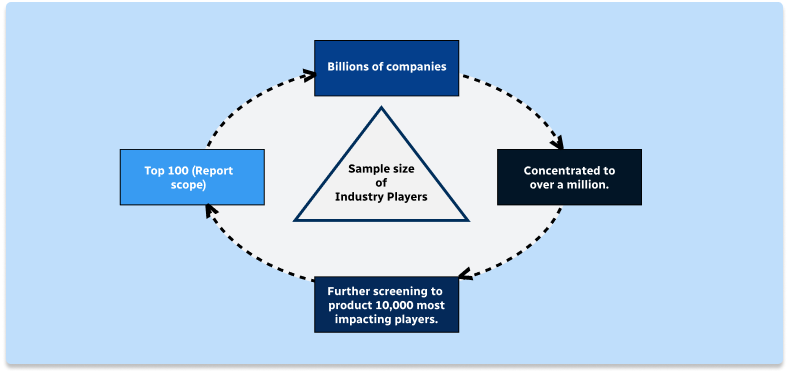
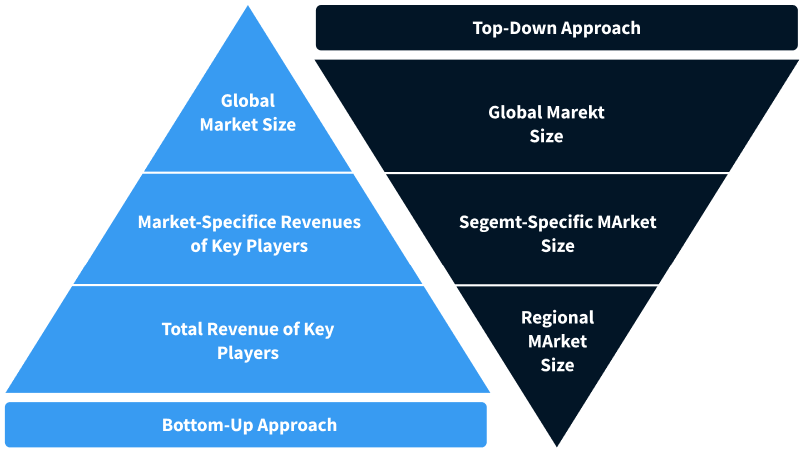
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
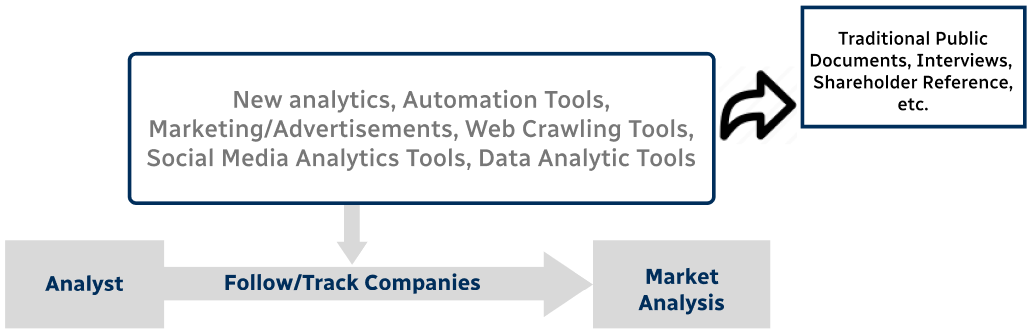
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


