Key Insights
The global Fondaparinux market is poised for substantial growth, projected to reach USD 691.9 million in 2024, with a compound annual growth rate (CAGR) of 6.8% expected to continue through the forecast period ending in 2033. This robust expansion is primarily driven by the increasing prevalence of venous thromboembolism (VTE) and deep vein thrombosis (DVT), particularly in aging populations and among individuals undergoing orthopedic surgeries, a key application area for Fondaparinux. The pharmaceutical industry's focus on developing innovative treatments for these conditions, coupled with heightened awareness and improved diagnostic capabilities, further fuels market demand. Moreover, the established efficacy and safety profile of Fondaparinux in preventing and treating VTE solidifies its position as a critical therapeutic agent, contributing to its steady market penetration. The market also benefits from increasing healthcare expenditure globally, enabling greater access to advanced anticoagulant therapies.
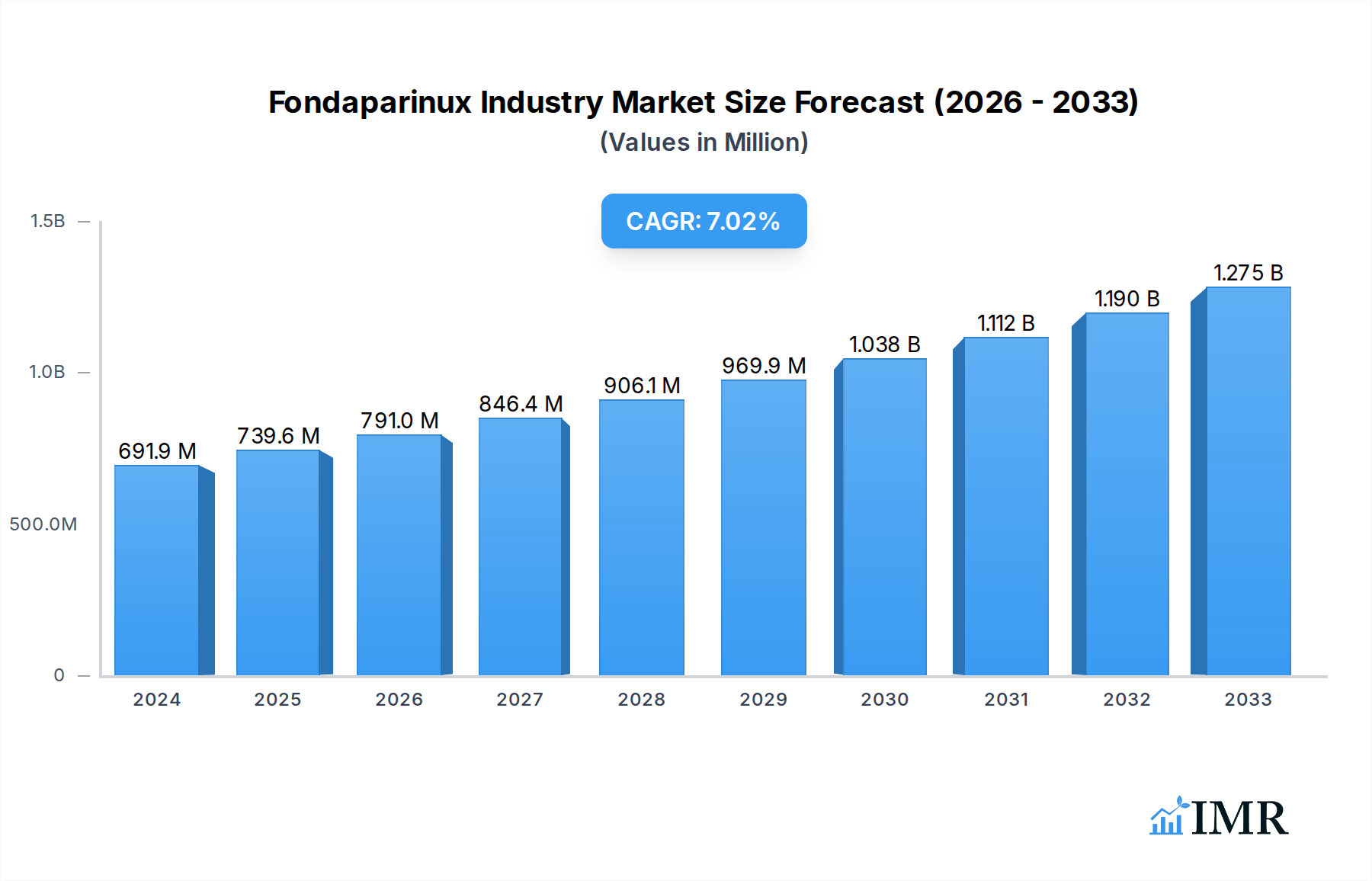
Fondaparinux Industry Market Size (In Million)

The Fondaparinux market dynamics are shaped by several key trends, including a growing preference for fixed-dose regimens and advancements in drug delivery systems aimed at enhancing patient compliance and convenience. While the market demonstrates a positive trajectory, certain restraints such as the availability of alternative anticoagulants and the potential for biosimilar competition pose challenges. However, the inherent advantages of Fondaparinux, particularly its targeted mechanism of action and established treatment protocols, are expected to mitigate these concerns. Geographically, North America and Europe currently dominate the market due to advanced healthcare infrastructure and high VTE incidence rates. Asia Pacific is anticipated to witness the fastest growth, driven by expanding healthcare access, increasing medical tourism, and a rising burden of chronic diseases. The market comprises both branded and generic segments, with generics playing an increasingly important role in expanding accessibility and affordability. Key players like Boehringer Ingelheim, Viatris, and Pfizer are actively involved in research, development, and market expansion strategies.
Fondaparinux Industry Company Market Share

Comprehensive Fondaparinux Industry Report: Market Dynamics, Growth Trends, and Future Outlook (2019-2033)
This in-depth report provides a thorough analysis of the global Fondaparinux industry, covering market dynamics, growth trajectories, regional dominance, product landscape, key drivers, challenges, opportunities, and a detailed outlook for the period of 2019–2033, with a base and estimated year of 2025. The report offers actionable insights for industry stakeholders, including pharmaceutical manufacturers, healthcare providers, investors, and policymakers, focusing on parent and child market segments to capture the full market spectrum.
Fondaparinux Industry Market Dynamics & Structure
The Fondaparinux industry is characterized by a moderately concentrated market structure, with key players actively engaged in strategic product development and market expansion. Technological innovation drivers are primarily focused on enhancing drug delivery systems and exploring new therapeutic applications for fondaparinux beyond its established role in venous thromboembolism (VTE) prophylaxis and treatment. Regulatory frameworks, particularly evolving guidelines for drug approvals and reimbursement policies, significantly influence market access and product uptake. Competitive product substitutes, such as direct oral anticoagulants (DOACs) and other low molecular weight heparins (LMWHs), present a continuous challenge, necessitating strong clinical evidence and cost-effectiveness for fondaparinux. End-user demographics, including an aging global population and a rising prevalence of conditions leading to VTE, are creating sustained demand. Mergers and acquisitions (M&A) trends indicate a strategic consolidation, with larger pharmaceutical companies aiming to expand their anticoagulation portfolios.
- Market Concentration: The market is dominated by a few key manufacturers, but a growing number of generic players are intensifying competition.
- Technological Innovation: Focus on improved formulations and potential combination therapies to enhance efficacy and patient convenience.
- Regulatory Landscape: Stringent approval processes and evolving reimbursement policies in major markets like North America and Europe are critical factors.
- Competitive Substitutes: High availability and adoption of DOACs and other anticoagulants necessitate robust differentiation strategies for fondaparinux.
- End-User Demographics: Increasing incidence of VTE due to sedentary lifestyles, obesity, and surgical procedures drives demand.
- M&A Trends: Strategic acquisitions by larger entities to gain market share and diversify product offerings.
Fondaparinux Industry Growth Trends & Insights
The Fondaparinux industry is poised for steady growth, driven by an increasing global demand for effective anticoagulation therapies, particularly in the prophylaxis and treatment of venous thromboembolism (VTE). Market size evolution is projected to be influenced by the expanding healthcare infrastructure in emerging economies and the continuous need for proven VTE management solutions in developed markets. Adoption rates are expected to remain robust, especially in hospital settings for post-operative patients and those with high VTE risk factors. Technological disruptions, while present in the broader anticoagulation market with novel oral anticoagulants, are less impactful on fondaparinux's established niche, which benefits from its specific safety profile and efficacy in certain patient populations. Consumer behavior shifts are leaning towards patient-centric care, demanding therapies that offer convenience and reduced risk of complications. The market is anticipated to witness a Compound Annual Growth Rate (CAGR) of approximately 5.2% from 2025 to 2033, reaching an estimated market value of $XXX million units by 2033. Market penetration is expected to deepen in regions with a higher prevalence of cardiovascular diseases and surgical procedures.
- Market Size Evolution: Consistent expansion driven by increasing VTE incidence and ongoing healthcare investments globally.
- Adoption Rates: High adoption in hospital settings for VTE prophylaxis in at-risk surgical patients and medical patients.
- Technological Disruptions: While DOACs are prevalent, fondaparinux retains its position due to specific clinical advantages and patient profiles.
- Consumer Behavior Shifts: Growing emphasis on preventative care and demand for effective, well-tolerated anticoagulation.
- CAGR Projection: An estimated CAGR of 5.2% from 2025 to 2033.
- Market Penetration: Deepening presence in both developed and emerging markets with robust healthcare systems.
Dominant Regions, Countries, or Segments in Fondaparinux Industry
The global Fondaparinux market demonstrates significant regional disparities, with North America currently leading in terms of market value and volume, primarily driven by a high incidence of VTE, advanced healthcare infrastructure, and favorable reimbursement policies for both branded and generic fondaparinux. The United States, in particular, is a dominant country due to its large patient population undergoing surgeries and a well-established VTE prevention protocol. The Generics segment within the Fondaparinux product type is experiencing substantial growth and is anticipated to become the dominant segment over the forecast period. This shift is fueled by increasing healthcare cost-containment pressures, leading to a higher preference for cost-effective generic alternatives to branded fondaparinux.
Key drivers for North America's dominance include:
- High Prevalence of VTE: Significant rates of deep vein thrombosis (DVT) and pulmonary embolism (PE) necessitate aggressive prophylactic and therapeutic interventions.
- Advanced Healthcare Infrastructure: Well-equipped hospitals and clinics with established protocols for VTE management and a high rate of surgical procedures.
- Reimbursement Policies: Robust insurance coverage and access programs that support the use of anticoagulants.
- Market Penetration of Generics: The availability and widespread acceptance of generic fondaparinux are increasing market share for this segment.
In terms of product segments, the Generics segment is outpacing the Branded segment due to:
- Cost-Effectiveness: Generics offer a significant cost advantage, making them more accessible to a wider patient population and healthcare systems focused on budget optimization.
- Patent Expirations: The expiry of patents for branded fondaparinux has opened the door for numerous generic manufacturers, increasing competition and driving down prices.
- Volume-Based Procurement (VBP): Initiatives like China's VBP program are actively promoting the procurement of generic drugs, further boosting the generics market.
- Increased Manufacturing Capacity: A growing number of pharmaceutical companies globally are investing in the production of generic fondaparinux, ensuring ample supply.
While North America leads, Europe also represents a significant market due to similar factors. Emerging markets in Asia-Pacific are showing promising growth potential, driven by improving healthcare access and a rising awareness of VTE. The anticipated market share of the generics segment is projected to reach approximately 70% by 2033.
Fondaparinux Industry Product Landscape
The Fondaparinux product landscape primarily revolves around subcutaneous injections, offering a synthetic pentasaccharide anticoagulant with a specific mechanism of action that selectively inhibits Factor Xa. Its therapeutic applications are well-defined for the prophylaxis and treatment of venous thromboembolism (VTE), including deep vein thrombosis (DVT) and pulmonary embolism (PE), particularly in patients undergoing orthopedic surgery or with other high-risk factors. Unique selling propositions include its predictable pharmacokinetic profile, lack of routine laboratory monitoring required for some other anticoagulants, and a generally favorable safety profile regarding heparin-induced thrombocytopenia (HIT). Technological advancements in this segment are largely focused on optimizing manufacturing processes for both branded and generic formulations to ensure consistent quality and cost-effectiveness, rather than radical new product introductions.
Key Drivers, Barriers & Challenges in Fondaparinux Industry
The Fondaparinux industry is propelled by several key drivers, including the increasing global incidence of venous thromboembolism (VTE) due to aging populations, sedentary lifestyles, and a rise in surgical procedures. Technological advancements in drug manufacturing ensure consistent quality and availability of both branded and generic forms. Favorable reimbursement policies in major healthcare systems also support market growth.
- Drivers:
- Rising VTE prevalence globally.
- Increasing number of orthopedic and major surgeries.
- Advancements in manufacturing efficiency for generics.
- Supportive reimbursement frameworks in key markets.
Conversely, the industry faces significant barriers and challenges. The continuous emergence and widespread adoption of direct oral anticoagulants (DOACs) represent a major competitive threat due to their oral administration convenience and comparable efficacy in many indications. Stringent regulatory approval processes and evolving pharmacovigilance requirements add to the complexity and cost of market entry and sustained product presence. Supply chain disruptions, especially for active pharmaceutical ingredients (APIs), and pricing pressures from generic competition can impact profitability.
- Barriers & Challenges:
- Competition from DOACs offering oral administration.
- Stringent regulatory hurdles and pharmacovigilance requirements.
- Pricing pressures due to generic market entry.
- Potential supply chain vulnerabilities for APIs.
- Need for ongoing post-market surveillance and safety monitoring.
Emerging Opportunities in Fondaparinux Industry
Emerging opportunities in the Fondaparinux industry lie in expanding its use in novel patient populations and geographical regions. Further research into its efficacy and safety in specific co-morbid conditions or as an adjunct therapy could unlock new therapeutic niches. The growing demand for cost-effective treatments in emerging economies presents a significant opportunity for generic fondaparinux manufacturers. Furthermore, the development of more convenient delivery mechanisms or improved formulations, while a long-term prospect, could enhance patient compliance and market competitiveness.
- Untapped Markets: Penetration into developing nations with increasing healthcare expenditure.
- Innovative Applications: Exploration of fondaparinux in underserved VTE patient groups or as part of combination therapies.
- Evolving Consumer Preferences: Adapting to patient demands for effective and accessible anticoagulation options.
Growth Accelerators in the Fondaparinux Industry Industry
The long-term growth of the Fondaparinux industry will be significantly accelerated by several key factors. The increasing global burden of VTE, driven by demographic shifts and lifestyle changes, will continue to fuel demand for effective anticoagulation therapies. Strategic partnerships between pharmaceutical companies and healthcare providers can streamline access and patient education, fostering wider adoption. Moreover, governments' increasing focus on population health and preventative care initiatives can lead to broader VTE screening and management programs, benefiting fondaparinux. The continuous drive for manufacturing efficiency and quality in generic production will ensure sustained affordability and availability, further solidifying its market position.
- Technological Breakthroughs: Continued optimization of manufacturing processes for enhanced purity and cost-effectiveness.
- Strategic Partnerships: Collaborations to improve patient access, education, and adherence programs.
- Market Expansion Strategies: Targeting emerging markets with growing healthcare needs and increasing disposable incomes.
Key Players Shaping the Fondaparinux Industry Market
The Fondaparinux industry landscape is shaped by a mix of established pharmaceutical giants and specialized generic manufacturers. These companies are instrumental in driving innovation, ensuring supply, and competing for market share.
- Boehringer Ingelheim International GmbH
- Viatris Inc
- Eugia
- Apotex Inc
- GSK plc
- Jiangsu Hengrui Medicine Co Ltd
- Aurobindo Pharma Ltd
- Scinopharm Taiwan Ltd
- Dr Reddy's Laboratories Ltd
- Pfizer Inc
Notable Milestones in Fondaparinux Industry Sector
The Fondaparinux industry has witnessed several significant developments that have impacted its market dynamics and trajectory. These milestones highlight evolving regulatory landscapes, market access initiatives, and the increasing importance of generic competition.
- September 2022: The Ministry of Health, Ontario, included fondaparinux (Arixtra and generic) under the exceptional access program reimbursement criteria for frequently requested drugs for the prophylaxis and treatment of venous thromboembolism. This decision is expected to improve patient access and utilization in Ontario.
- February 2022: The Joint Procurement Office (JPO) of China issued a notice to collect drug information related to the seventh batch of volume-based procurement (VBP) of fondaparinux, which will boost China's national bulk buying of drugs. This initiative is anticipated to significantly increase the volume and market penetration of fondaparinux in China, particularly for generic versions.
In-Depth Fondaparinux Industry Market Outlook
The Fondaparinux industry is projected to exhibit sustained growth, underpinned by persistent global demand for effective VTE management. Key growth accelerators, including the increasing prevalence of VTE and strategic government initiatives promoting the use of essential medicines, will continue to drive market expansion. The ongoing trend towards cost-effective healthcare solutions favors the robust growth of the generics segment, ensuring broader accessibility. Strategic partnerships and market expansion into underserved regions represent significant opportunities for future revenue generation. The industry is well-positioned to meet the evolving needs of healthcare systems and patients seeking reliable anticoagulation therapies.
- Future Market Potential: Continued strong demand driven by VTE incidence and an aging global population.
- Strategic Opportunities: Expansion into emerging markets and exploration of niche therapeutic applications.
- Generic Dominance: The generics segment is expected to maintain its leading position due to cost-effectiveness and increasing VBP programs.
- Innovation Focus: Continued emphasis on manufacturing efficiency, quality control, and supply chain resilience.
Fondaparinux Industry Segmentation
-
1. Product Type
- 1.1. Branded
- 1.2. Generics
Fondaparinux Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
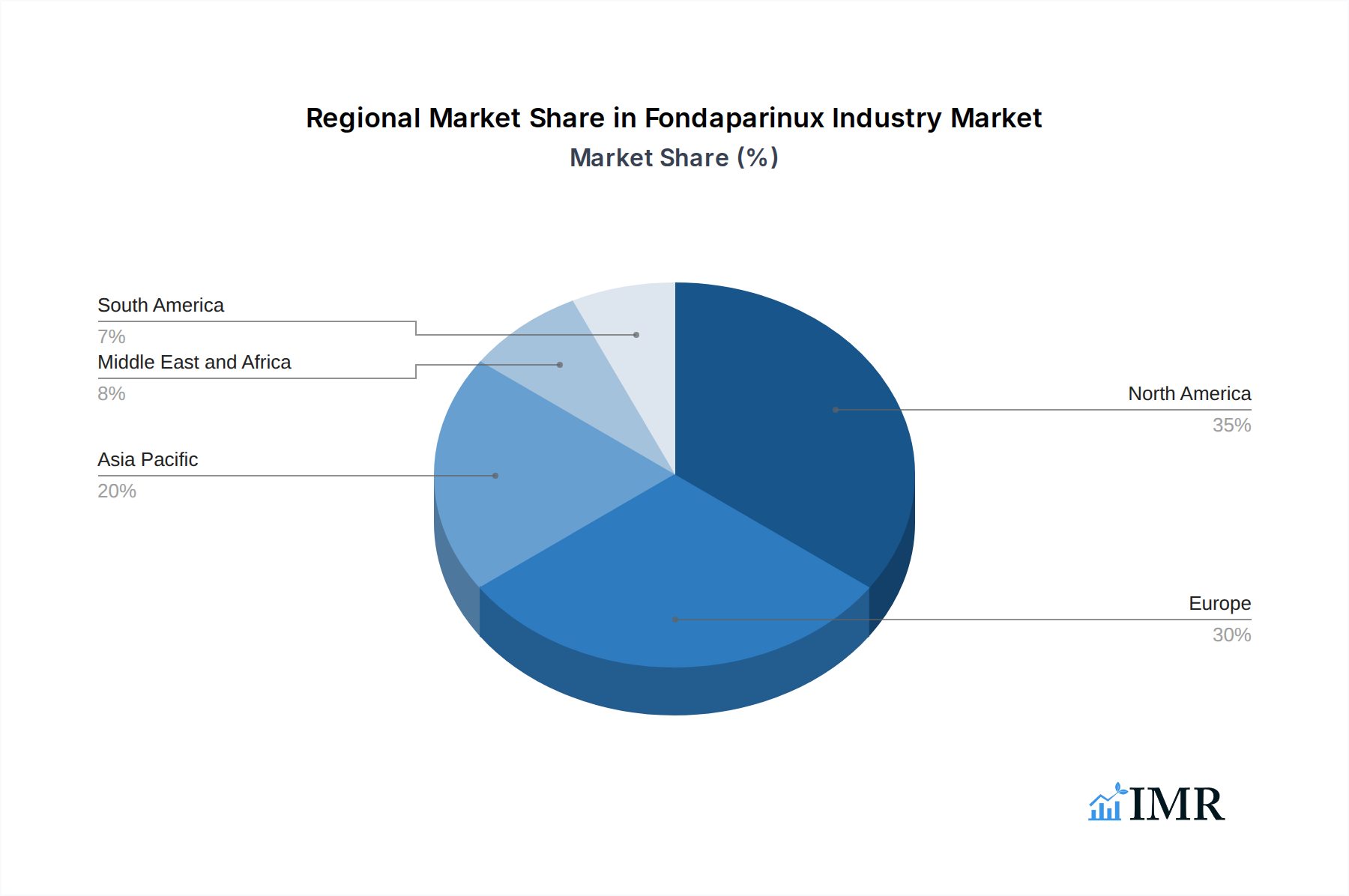
Fondaparinux Industry Regional Market Share

Geographic Coverage of Fondaparinux Industry
Fondaparinux Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.3% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. IMR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product Type
- 5.1.1. Branded
- 5.1.2. Generics
- 5.2. Market Analysis, Insights and Forecast - by Region
- 5.2.1. North America
- 5.2.2. Europe
- 5.2.3. Asia Pacific
- 5.2.4. Middle East and Africa
- 5.2.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Product Type
- 6. Global Fondaparinux Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product Type
- 6.1.1. Branded
- 6.1.2. Generics
- 6.1. Market Analysis, Insights and Forecast - by Product Type
- 7. North America Fondaparinux Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Product Type
- 7.1.1. Branded
- 7.1.2. Generics
- 7.1. Market Analysis, Insights and Forecast - by Product Type
- 8. Europe Fondaparinux Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Product Type
- 8.1.1. Branded
- 8.1.2. Generics
- 8.1. Market Analysis, Insights and Forecast - by Product Type
- 9. Asia Pacific Fondaparinux Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Product Type
- 9.1.1. Branded
- 9.1.2. Generics
- 9.1. Market Analysis, Insights and Forecast - by Product Type
- 10. Middle East and Africa Fondaparinux Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Product Type
- 10.1.1. Branded
- 10.1.2. Generics
- 10.1. Market Analysis, Insights and Forecast - by Product Type
- 11. South America Fondaparinux Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Product Type
- 11.1.1. Branded
- 11.1.2. Generics
- 11.1. Market Analysis, Insights and Forecast - by Product Type
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Boehringer Ingelheim International GmbH
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Viatris Inc
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Eugia
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Apotex Inc
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 GSK plc
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 Jiangsu Hengrui Medicine Co Ltd
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Aurobindo Pharma Ltd
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Scinopharm Taiwan Ltd
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Dr Reddy's Laboratories Ltd
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.10 Pfizer Inc
- 12.1.10.1. Company Overview
- 12.1.10.2. Products
- 12.1.10.3. Company Financials
- 12.1.10.4. SWOT Analysis
- 12.1.1 Boehringer Ingelheim International GmbH
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Fondaparinux Industry Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: Global Fondaparinux Industry Volume Breakdown (k unit, %) by Region 2025 & 2033
- Figure 3: North America Fondaparinux Industry Revenue (undefined), by Product Type 2025 & 2033
- Figure 4: North America Fondaparinux Industry Volume (k unit), by Product Type 2025 & 2033
- Figure 5: North America Fondaparinux Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 6: North America Fondaparinux Industry Volume Share (%), by Product Type 2025 & 2033
- Figure 7: North America Fondaparinux Industry Revenue (undefined), by Country 2025 & 2033
- Figure 8: North America Fondaparinux Industry Volume (k unit), by Country 2025 & 2033
- Figure 9: North America Fondaparinux Industry Revenue Share (%), by Country 2025 & 2033
- Figure 10: North America Fondaparinux Industry Volume Share (%), by Country 2025 & 2033
- Figure 11: Europe Fondaparinux Industry Revenue (undefined), by Product Type 2025 & 2033
- Figure 12: Europe Fondaparinux Industry Volume (k unit), by Product Type 2025 & 2033
- Figure 13: Europe Fondaparinux Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 14: Europe Fondaparinux Industry Volume Share (%), by Product Type 2025 & 2033
- Figure 15: Europe Fondaparinux Industry Revenue (undefined), by Country 2025 & 2033
- Figure 16: Europe Fondaparinux Industry Volume (k unit), by Country 2025 & 2033
- Figure 17: Europe Fondaparinux Industry Revenue Share (%), by Country 2025 & 2033
- Figure 18: Europe Fondaparinux Industry Volume Share (%), by Country 2025 & 2033
- Figure 19: Asia Pacific Fondaparinux Industry Revenue (undefined), by Product Type 2025 & 2033
- Figure 20: Asia Pacific Fondaparinux Industry Volume (k unit), by Product Type 2025 & 2033
- Figure 21: Asia Pacific Fondaparinux Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 22: Asia Pacific Fondaparinux Industry Volume Share (%), by Product Type 2025 & 2033
- Figure 23: Asia Pacific Fondaparinux Industry Revenue (undefined), by Country 2025 & 2033
- Figure 24: Asia Pacific Fondaparinux Industry Volume (k unit), by Country 2025 & 2033
- Figure 25: Asia Pacific Fondaparinux Industry Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Fondaparinux Industry Volume Share (%), by Country 2025 & 2033
- Figure 27: Middle East and Africa Fondaparinux Industry Revenue (undefined), by Product Type 2025 & 2033
- Figure 28: Middle East and Africa Fondaparinux Industry Volume (k unit), by Product Type 2025 & 2033
- Figure 29: Middle East and Africa Fondaparinux Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 30: Middle East and Africa Fondaparinux Industry Volume Share (%), by Product Type 2025 & 2033
- Figure 31: Middle East and Africa Fondaparinux Industry Revenue (undefined), by Country 2025 & 2033
- Figure 32: Middle East and Africa Fondaparinux Industry Volume (k unit), by Country 2025 & 2033
- Figure 33: Middle East and Africa Fondaparinux Industry Revenue Share (%), by Country 2025 & 2033
- Figure 34: Middle East and Africa Fondaparinux Industry Volume Share (%), by Country 2025 & 2033
- Figure 35: South America Fondaparinux Industry Revenue (undefined), by Product Type 2025 & 2033
- Figure 36: South America Fondaparinux Industry Volume (k unit), by Product Type 2025 & 2033
- Figure 37: South America Fondaparinux Industry Revenue Share (%), by Product Type 2025 & 2033
- Figure 38: South America Fondaparinux Industry Volume Share (%), by Product Type 2025 & 2033
- Figure 39: South America Fondaparinux Industry Revenue (undefined), by Country 2025 & 2033
- Figure 40: South America Fondaparinux Industry Volume (k unit), by Country 2025 & 2033
- Figure 41: South America Fondaparinux Industry Revenue Share (%), by Country 2025 & 2033
- Figure 42: South America Fondaparinux Industry Volume Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Fondaparinux Industry Revenue undefined Forecast, by Product Type 2020 & 2033
- Table 2: Global Fondaparinux Industry Volume k unit Forecast, by Product Type 2020 & 2033
- Table 3: Global Fondaparinux Industry Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Fondaparinux Industry Volume k unit Forecast, by Region 2020 & 2033
- Table 5: Global Fondaparinux Industry Revenue undefined Forecast, by Product Type 2020 & 2033
- Table 6: Global Fondaparinux Industry Volume k unit Forecast, by Product Type 2020 & 2033
- Table 7: Global Fondaparinux Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 8: Global Fondaparinux Industry Volume k unit Forecast, by Country 2020 & 2033
- Table 9: United States Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: United States Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 11: Canada Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 12: Canada Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 13: Mexico Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Mexico Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 15: Global Fondaparinux Industry Revenue undefined Forecast, by Product Type 2020 & 2033
- Table 16: Global Fondaparinux Industry Volume k unit Forecast, by Product Type 2020 & 2033
- Table 17: Global Fondaparinux Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 18: Global Fondaparinux Industry Volume k unit Forecast, by Country 2020 & 2033
- Table 19: Germany Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 21: United Kingdom Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: United Kingdom Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 23: France Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: France Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 25: Italy Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Italy Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 27: Spain Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Spain Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 29: Rest of Europe Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 30: Rest of Europe Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 31: Global Fondaparinux Industry Revenue undefined Forecast, by Product Type 2020 & 2033
- Table 32: Global Fondaparinux Industry Volume k unit Forecast, by Product Type 2020 & 2033
- Table 33: Global Fondaparinux Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 34: Global Fondaparinux Industry Volume k unit Forecast, by Country 2020 & 2033
- Table 35: China Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: China Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 37: Japan Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 38: Japan Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 39: India Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 40: India Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 41: Australia Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Australia Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 43: South Korea Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: South Korea Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 45: Rest of Asia Pacific Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 47: Global Fondaparinux Industry Revenue undefined Forecast, by Product Type 2020 & 2033
- Table 48: Global Fondaparinux Industry Volume k unit Forecast, by Product Type 2020 & 2033
- Table 49: Global Fondaparinux Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 50: Global Fondaparinux Industry Volume k unit Forecast, by Country 2020 & 2033
- Table 51: GCC Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 52: GCC Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 53: South Africa Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 54: South Africa Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 55: Rest of Middle East and Africa Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 56: Rest of Middle East and Africa Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 57: Global Fondaparinux Industry Revenue undefined Forecast, by Product Type 2020 & 2033
- Table 58: Global Fondaparinux Industry Volume k unit Forecast, by Product Type 2020 & 2033
- Table 59: Global Fondaparinux Industry Revenue undefined Forecast, by Country 2020 & 2033
- Table 60: Global Fondaparinux Industry Volume k unit Forecast, by Country 2020 & 2033
- Table 61: Brazil Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 62: Brazil Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 63: Argentina Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 64: Argentina Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
- Table 65: Rest of South America Fondaparinux Industry Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 66: Rest of South America Fondaparinux Industry Volume (k unit) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Fondaparinux Industry?
The projected CAGR is approximately 6.3%.
2. Which companies are prominent players in the Fondaparinux Industry?
Key companies in the market include Boehringer Ingelheim International GmbH, Viatris Inc, Eugia, Apotex Inc , GSK plc, Jiangsu Hengrui Medicine Co Ltd, Aurobindo Pharma Ltd, Scinopharm Taiwan Ltd, Dr Reddy's Laboratories Ltd, Pfizer Inc.
3. What are the main segments of the Fondaparinux Industry?
The market segments include Product Type.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
Growing Burden of Cardiovascular Disease; Increasing Geriatric Population; Increasing Research and Development Activities.
6. What are the notable trends driving market growth?
The Generics Segment is Expected to Have the Highest CAGR over the Forecast Period.
7. Are there any restraints impacting market growth?
Lack of Awareness about Venous Thromboembolism Conditions.
8. Can you provide examples of recent developments in the market?
September 2022: The Ministry of Health, Ontario, included fondaparinux (Arixtra and generic) under the exceptional access program reimbursement criteria for frequently requested drugs for the prophylaxis and treatment of venous thromboembolism.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A and volume, measured in k unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Fondaparinux Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Fondaparinux Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Fondaparinux Industry?
To stay informed about further developments, trends, and reports in the Fondaparinux Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
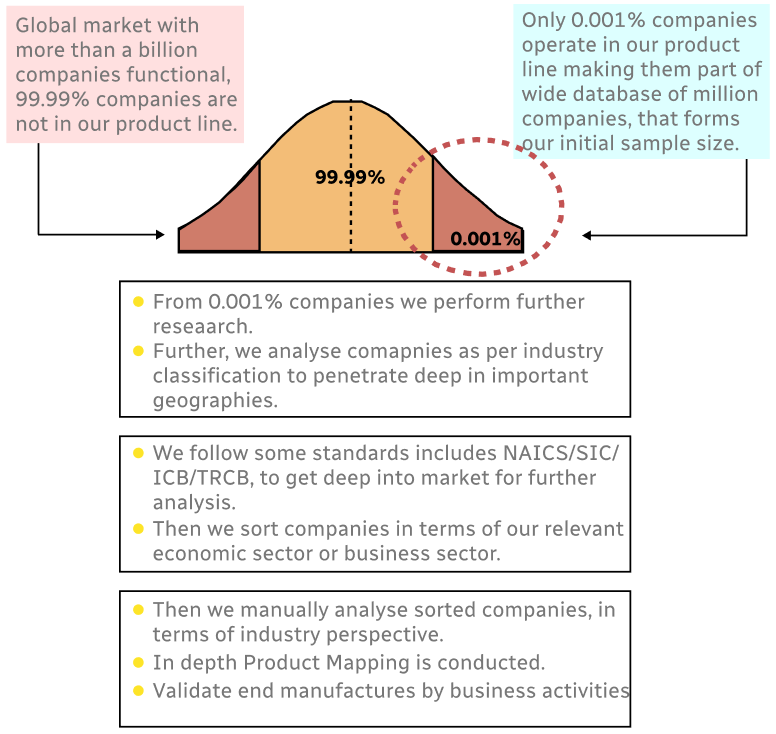
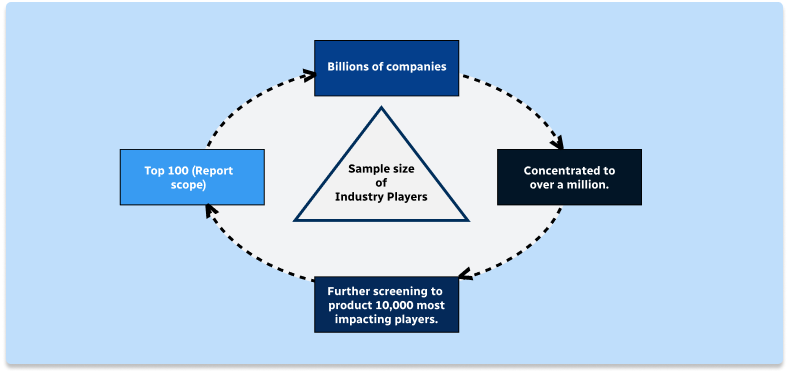
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
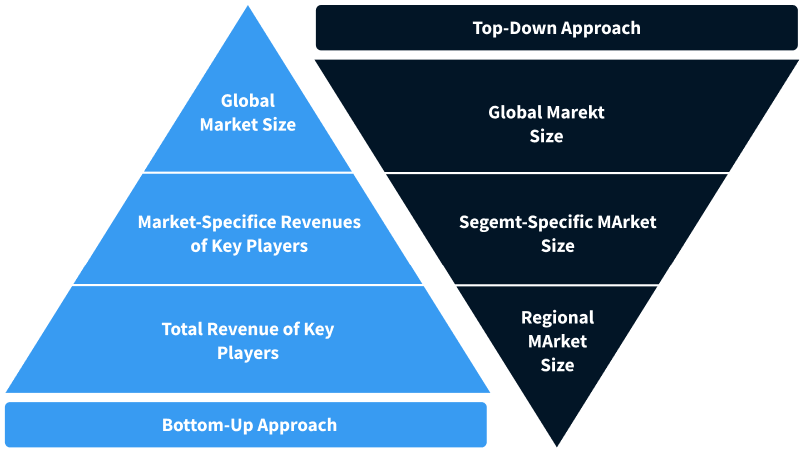
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
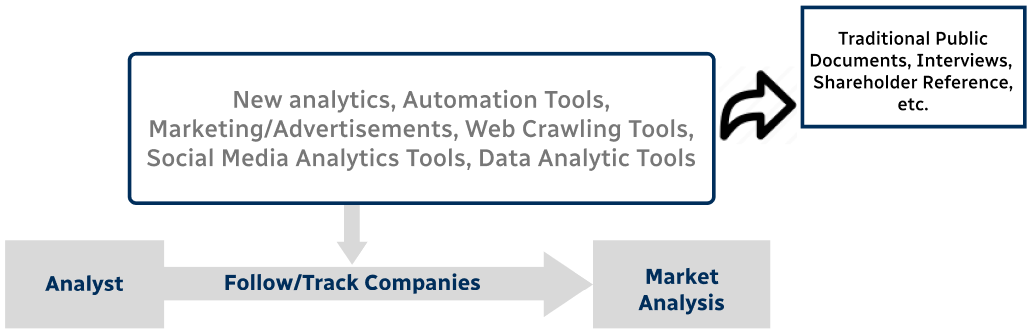
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


