Key Insights
The United States In Vitro Diagnostics (IVD) market is poised for significant expansion, projected to reach a robust $40.85 million by 2025, with a projected Compound Annual Growth Rate (CAGR) of 5.57% over the forecast period of 2025-2033. This impressive growth is fueled by several key drivers, including the increasing prevalence of chronic and infectious diseases, a growing aging population susceptible to various health conditions, and significant advancements in diagnostic technologies. The rising demand for early disease detection and personalized medicine further propels the market forward. Innovations in molecular diagnostics, such as PCR and next-generation sequencing, are becoming increasingly vital for accurate and rapid disease identification, while the continuous development of user-friendly and automated instruments in clinical chemistry and immuno-diagnostics contributes to greater efficiency in healthcare settings.
-Market.png)
United States In Vitro Diagnostics (IVD) Market Market Size (In Million)

The IVD market in the United States is characterized by dynamic trends and a competitive landscape. The shift towards point-of-care (POC) testing and home-based diagnostics is gaining momentum, driven by the need for faster results and increased patient convenience. Furthermore, the integration of artificial intelligence (AI) and machine learning (ML) in IVD platforms is revolutionizing data analysis and diagnostic accuracy, leading to more informed clinical decisions. Key segments expected to witness substantial growth include molecular diagnostics and immuno-diagnostics, owing to their critical role in infectious disease surveillance and the management of complex conditions like cancer and autoimmune disorders. While technological advancements and increasing healthcare expenditure act as strong growth drivers, challenges such as stringent regulatory hurdles and the high cost of advanced diagnostic equipment may present some restraints. However, the overall outlook remains highly optimistic due to the persistent need for accurate and timely diagnostic solutions in the US healthcare ecosystem.
-Market.png)
United States In Vitro Diagnostics (IVD) Market Company Market Share

Here's a comprehensive, SEO-optimized report description for the United States In Vitro Diagnostics (IVD) Market, integrating high-traffic keywords and structured as requested.
This in-depth report offers a strategic overview of the United States In Vitro Diagnostics (IVD) Market, a critical and rapidly evolving sector within the healthcare industry. Covering the period from 2019 to 2033, with a base year of 2025, this analysis delves into market dynamics, growth trends, product landscapes, key players, and emerging opportunities. Leveraging extensive data and expert insights, this report is designed to equip industry professionals, investors, and stakeholders with the knowledge needed to navigate the complexities of the US IVD market. Discover the latest advancements in molecular diagnostics, clinical chemistry, immuno diagnostics, and haematology, alongside in-depth analysis of diagnostic instruments, reagents, and disposable IVD solutions. This report also examines crucial applications such as infectious diseases, diabetes, cancer diagnostics, and cardiology, and identifies the key end-users including diagnostic laboratories and hospitals.
United States In Vitro Diagnostics (IVD) Market Market Dynamics & Structure
The United States IVD market is characterized by a dynamic interplay of technological innovation, stringent regulatory oversight, and evolving end-user demands. Market concentration is moderately high, with key players like Siemens Healthcare GmbH, Becton Dickinson and Company, F Hoffmann-La Roche AG, Bio-Rad Laboratories Inc, Thermo Fisher Scientific Inc, Abbott Laboratories, Agilent Technologies Inc, Danaher Corporation, Qiagen, bioMerieux SA, Sysmex Corporation, and Arkray Inc holding significant shares. Technological innovation is primarily driven by the demand for faster, more accurate, and minimally invasive diagnostic methods, particularly in areas like personalized medicine and early disease detection. The regulatory framework, overseen by the FDA, plays a crucial role in market entry and product approvals, ensuring patient safety and diagnostic efficacy. Competitive product substitutes are emerging with advancements in point-of-care testing and home-use diagnostics, challenging traditional laboratory-based models. End-user demographics are shifting towards a greater emphasis on preventative healthcare and chronic disease management, influencing the demand for specific IVD tests and platforms. Mergers and acquisitions (M&A) activity remains a significant trend, with companies consolidating to expand their product portfolios, geographic reach, and technological capabilities. For instance, several smaller companies have been acquired by larger entities to gain access to novel technologies or established market segments.
- Market Concentration: Moderately concentrated with a few dominant players and a growing number of specialized niche providers.
- Technological Innovation Drivers: Demand for precision medicine, AI-driven diagnostics, liquid biopsies, and rapid point-of-care testing.
- Regulatory Frameworks: Stringent FDA regulations for product clearance and post-market surveillance, impacting time-to-market.
- Competitive Product Substitutes: Rise of point-of-care (POC) devices, home testing kits, and integrated diagnostic platforms.
- End-User Demographics: Increasing prevalence of chronic diseases, aging population, and growing consumer awareness of health monitoring.
- M&A Trends: Continuous consolidation to acquire innovative technologies, expand market access, and achieve economies of scale. Deal volumes in the IVD sector have seen consistent activity over the past five years, with strategic acquisitions focused on areas like molecular diagnostics and companion diagnostics.
United States In Vitro Diagnostics (IVD) Market Growth Trends & Insights
The United States In Vitro Diagnostics (IVD) Market is poised for substantial growth, driven by an escalating demand for advanced diagnostic solutions across various healthcare settings. The market size is projected to witness a significant expansion, fueled by increasing investments in research and development, the growing prevalence of chronic and infectious diseases, and the continuous integration of cutting-edge technologies. Adoption rates for sophisticated diagnostic tests, particularly in molecular diagnostics and immuno diagnostics, are on a steady rise, reflecting their crucial role in disease identification, prognosis, and treatment monitoring. Technological disruptions are rapidly reshaping the landscape, with the advent of artificial intelligence (AI) in image analysis, automation in laboratory workflows, and the development of more sensitive and specific assays. Consumer behavior shifts are also playing a pivotal role; individuals are becoming more proactive in managing their health, leading to a greater demand for early detection and personalized screening. This translates to increased market penetration of home-use diagnostic kits and point-of-care testing devices. The market is expected to grow at a Compound Annual Growth Rate (CAGR) of approximately xx% during the forecast period, reaching an estimated market value of $XX million by 2033. The growing emphasis on preventative healthcare, coupled with favorable reimbursement policies for diagnostic procedures, further accelerates market expansion. Furthermore, the integration of digital health technologies and data analytics in IVD is enhancing diagnostic efficiency and patient outcomes, contributing to robust market growth. The expansion of telehealth services also indirectly supports the IVD market by enabling remote patient monitoring and facilitating the timely interpretation of diagnostic results. The increasing focus on companion diagnostics for targeted therapies in oncology also represents a significant growth driver.
Dominant Regions, Countries, or Segments in United States In Vitro Diagnostics (IVD) Market
Within the vast United States IVD market, certain segments and applications demonstrate exceptional growth and dominance, driven by a confluence of factors including technological adoption, healthcare infrastructure, and disease burden. Molecular Diagnostics stands out as a leading segment, experiencing a remarkable surge in demand due to its accuracy in identifying infectious agents, genetic predispositions, and personalized treatment markers. This dominance is further bolstered by the increasing incidence of infectious diseases, the growing focus on genetic testing for hereditary conditions, and the rapid advancements in next-generation sequencing (NGS) technologies, making it a cornerstone of modern diagnostics.
In terms of product types, Reagents consistently represent a substantial portion of the market revenue, owing to their consumable nature and their indispensable role in all IVD testing procedures. The continuous development of novel reagents with enhanced sensitivity and specificity further solidifies their market position.
From an application perspective, Cancer/Oncology diagnostics are a major growth engine. The rising cancer incidence rates across the United States, coupled with the development of advanced diagnostic tools for early detection, staging, and treatment selection, including companion diagnostics, contribute significantly to this segment's prominence. The increasing adoption of disposable IVD products also marks a significant trend, driven by their convenience, reduced risk of cross-contamination, and suitability for point-of-care settings.
The Diagnostic Laboratories end-user segment remains the largest contributor to market revenue. These facilities are at the forefront of performing a wide array of diagnostic tests, from routine screenings to complex specialized analyses. Their robust infrastructure, skilled workforce, and high test volumes underpin their market dominance.
Key drivers for these dominant segments include:
- Molecular Diagnostics: High accuracy for pathogen detection (e.g., COVID-19, influenza), growth in genetic testing, advancements in PCR and NGS.
- Reagents: Essential for all tests, continuous innovation for improved performance, widespread use across all IVD segments.
- Cancer/Oncology: Increasing cancer prevalence, demand for early detection and personalized therapies, advancements in liquid biopsies and targeted therapies.
- Disposable IVD: Focus on infection control, convenience in point-of-care settings, growing demand in home healthcare.
- Diagnostic Laboratories: High volume of testing, comprehensive service offerings, centralized testing hubs.
The economic policies promoting healthcare access, federal and state funding for disease research, and the robust healthcare infrastructure across the US, particularly in major metropolitan hubs, create a fertile ground for these segments to flourish. The market share of Molecular Diagnostics within the broader IVD market is estimated to be around xx%, while Reagents contribute roughly xx% to the overall market value. Cancer diagnostics alone represent an estimated xx% of the total application-driven market.
United States In Vitro Diagnostics (IVD) Market Product Landscape
The United States IVD market is characterized by a continuous stream of product innovations aimed at enhancing diagnostic accuracy, efficiency, and accessibility. Key product developments include advancements in automated immunoassay analyzers for faster and more reliable detection of biomarkers, high-throughput molecular diagnostic platforms for comprehensive genetic analysis, and integrated systems that combine multiple testing capabilities. Furthermore, there is a significant focus on the development of novel reagents with improved sensitivity and specificity for early disease detection, as well as the creation of user-friendly, disposable IVD devices for point-of-care and home-use applications. Performance metrics are continuously being pushed, with advancements leading to reduced turnaround times, lower detection limits for analytes, and enhanced multiplexing capabilities, enabling the simultaneous detection of multiple targets from a single sample.
Key Drivers, Barriers & Challenges in United States In Vitro Diagnostics (IVD) Market
Key Drivers:
- Technological Advancements: Continuous innovation in areas like molecular diagnostics, liquid biopsies, and AI-powered analysis fuels market growth. The development of rapid and accurate tests for emerging infectious diseases also acts as a significant driver.
- Increasing Prevalence of Chronic and Infectious Diseases: The rising incidence of conditions like diabetes, cancer, cardiovascular diseases, and infectious outbreaks necessitates advanced diagnostic solutions, boosting demand.
- Growing Demand for Personalized Medicine: The shift towards tailored treatment approaches requires precise diagnostic tools, particularly companion diagnostics, driving the adoption of sophisticated IVD tests.
- Favorable Reimbursement Policies: Government and private payers increasingly recognize the value of diagnostic testing, leading to improved reimbursement, which encourages adoption and investment.
- Aging Population: The demographic shift towards an older population cohort generally experiences a higher incidence of chronic diseases, thus increasing the demand for diagnostic testing.
Barriers & Challenges:
- Stringent Regulatory Hurdles: The FDA's rigorous approval process can lead to lengthy timelines and substantial costs for new product introductions, potentially slowing market entry.
- High Cost of Advanced Technologies: The initial investment in sophisticated IVD instruments and platforms can be substantial, posing a barrier for smaller healthcare facilities and laboratories.
- Data Security and Privacy Concerns: The increasing use of digital health and AI in IVD raises concerns about the security and privacy of sensitive patient data, requiring robust cybersecurity measures.
- Skilled Workforce Shortage: A lack of trained professionals to operate and interpret results from advanced IVD equipment can hinder adoption and efficient utilization.
- Reimbursement Policy Uncertainty: While generally favorable, potential changes or uncertainties in reimbursement policies can impact market growth and investment decisions. For instance, shifts in Medicare or private insurance coverage for specific tests can create market volatility.
Emerging Opportunities in United States In Vitro Diagnostics (IVD) Market
Emerging opportunities in the United States IVD market lie in the expanding applications of liquid biopsies for non-invasive cancer detection and monitoring, and the continued development of point-of-care (POC) testing solutions for decentralized healthcare settings and rapid diagnosis. The integration of Artificial Intelligence (AI) and machine learning in diagnostic algorithms promises to enhance accuracy and efficiency, particularly in areas like image analysis and predictive diagnostics. Furthermore, there is a growing demand for companion diagnostics to guide targeted therapies in oncology and other disease areas, creating a niche for specialized IVD development. The increasing consumer interest in at-home testing kits for various health parameters presents a significant opportunity for market expansion beyond traditional clinical settings. The growing adoption of telehealth also opens avenues for remote diagnostic support and monitoring.
Growth Accelerators in the United States In Vitro Diagnostics (IVD) Market Industry
Long-term growth in the United States IVD market is being significantly accelerated by breakthroughs in genomic sequencing technologies, enabling more precise and personalized disease risk assessment and treatment selection. Strategic partnerships between IVD manufacturers, pharmaceutical companies, and academic research institutions are fostering the development of innovative companion diagnostics and novel therapeutic targets. Market expansion strategies, including the increased focus on developing integrated diagnostic platforms that offer multiplex testing capabilities, are streamlining laboratory workflows and improving efficiency. Furthermore, the growing emphasis on preventative healthcare and early disease detection is driving sustained demand for a wide range of IVD solutions. The increasing investment in point-of-care diagnostics and home-use testing technologies is also a key accelerator, making diagnostics more accessible and convenient for patients.
Key Players Shaping the United States In Vitro Diagnostics (IVD) Market Market
- Siemens Healthcare GmbH
- Becton Dickinson and Company
- F Hoffmann-La Roche AG
- Bio-Rad Laboratories Inc
- Thermo Fisher Scientific Inc
- Abbott Laboratories
- Agilent Technologies Inc
- Danaher Corporation
- Qiagen
- bioMerieux SA
- Sysmex Corporation
- Arkray Inc
Notable Milestones in United States In Vitro Diagnostics (IVD) Market Sector
- July 2022: The United States Food and Drug Administration (FDA) cleared The Lumipulse G β-Amyloid Ratio (1-42/1-40) test, manufactured by Fujirebio Inc., marking the first diagnostic laboratory test to aid in the diagnosis of Alzheimer's disease.
- July 2022: Roche announced that the United States Food and Drug Administration (FDA) granted Breakthrough Device Designation to the Elecsys Amyloid Plasma Panel, an innovative new solution designed to enable earlier detection of Alzheimer's disease.
In-Depth United States In Vitro Diagnostics (IVD) Market Market Outlook
The future outlook for the United States In Vitro Diagnostics (IVD) Market is exceptionally positive, with sustained growth anticipated due to ongoing technological advancements and evolving healthcare demands. Strategic opportunities abound in the expansion of personalized medicine through companion diagnostics and advanced genomic testing, as well as in the burgeoning field of liquid biopsies for early cancer detection. The increasing adoption of point-of-care diagnostics and at-home testing solutions will further democratize access to diagnostics, catering to a growing consumer desire for proactive health management. The integration of AI and machine learning into diagnostic platforms is expected to unlock new levels of accuracy and efficiency. Continued investment in research and development, coupled with collaborations between industry stakeholders, will drive innovation and solidify the market's trajectory towards providing more precise, rapid, and accessible diagnostic solutions for a healthier future.
United States In Vitro Diagnostics (IVD) Market Segmentation
-
1. Test Type
- 1.1. Clinical Chemistry
- 1.2. Molecular Diagnostics
- 1.3. Immuno Diagnostics
- 1.4. Haematology
- 1.5. Other Test Types
-
2. Product
- 2.1. Instruments
- 2.2. Reagents
- 2.3. Other Products
-
3. Usability
- 3.1. Disposable IVD
- 3.2. Reusable IVD
-
4. Application
- 4.1. Infectious Diseases
- 4.2. Diabetes
- 4.3. Cancer/Oncology
- 4.4. Cardiology
- 4.5. Autoimmune Diseases
- 4.6. Nephrology
- 4.7. Other Applications
-
5. End User
- 5.1. Diagnostic Laboratories
- 5.2. Hospitals and Clinics
- 5.3. Other End Users
United States In Vitro Diagnostics (IVD) Market Segmentation By Geography
- 1. United States
-Market.png)
United States In Vitro Diagnostics (IVD) Market Regional Market Share

Geographic Coverage of United States In Vitro Diagnostics (IVD) Market
United States In Vitro Diagnostics (IVD) Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.57% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. IMR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Test Type
- 5.1.1. Clinical Chemistry
- 5.1.2. Molecular Diagnostics
- 5.1.3. Immuno Diagnostics
- 5.1.4. Haematology
- 5.1.5. Other Test Types
- 5.2. Market Analysis, Insights and Forecast - by Product
- 5.2.1. Instruments
- 5.2.2. Reagents
- 5.2.3. Other Products
- 5.3. Market Analysis, Insights and Forecast - by Usability
- 5.3.1. Disposable IVD
- 5.3.2. Reusable IVD
- 5.4. Market Analysis, Insights and Forecast - by Application
- 5.4.1. Infectious Diseases
- 5.4.2. Diabetes
- 5.4.3. Cancer/Oncology
- 5.4.4. Cardiology
- 5.4.5. Autoimmune Diseases
- 5.4.6. Nephrology
- 5.4.7. Other Applications
- 5.5. Market Analysis, Insights and Forecast - by End User
- 5.5.1. Diagnostic Laboratories
- 5.5.2. Hospitals and Clinics
- 5.5.3. Other End Users
- 5.6. Market Analysis, Insights and Forecast - by Region
- 5.6.1. United States
- 5.1. Market Analysis, Insights and Forecast - by Test Type
- 6. United States In Vitro Diagnostics (IVD) Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Test Type
- 6.1.1. Clinical Chemistry
- 6.1.2. Molecular Diagnostics
- 6.1.3. Immuno Diagnostics
- 6.1.4. Haematology
- 6.1.5. Other Test Types
- 6.2. Market Analysis, Insights and Forecast - by Product
- 6.2.1. Instruments
- 6.2.2. Reagents
- 6.2.3. Other Products
- 6.3. Market Analysis, Insights and Forecast - by Usability
- 6.3.1. Disposable IVD
- 6.3.2. Reusable IVD
- 6.4. Market Analysis, Insights and Forecast - by Application
- 6.4.1. Infectious Diseases
- 6.4.2. Diabetes
- 6.4.3. Cancer/Oncology
- 6.4.4. Cardiology
- 6.4.5. Autoimmune Diseases
- 6.4.6. Nephrology
- 6.4.7. Other Applications
- 6.5. Market Analysis, Insights and Forecast - by End User
- 6.5.1. Diagnostic Laboratories
- 6.5.2. Hospitals and Clinics
- 6.5.3. Other End Users
- 6.1. Market Analysis, Insights and Forecast - by Test Type
- 7. Competitive Analysis
- 7.1. Company Profiles
- 7.1.1 Siemens Healthcare GmbH
- 7.1.1.1. Company Overview
- 7.1.1.2. Products
- 7.1.1.3. Company Financials
- 7.1.1.4. SWOT Analysis
- 7.1.2 Becton Dickinson and Company
- 7.1.2.1. Company Overview
- 7.1.2.2. Products
- 7.1.2.3. Company Financials
- 7.1.2.4. SWOT Analysis
- 7.1.3 F Hoffmann-La Roche AG
- 7.1.3.1. Company Overview
- 7.1.3.2. Products
- 7.1.3.3. Company Financials
- 7.1.3.4. SWOT Analysis
- 7.1.4 Bio-Rad Laboratories Inc
- 7.1.4.1. Company Overview
- 7.1.4.2. Products
- 7.1.4.3. Company Financials
- 7.1.4.4. SWOT Analysis
- 7.1.5 Thermo Fisher Scientific Inc
- 7.1.5.1. Company Overview
- 7.1.5.2. Products
- 7.1.5.3. Company Financials
- 7.1.5.4. SWOT Analysis
- 7.1.6 Abbott Laboratories
- 7.1.6.1. Company Overview
- 7.1.6.2. Products
- 7.1.6.3. Company Financials
- 7.1.6.4. SWOT Analysis
- 7.1.7 AgilentTechnologies Inc *List Not Exhaustive
- 7.1.7.1. Company Overview
- 7.1.7.2. Products
- 7.1.7.3. Company Financials
- 7.1.7.4. SWOT Analysis
- 7.1.8 Danaher Corporation
- 7.1.8.1. Company Overview
- 7.1.8.2. Products
- 7.1.8.3. Company Financials
- 7.1.8.4. SWOT Analysis
- 7.1.9 Qiagen
- 7.1.9.1. Company Overview
- 7.1.9.2. Products
- 7.1.9.3. Company Financials
- 7.1.9.4. SWOT Analysis
- 7.1.10 bioMerieux SA
- 7.1.10.1. Company Overview
- 7.1.10.2. Products
- 7.1.10.3. Company Financials
- 7.1.10.4. SWOT Analysis
- 7.1.11 Sysmex Corporation
- 7.1.11.1. Company Overview
- 7.1.11.2. Products
- 7.1.11.3. Company Financials
- 7.1.11.4. SWOT Analysis
- 7.1.12 Arkray Inc
- 7.1.12.1. Company Overview
- 7.1.12.2. Products
- 7.1.12.3. Company Financials
- 7.1.12.4. SWOT Analysis
- 7.1.1 Siemens Healthcare GmbH
- 7.2. Market Entropy
- 7.2.1 Company's Key Areas Served
- 7.2.2 Recent Developments
- 7.3. Company Market Share Analysis 2025
- 7.3.1 Top 5 Companies Market Share Analysis
- 7.3.2 Top 3 Companies Market Share Analysis
- 7.4. List of Potential Customers
- 8. Research Methodology
List of Figures
- Figure 1: United States In Vitro Diagnostics (IVD) Market Revenue Breakdown (Million, %) by Product 2025 & 2033
- Figure 2: United States In Vitro Diagnostics (IVD) Market Share (%) by Company 2025
List of Tables
- Table 1: United States In Vitro Diagnostics (IVD) Market Revenue Million Forecast, by Test Type 2020 & 2033
- Table 2: United States In Vitro Diagnostics (IVD) Market Revenue Million Forecast, by Product 2020 & 2033
- Table 3: United States In Vitro Diagnostics (IVD) Market Revenue Million Forecast, by Usability 2020 & 2033
- Table 4: United States In Vitro Diagnostics (IVD) Market Revenue Million Forecast, by Application 2020 & 2033
- Table 5: United States In Vitro Diagnostics (IVD) Market Revenue Million Forecast, by End User 2020 & 2033
- Table 6: United States In Vitro Diagnostics (IVD) Market Revenue Million Forecast, by Region 2020 & 2033
- Table 7: United States In Vitro Diagnostics (IVD) Market Revenue Million Forecast, by Test Type 2020 & 2033
- Table 8: United States In Vitro Diagnostics (IVD) Market Revenue Million Forecast, by Product 2020 & 2033
- Table 9: United States In Vitro Diagnostics (IVD) Market Revenue Million Forecast, by Usability 2020 & 2033
- Table 10: United States In Vitro Diagnostics (IVD) Market Revenue Million Forecast, by Application 2020 & 2033
- Table 11: United States In Vitro Diagnostics (IVD) Market Revenue Million Forecast, by End User 2020 & 2033
- Table 12: United States In Vitro Diagnostics (IVD) Market Revenue Million Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the United States In Vitro Diagnostics (IVD) Market?
The projected CAGR is approximately 5.57%.
2. Which companies are prominent players in the United States In Vitro Diagnostics (IVD) Market?
Key companies in the market include Siemens Healthcare GmbH, Becton Dickinson and Company, F Hoffmann-La Roche AG, Bio-Rad Laboratories Inc, Thermo Fisher Scientific Inc, Abbott Laboratories, AgilentTechnologies Inc *List Not Exhaustive, Danaher Corporation, Qiagen, bioMerieux SA, Sysmex Corporation, Arkray Inc.
3. What are the main segments of the United States In Vitro Diagnostics (IVD) Market?
The market segments include Test Type, Product, Usability, Application, End User.
4. Can you provide details about the market size?
The market size is estimated to be USD 40.85 Million as of 2022.
5. What are some drivers contributing to market growth?
High Burden of Chronic Diseases; Increasing Use of Point-of-care (POC) Diagnostics Spurring the IVD Market; Increasing Government Healthcare Expenditure and Consumer's Healthcare Spending; Advanced Technologies Fueling the IVD Market; Increasing Awareness and Acceptance of Personalized Medicine and Companion Diagnostics.
6. What are the notable trends driving market growth?
Cancer/Oncology Segment Segment is Expected to Witness Growth Over the Forecast Period.
7. Are there any restraints impacting market growth?
Lack of Proper Reimbursement; Stringent Regulatory Framework; Need For High Complexity Testing Centers.
8. Can you provide examples of recent developments in the market?
In July 2022, the United States Food and Drug Administration (FDA) cleared The Lumipulse G β-Amyloid Ratio (1-42/1-40) test, manufactured by Fujirebio Inc., the first diagnostic laboratory test to aid in the diagnosis of Alzheimer's disease.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3800, USD 4500, and USD 5800 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "United States In Vitro Diagnostics (IVD) Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the United States In Vitro Diagnostics (IVD) Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the United States In Vitro Diagnostics (IVD) Market?
To stay informed about further developments, trends, and reports in the United States In Vitro Diagnostics (IVD) Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
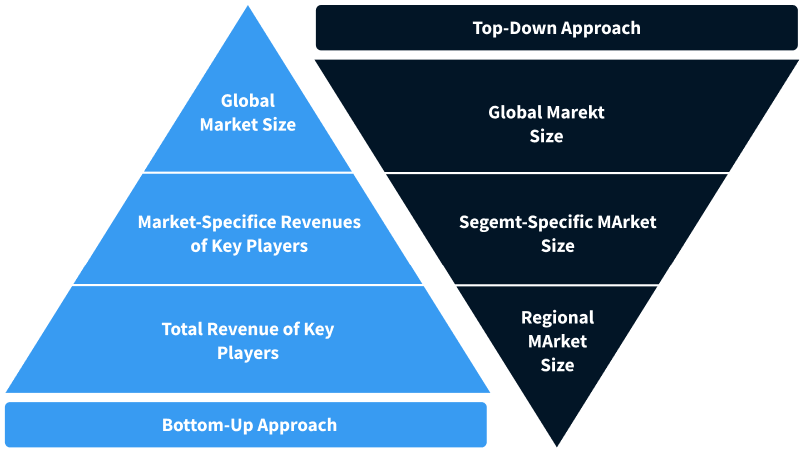
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


