Key Insights
The North America Autoimmune Disease Diagnostics Market is poised for significant expansion, projected to reach $2.05 Million by 2025, with a robust Compound Annual Growth Rate (CAGR) of 5.53% through 2033. This growth is propelled by an increasing prevalence of autoimmune disorders, heightened awareness among patients and healthcare providers, and advancements in diagnostic technologies. Key drivers include the rising incidence of conditions like Rheumatoid Arthritis, Systemic Lupus Erythematosus (SLE), and Multiple Sclerosis, which necessitate early and accurate diagnosis for effective management. Furthermore, technological innovations in immunologic assays and antibody tests are enhancing diagnostic precision, enabling earlier detection and better patient outcomes. The market's trajectory is also influenced by increasing healthcare expenditure and government initiatives aimed at improving access to diagnostics, particularly in countries like the United States.
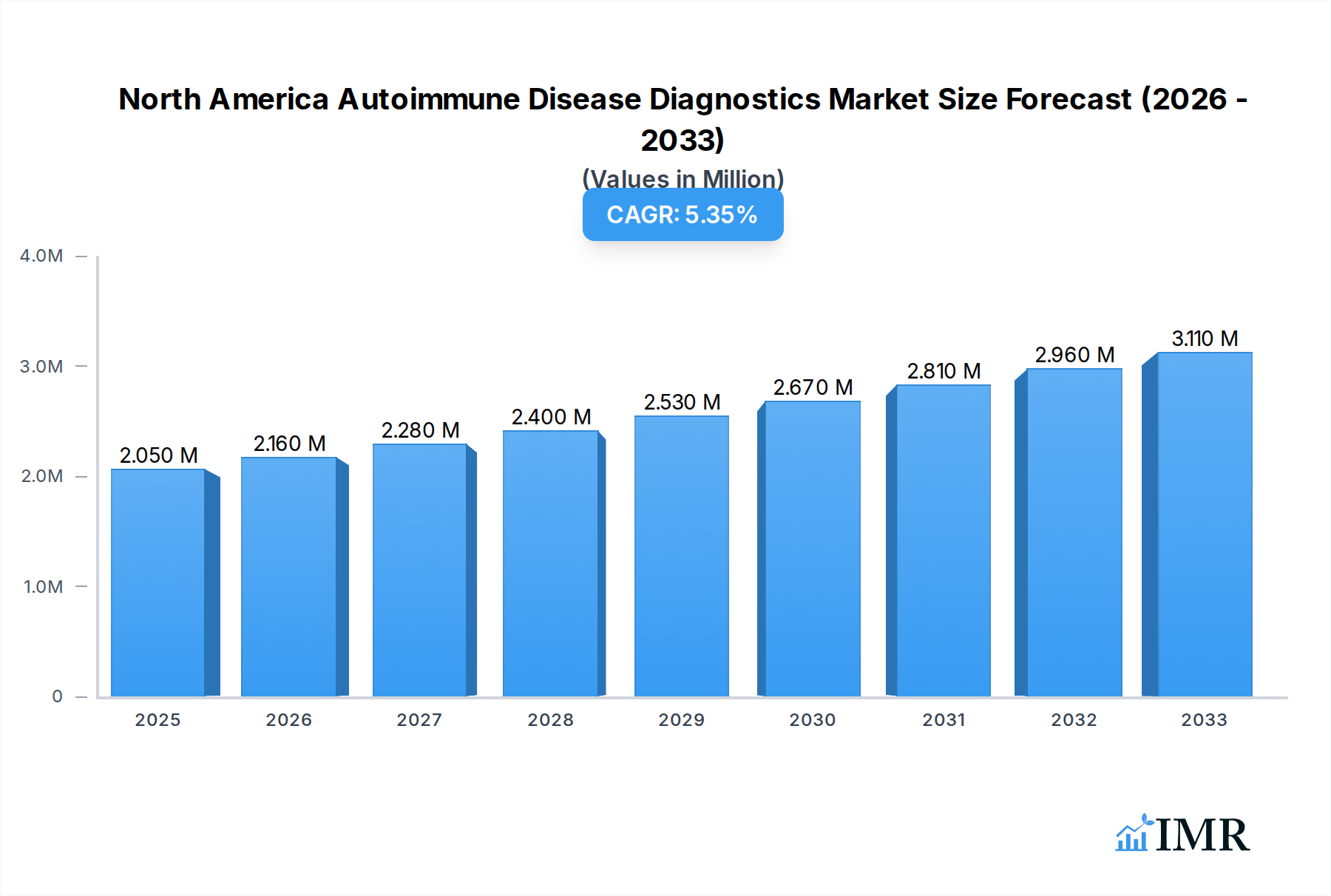
North America Autoimmune Disease Diagnostics Market Market Size (In Million)

The market is segmented across various disease types, including Systemic Autoimmune Diseases (such as Rheumatoid Arthritis, Psoriasis, SLE, and Multiple Sclerosis) and Localized Autoimmune Diseases (including Inflammatory Bowel Disease, Type 1 Diabetes, and Thyroid disorders). Diagnostic test categories span Regular Laboratory Tests, Inflammatory Markers, Immunologic Assays, and Antibody Tests, with immunologic and antibody tests expected to witness substantial growth due to their specificity and sensitivity. While the market is driven by these factors, potential restraints include the high cost of advanced diagnostic equipment and the need for skilled personnel to operate them. Geographically, the United States dominates the North American market due to its advanced healthcare infrastructure and high patient population, followed by Canada and Mexico. Key players like Abbott Laboratories, Siemens Healthineers Inc., and Thermo Fisher Scientific are actively investing in research and development to introduce novel diagnostic solutions, further shaping the market landscape.
North America Autoimmune Disease Diagnostics Market Company Market Share

North America Autoimmune Disease Diagnostics Market: Comprehensive Analysis and Future Outlook (2019–2033)
Report Description:
Dive deep into the dynamic North America Autoimmune Disease Diagnostics Market with this comprehensive report, meticulously designed to offer unparalleled insights for industry stakeholders. Spanning from 2019 to 2033, with a detailed focus on the base year 2025 and the forecast period 2025–2033, this report meticulously analyzes market dynamics, growth trends, regional dominance, product landscape, key drivers, emerging opportunities, and the strategic moves of leading players. Leveraging high-traffic SEO keywords such as "autoimmune disease diagnostics," "North America diagnostics market," "rheumatoid arthritis diagnosis," "lupus testing," "multiple sclerosis diagnostics," and "inflammatory bowel disease testing," this report is optimized for maximum search engine visibility and engagement. We provide granular analysis of parent and child markets, including detailed breakdowns by Disease Type (Systemic Autoimmune Disease, Localized Autoimmune Disease) and Diagnostic Test categories (Inflammatory Markers, Antibody Tests, Immunologic Assays). Essential quantitative data, including market share percentages, CAGR projections, and unit values in Millions, are presented to support strategic decision-making. This report is your indispensable guide to navigating the complexities and capitalizing on the significant growth potential within the North America autoimmune disease diagnostics sector.
North America Autoimmune Disease Diagnostics Market Dynamics & Structure
The North America autoimmune disease diagnostics market is characterized by a moderately concentrated landscape, with a few prominent players like Abbott Laboratories, Siemens Healthineers Inc., Thermo Fisher Scientific, and F Hoffmann-la Roche holding significant market shares. Technological innovation serves as a primary driver, with continuous advancements in assay development, automation, and multiplexing capabilities enhancing diagnostic accuracy and speed. The regulatory framework, particularly stringent FDA approvals in the United States and Health Canada's oversight, shapes product development and market entry strategies. Competitive product substitutes are evolving, with the increasing adoption of molecular diagnostics and advanced imaging techniques offering alternative diagnostic pathways. End-user demographics are shifting, with an aging population and a growing prevalence of autoimmune conditions driving demand. Mergers and Acquisitions (M&A) are a notable trend, aimed at expanding product portfolios, geographical reach, and technological capabilities. For instance, the acquisition of smaller, innovative diagnostic firms by larger corporations is common, further consolidating the market.
- Market Concentration: Moderate, with key players dominating specific segments.
- Technological Innovation: Driven by enhanced assay sensitivity, automation, and point-of-care solutions.
- Regulatory Framework: Robust, requiring stringent validation and approval processes.
- Competitive Landscape: Features a mix of established diagnostics giants and specialized biotechnology firms.
- End-User Demographics: Increasing prevalence of autoimmune diseases and an aging population are key drivers.
- M&A Trends: Strategic acquisitions and partnerships to enhance market presence and technological prowess.
North America Autoimmune Disease Diagnostics Market Growth Trends & Insights
The North America autoimmune disease diagnostics market is poised for robust expansion driven by a confluence of factors including increasing disease prevalence, heightened awareness, and significant advancements in diagnostic technologies. The market size is projected to witness a compound annual growth rate (CAGR) of approximately 7.5% from 2025 to 2033. This growth is underpinned by a rising incidence of systemic autoimmune diseases like Rheumatoid Arthritis (RA) and Systemic Lupus Erythematosus (SLE), as well as localized conditions such as Type 1 Diabetes and Thyroid disorders. Adoption rates of sophisticated diagnostic tests, particularly immunologic assays and antibody tests, are accelerating due to their improved accuracy and ability to detect autoimmune markers earlier in disease progression. Technological disruptions, including the integration of artificial intelligence (AI) in image analysis for conditions like Multiple Sclerosis (MS) and the development of highly sensitive multiplex assays capable of detecting multiple autoantibodies simultaneously, are revolutionizing diagnosis. Consumer behavior is also shifting, with patients becoming more proactive in seeking early diagnosis and personalized treatment plans, thus fueling demand for comprehensive diagnostic solutions. The continuous innovation in developing more accurate, faster, and cost-effective diagnostic kits is a critical factor influencing market penetration. For example, the development of novel biomarkers and the refinement of existing immunoassay platforms are enabling earlier and more precise diagnoses, reducing the time to treatment and improving patient outcomes. Furthermore, the increasing focus on personalized medicine is driving the demand for genetic testing and companion diagnostics for autoimmune diseases.
Dominant Regions, Countries, or Segments in North America Autoimmune Disease Diagnostics Market
The United States stands as the dominant region within the North America autoimmune disease diagnostics market, driven by several compelling factors that contribute to its significant market share and sustained growth potential. The country boasts a well-established and advanced healthcare infrastructure, characterized by a high density of specialized clinics, hospitals, and diagnostic laboratories equipped with cutting-edge technology. This infrastructure is crucial for the widespread adoption and accessibility of complex autoimmune disease diagnostic tests. Furthermore, the United States has the highest prevalence of many autoimmune diseases, including Rheumatoid Arthritis, Multiple Sclerosis, and Systemic Lupus Erythematosus, creating a substantial and consistent demand for diagnostic solutions.
Within the Disease Type segment, Systemic Autoimmune Diseases are the primary revenue generators. Specifically, Rheumatoid Arthritis (RA) and Systemic Lupus Erythematosus (SLE) represent significant market segments due to their high prevalence, chronic nature, and the continuous need for monitoring and management. The increasing diagnosis and management of Multiple Sclerosis (MS) also contribute substantially to the market's growth.
In terms of Diagnostic Test categories, Immunologic Assays and Antibody Tests are the leading segments. These tests, including enzyme-linked immunosorbent assays (ELISA), chemiluminescent immunoassays (CLIA), and immunofluorescence assays (IFA), are fundamental for identifying specific autoantibodies that are hallmarks of autoimmune conditions. The development of more sensitive and specific antibody panels for conditions like Sjogren's syndrome, myositis, and vasculitis further solidifies the dominance of these testing modalities. The advancements in Inflammatory Markers testing also play a crucial role in the initial screening and assessment of inflammatory activity associated with autoimmune diseases.
- Dominant Country: United States, due to its advanced healthcare system, high disease prevalence, and robust R&D investments.
- Dominant Disease Type Segment: Systemic Autoimmune Diseases (Rheumatoid Arthritis, Systemic Lupus Erythematosus, Multiple Sclerosis) driven by high incidence and chronic management needs.
- Dominant Diagnostic Test Segment: Immunologic Assays and Antibody Tests, crucial for identifying specific autoantibodies and disease biomarkers.
- Key Drivers:
- High prevalence of autoimmune conditions.
- Advanced healthcare infrastructure and accessibility to diagnostics.
- Significant investment in R&D for novel diagnostic tools.
- Growing awareness and demand for early and accurate diagnosis.
- Supportive reimbursement policies for diagnostic procedures.
- Growth Potential: Continued innovation in multiplex assays and personalized diagnostics will further fuel growth in these dominant segments.
North America Autoimmune Disease Diagnostics Market Product Landscape
The product landscape in the North America autoimmune disease diagnostics market is characterized by a diverse array of innovative diagnostic tools and platforms. Leading companies are focusing on developing highly sensitive and specific assays for detecting a broad spectrum of autoantibodies, aiding in the early and accurate diagnosis of various autoimmune conditions. Innovations include the development of multiplex assays that can simultaneously test for multiple autoantibodies from a single patient sample, significantly improving efficiency and reducing diagnostic time. Furthermore, advancements in automation and point-of-care testing are enhancing accessibility and convenience for both healthcare providers and patients. Examples of product innovation include the development of new immunoassay kits for specific autoimmune markers and the integration of digital technologies for data analysis and interpretation. The unique selling proposition of these products often lies in their enhanced performance metrics such as higher sensitivity, specificity, and faster turnaround times.
Key Drivers, Barriers & Challenges in North America Autoimmune Disease Diagnostics Market
The North America autoimmune disease diagnostics market is propelled by several key drivers, primarily the escalating prevalence of autoimmune conditions across all age groups. Heightened patient and physician awareness regarding the importance of early diagnosis and intervention significantly boosts demand for sophisticated diagnostic tools. Technological advancements, particularly in areas like multiplex immunoassay platforms and molecular diagnostics, are crucial for improving diagnostic accuracy and breadth. Furthermore, supportive government initiatives and increasing healthcare expenditure in regions like the United States contribute to market growth.
However, the market faces significant barriers and challenges. High development costs for new diagnostic assays and the stringent regulatory approval processes, especially in the US, can impede market entry for smaller players. Reimbursement policies for certain advanced diagnostic tests can be complex and vary across different healthcare systems, impacting adoption rates. The competitive pressure from established players and the need for continuous innovation to stay ahead also present challenges. Supply chain disruptions, as witnessed globally, can affect the availability of raw materials and finished diagnostic kits.
- Key Drivers:
- Rising prevalence of autoimmune diseases.
- Increased awareness and demand for early diagnosis.
- Technological advancements in diagnostic assays.
- Growing healthcare expenditure.
- Key Barriers & Challenges:
- High R&D and regulatory approval costs.
- Complex reimbursement landscape.
- Intense competition and need for rapid innovation.
- Potential supply chain disruptions.
Emerging Opportunities in North America Autoimmune Disease Diagnostics Market
Emerging opportunities within the North America autoimmune disease diagnostics market are abundant, driven by unmet diagnostic needs and technological advancements. The development of novel biomarkers for early disease detection and prediction of treatment response presents a significant avenue for growth. Personalized diagnostics, including genetic testing for predisposition and companion diagnostics to guide targeted therapies, are gaining traction. The expansion of point-of-care diagnostic solutions for autoimmune diseases can enhance accessibility, especially in remote areas or for patients with mobility issues. Furthermore, the increasing focus on less common autoimmune diseases offers untapped market potential, necessitating the development of specialized diagnostic kits and panels. The integration of AI and machine learning for analyzing complex diagnostic data and identifying subtle disease patterns also represents a promising frontier.
Growth Accelerators in the North America Autoimmune Disease Diagnostics Market Industry
Several factors are acting as growth accelerators for the North America autoimmune disease diagnostics market. Continuous technological breakthroughs, such as the development of highly sensitive and specific multiplex assays and advancements in liquid biopsy techniques, are significantly enhancing diagnostic capabilities. Strategic partnerships between diagnostic companies and pharmaceutical firms are accelerating the development and commercialization of companion diagnostics, crucial for the growing field of personalized medicine in autoimmune disorders. Market expansion strategies, including increased penetration in underserved regions and the development of more affordable diagnostic solutions, are also driving long-term growth. The growing emphasis on preventative healthcare and early disease intervention by healthcare systems and payers further fuels the demand for advanced diagnostic tools.
Key Players Shaping the North America Autoimmune Disease Diagnostics Market Market
- Abbott Laboratories
- Siemens Healthineers Inc.
- Thermo Fisher Scientific
- F Hoffmann-la Roche
- Grifols SA
- Werfen Group
- Bio-rad Laboratories
- Myriad Genetics
- Euroimmun AG (Perkinelmer Inc.)
- Oncimmune
- HYCOR Biomedical
- Trinity Biotech
Notable Milestones in North America Autoimmune Disease Diagnostics Market Sector
- February 2023: Edesa Biotech received approval from Health Canada for a phase II clinical trial of its EB06 monoclonal antibody candidate to treat vitiligo, a life-altering autoimmune disease.
- June 2022: Thermo Scientific received United States FDA clearance for the EliA RNA Pol III and EliA Rib-P tests for aiding in the diagnosis of systemic sclerosis and systemic lupus erythematosus (SLE).
In-Depth North America Autoimmune Disease Diagnostics Market Market Outlook
The future outlook for the North America autoimmune disease diagnostics market is exceptionally positive, fueled by ongoing technological innovation and an expanding understanding of autoimmune pathology. Growth accelerators like the development of novel biomarkers, advancements in next-generation sequencing for genetic predisposition studies, and the increasing utility of AI in diagnostic interpretation will continue to shape the market. Strategic partnerships focused on co-developing diagnostic and therapeutic solutions will further enhance market integration and efficiency. The market is poised to benefit from the growing emphasis on early diagnosis and personalized treatment approaches, leading to a greater demand for comprehensive and highly accurate diagnostic testing. Emerging opportunities in areas like rare autoimmune diseases and the integration of digital health platforms into diagnostic workflows present significant untapped potential for market expansion and sustained growth.
North America Autoimmune Disease Diagnostics Market Segmentation
-
1. Disease Type
-
1.1. Systemic Autoimmune Disease
- 1.1.1. Rheumatoid Arthritis
- 1.1.2. Psoriasis
- 1.1.3. Systemic Lupus Erythematosus (SLE)
- 1.1.4. Multiple Sclerosis
- 1.1.5. Other Disease Types
-
1.2. Localized Autoimmune Disease
- 1.2.1. Inflammatory Bowel disease
- 1.2.2. Type 1 Diabetes
- 1.2.3. Thyroid
- 1.2.4. Other Localized Autoimmune Diseases
-
1.1. Systemic Autoimmune Disease
-
2. Diagnostic Test
- 2.1. Regular Laboratory Tests
- 2.2. Inflammatory Markers
- 2.3. Immunologic Assays
- 2.4. Antibody Tests
- 2.5. Other Tests
-
3. Geography
- 3.1. United States
- 3.2. Canada
- 3.3. Mexico
North America Autoimmune Disease Diagnostics Market Segmentation By Geography
- 1. United States
- 2. Canada
- 3. Mexico
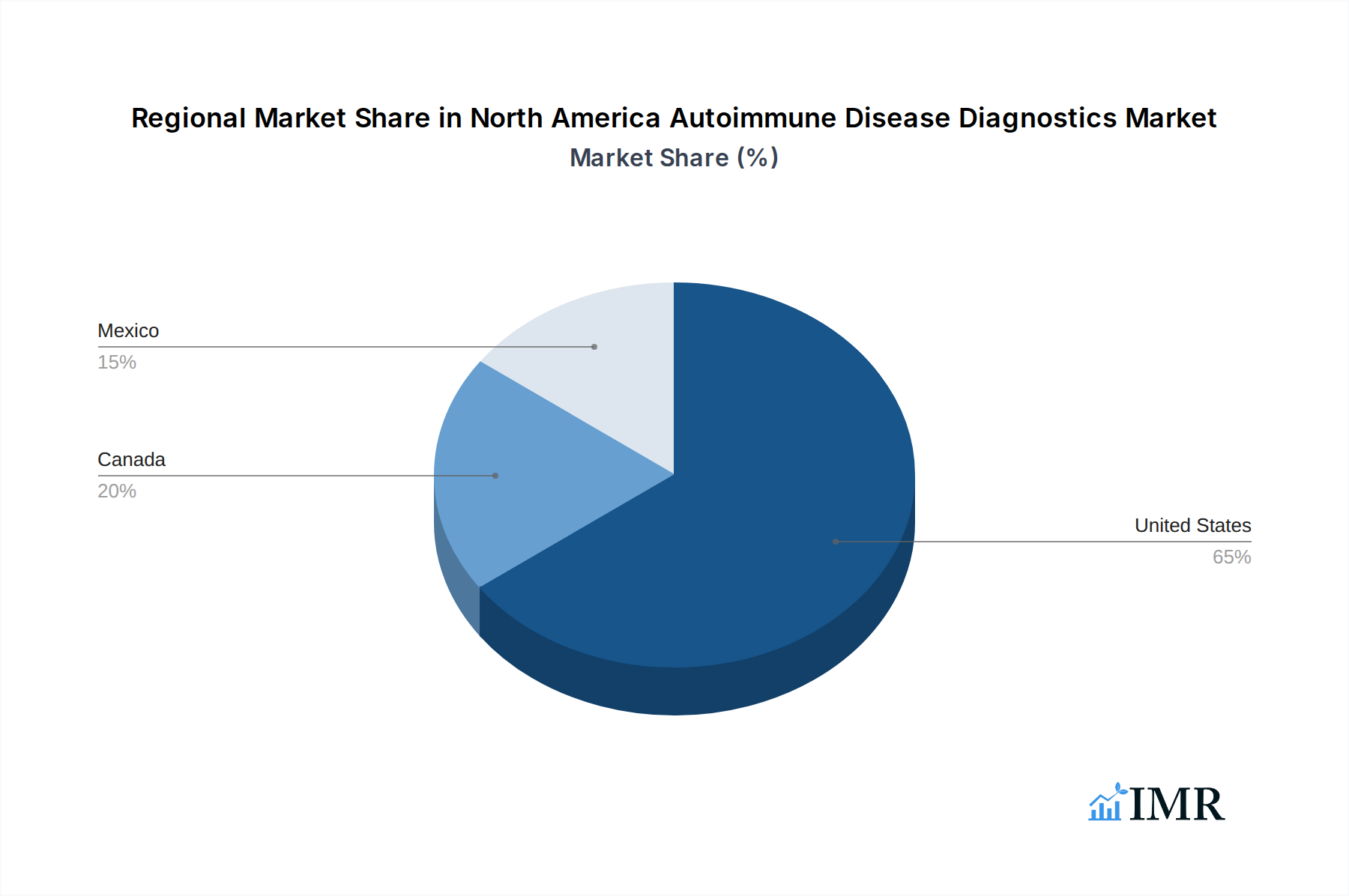
North America Autoimmune Disease Diagnostics Market Regional Market Share

Geographic Coverage of North America Autoimmune Disease Diagnostics Market
North America Autoimmune Disease Diagnostics Market REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.53% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. IMR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Disease Type
- 5.1.1. Systemic Autoimmune Disease
- 5.1.1.1. Rheumatoid Arthritis
- 5.1.1.2. Psoriasis
- 5.1.1.3. Systemic Lupus Erythematosus (SLE)
- 5.1.1.4. Multiple Sclerosis
- 5.1.1.5. Other Disease Types
- 5.1.2. Localized Autoimmune Disease
- 5.1.2.1. Inflammatory Bowel disease
- 5.1.2.2. Type 1 Diabetes
- 5.1.2.3. Thyroid
- 5.1.2.4. Other Localized Autoimmune Diseases
- 5.1.1. Systemic Autoimmune Disease
- 5.2. Market Analysis, Insights and Forecast - by Diagnostic Test
- 5.2.1. Regular Laboratory Tests
- 5.2.2. Inflammatory Markers
- 5.2.3. Immunologic Assays
- 5.2.4. Antibody Tests
- 5.2.5. Other Tests
- 5.3. Market Analysis, Insights and Forecast - by Geography
- 5.3.1. United States
- 5.3.2. Canada
- 5.3.3. Mexico
- 5.4. Market Analysis, Insights and Forecast - by Region
- 5.4.1. United States
- 5.4.2. Canada
- 5.4.3. Mexico
- 5.1. Market Analysis, Insights and Forecast - by Disease Type
- 6. North America Autoimmune Disease Diagnostics Market Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Disease Type
- 6.1.1. Systemic Autoimmune Disease
- 6.1.1.1. Rheumatoid Arthritis
- 6.1.1.2. Psoriasis
- 6.1.1.3. Systemic Lupus Erythematosus (SLE)
- 6.1.1.4. Multiple Sclerosis
- 6.1.1.5. Other Disease Types
- 6.1.2. Localized Autoimmune Disease
- 6.1.2.1. Inflammatory Bowel disease
- 6.1.2.2. Type 1 Diabetes
- 6.1.2.3. Thyroid
- 6.1.2.4. Other Localized Autoimmune Diseases
- 6.1.1. Systemic Autoimmune Disease
- 6.2. Market Analysis, Insights and Forecast - by Diagnostic Test
- 6.2.1. Regular Laboratory Tests
- 6.2.2. Inflammatory Markers
- 6.2.3. Immunologic Assays
- 6.2.4. Antibody Tests
- 6.2.5. Other Tests
- 6.3. Market Analysis, Insights and Forecast - by Geography
- 6.3.1. United States
- 6.3.2. Canada
- 6.3.3. Mexico
- 6.1. Market Analysis, Insights and Forecast - by Disease Type
- 7. United States North America Autoimmune Disease Diagnostics Market Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Disease Type
- 7.1.1. Systemic Autoimmune Disease
- 7.1.1.1. Rheumatoid Arthritis
- 7.1.1.2. Psoriasis
- 7.1.1.3. Systemic Lupus Erythematosus (SLE)
- 7.1.1.4. Multiple Sclerosis
- 7.1.1.5. Other Disease Types
- 7.1.2. Localized Autoimmune Disease
- 7.1.2.1. Inflammatory Bowel disease
- 7.1.2.2. Type 1 Diabetes
- 7.1.2.3. Thyroid
- 7.1.2.4. Other Localized Autoimmune Diseases
- 7.1.1. Systemic Autoimmune Disease
- 7.2. Market Analysis, Insights and Forecast - by Diagnostic Test
- 7.2.1. Regular Laboratory Tests
- 7.2.2. Inflammatory Markers
- 7.2.3. Immunologic Assays
- 7.2.4. Antibody Tests
- 7.2.5. Other Tests
- 7.3. Market Analysis, Insights and Forecast - by Geography
- 7.3.1. United States
- 7.3.2. Canada
- 7.3.3. Mexico
- 7.1. Market Analysis, Insights and Forecast - by Disease Type
- 8. Canada North America Autoimmune Disease Diagnostics Market Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Disease Type
- 8.1.1. Systemic Autoimmune Disease
- 8.1.1.1. Rheumatoid Arthritis
- 8.1.1.2. Psoriasis
- 8.1.1.3. Systemic Lupus Erythematosus (SLE)
- 8.1.1.4. Multiple Sclerosis
- 8.1.1.5. Other Disease Types
- 8.1.2. Localized Autoimmune Disease
- 8.1.2.1. Inflammatory Bowel disease
- 8.1.2.2. Type 1 Diabetes
- 8.1.2.3. Thyroid
- 8.1.2.4. Other Localized Autoimmune Diseases
- 8.1.1. Systemic Autoimmune Disease
- 8.2. Market Analysis, Insights and Forecast - by Diagnostic Test
- 8.2.1. Regular Laboratory Tests
- 8.2.2. Inflammatory Markers
- 8.2.3. Immunologic Assays
- 8.2.4. Antibody Tests
- 8.2.5. Other Tests
- 8.3. Market Analysis, Insights and Forecast - by Geography
- 8.3.1. United States
- 8.3.2. Canada
- 8.3.3. Mexico
- 8.1. Market Analysis, Insights and Forecast - by Disease Type
- 9. Mexico North America Autoimmune Disease Diagnostics Market Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Disease Type
- 9.1.1. Systemic Autoimmune Disease
- 9.1.1.1. Rheumatoid Arthritis
- 9.1.1.2. Psoriasis
- 9.1.1.3. Systemic Lupus Erythematosus (SLE)
- 9.1.1.4. Multiple Sclerosis
- 9.1.1.5. Other Disease Types
- 9.1.2. Localized Autoimmune Disease
- 9.1.2.1. Inflammatory Bowel disease
- 9.1.2.2. Type 1 Diabetes
- 9.1.2.3. Thyroid
- 9.1.2.4. Other Localized Autoimmune Diseases
- 9.1.1. Systemic Autoimmune Disease
- 9.2. Market Analysis, Insights and Forecast - by Diagnostic Test
- 9.2.1. Regular Laboratory Tests
- 9.2.2. Inflammatory Markers
- 9.2.3. Immunologic Assays
- 9.2.4. Antibody Tests
- 9.2.5. Other Tests
- 9.3. Market Analysis, Insights and Forecast - by Geography
- 9.3.1. United States
- 9.3.2. Canada
- 9.3.3. Mexico
- 9.1. Market Analysis, Insights and Forecast - by Disease Type
- 10. Competitive Analysis
- 10.1. Company Profiles
- 10.1.1 Oncimmune
- 10.1.1.1. Company Overview
- 10.1.1.2. Products
- 10.1.1.3. Company Financials
- 10.1.1.4. SWOT Analysis
- 10.1.2 HYCOR Biomedical
- 10.1.2.1. Company Overview
- 10.1.2.2. Products
- 10.1.2.3. Company Financials
- 10.1.2.4. SWOT Analysis
- 10.1.3 Grifols SA
- 10.1.3.1. Company Overview
- 10.1.3.2. Products
- 10.1.3.3. Company Financials
- 10.1.3.4. SWOT Analysis
- 10.1.4 Werfen Group
- 10.1.4.1. Company Overview
- 10.1.4.2. Products
- 10.1.4.3. Company Financials
- 10.1.4.4. SWOT Analysis
- 10.1.5 Abbott Laboratories
- 10.1.5.1. Company Overview
- 10.1.5.2. Products
- 10.1.5.3. Company Financials
- 10.1.5.4. SWOT Analysis
- 10.1.6 Siemens Healthineers Inc
- 10.1.6.1. Company Overview
- 10.1.6.2. Products
- 10.1.6.3. Company Financials
- 10.1.6.4. SWOT Analysis
- 10.1.7 Bio-rad Laboratories
- 10.1.7.1. Company Overview
- 10.1.7.2. Products
- 10.1.7.3. Company Financials
- 10.1.7.4. SWOT Analysis
- 10.1.8 Myriad Genetics
- 10.1.8.1. Company Overview
- 10.1.8.2. Products
- 10.1.8.3. Company Financials
- 10.1.8.4. SWOT Analysis
- 10.1.9 Euroimmun AG (Perkinelmer Inc )
- 10.1.9.1. Company Overview
- 10.1.9.2. Products
- 10.1.9.3. Company Financials
- 10.1.9.4. SWOT Analysis
- 10.1.10 F Hoffmann-la Roche
- 10.1.10.1. Company Overview
- 10.1.10.2. Products
- 10.1.10.3. Company Financials
- 10.1.10.4. SWOT Analysis
- 10.1.11 Thermo Fisher Scientific
- 10.1.11.1. Company Overview
- 10.1.11.2. Products
- 10.1.11.3. Company Financials
- 10.1.11.4. SWOT Analysis
- 10.1.12 Trinity Biotech
- 10.1.12.1. Company Overview
- 10.1.12.2. Products
- 10.1.12.3. Company Financials
- 10.1.12.4. SWOT Analysis
- 10.1.1 Oncimmune
- 10.2. Market Entropy
- 10.2.1 Company's Key Areas Served
- 10.2.2 Recent Developments
- 10.3. Company Market Share Analysis 2025
- 10.3.1 Top 5 Companies Market Share Analysis
- 10.3.2 Top 3 Companies Market Share Analysis
- 10.4. List of Potential Customers
- 11. Research Methodology
List of Figures
- Figure 1: North America Autoimmune Disease Diagnostics Market Revenue Breakdown (Million, %) by Product 2025 & 2033
- Figure 2: North America Autoimmune Disease Diagnostics Market Share (%) by Company 2025
List of Tables
- Table 1: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Disease Type 2020 & 2033
- Table 2: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Disease Type 2020 & 2033
- Table 3: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Diagnostic Test 2020 & 2033
- Table 4: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Diagnostic Test 2020 & 2033
- Table 5: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Geography 2020 & 2033
- Table 6: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Geography 2020 & 2033
- Table 7: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Region 2020 & 2033
- Table 8: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Region 2020 & 2033
- Table 9: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Disease Type 2020 & 2033
- Table 10: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Disease Type 2020 & 2033
- Table 11: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Diagnostic Test 2020 & 2033
- Table 12: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Diagnostic Test 2020 & 2033
- Table 13: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Geography 2020 & 2033
- Table 14: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Geography 2020 & 2033
- Table 15: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Country 2020 & 2033
- Table 16: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 17: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Disease Type 2020 & 2033
- Table 18: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Disease Type 2020 & 2033
- Table 19: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Diagnostic Test 2020 & 2033
- Table 20: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Diagnostic Test 2020 & 2033
- Table 21: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Geography 2020 & 2033
- Table 22: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Geography 2020 & 2033
- Table 23: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Country 2020 & 2033
- Table 24: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Country 2020 & 2033
- Table 25: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Disease Type 2020 & 2033
- Table 26: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Disease Type 2020 & 2033
- Table 27: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Diagnostic Test 2020 & 2033
- Table 28: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Diagnostic Test 2020 & 2033
- Table 29: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Geography 2020 & 2033
- Table 30: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Geography 2020 & 2033
- Table 31: North America Autoimmune Disease Diagnostics Market Revenue Million Forecast, by Country 2020 & 2033
- Table 32: North America Autoimmune Disease Diagnostics Market Volume K Unit Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the North America Autoimmune Disease Diagnostics Market?
The projected CAGR is approximately 5.53%.
2. Which companies are prominent players in the North America Autoimmune Disease Diagnostics Market?
Key companies in the market include Oncimmune, HYCOR Biomedical, Grifols SA, Werfen Group, Abbott Laboratories, Siemens Healthineers Inc, Bio-rad Laboratories, Myriad Genetics, Euroimmun AG (Perkinelmer Inc ), F Hoffmann-la Roche, Thermo Fisher Scientific, Trinity Biotech.
3. What are the main segments of the North America Autoimmune Disease Diagnostics Market?
The market segments include Disease Type, Diagnostic Test, Geography.
4. Can you provide details about the market size?
The market size is estimated to be USD 2.05 Million as of 2022.
5. What are some drivers contributing to market growth?
Increasing Prevalence and Growing Public Awareness of Autoimmune Diseases; Technological Advancements in Autoimmune Disease Diagnostics.
6. What are the notable trends driving market growth?
Immunologic Assays Segment is Expected to Hold a Significant Market Share Over the Forecast Period.
7. Are there any restraints impacting market growth?
Slow Turnaround Time for Autoimmune Disease Diagnostic Test Results; High Frequency of False Positive Result.
8. Can you provide examples of recent developments in the market?
February 2023: Edesa Biotech received approval from Health Canada for a phase II clinical trial of its EB06 monoclonal antibody candidate to treat vitiligo, a life-altering autoimmune disease.
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in Million and volume, measured in K Unit.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "North America Autoimmune Disease Diagnostics Market," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the North America Autoimmune Disease Diagnostics Market report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the North America Autoimmune Disease Diagnostics Market?
To stay informed about further developments, trends, and reports in the North America Autoimmune Disease Diagnostics Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
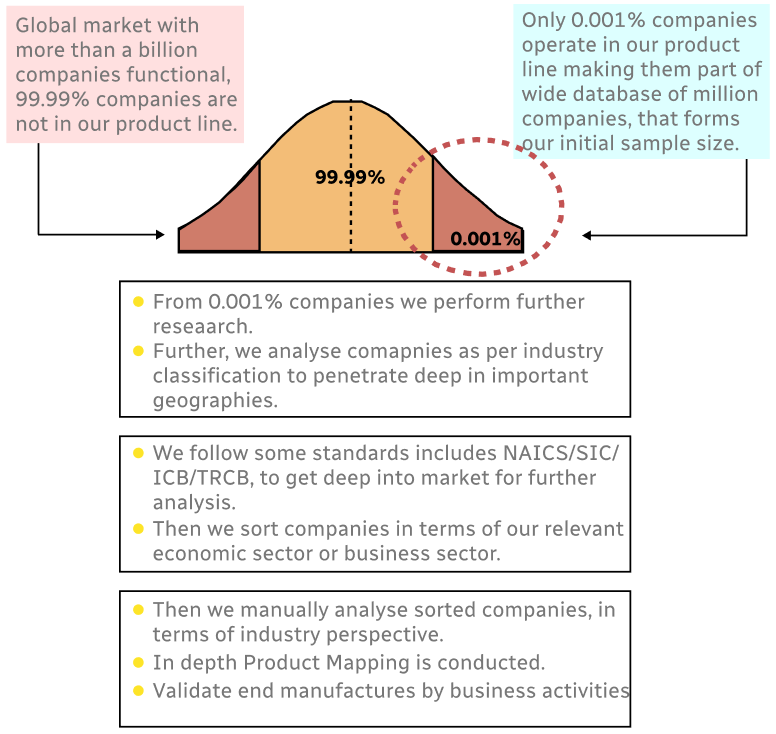
Step 1 - Identification of Relevant Samples Size from Population Database
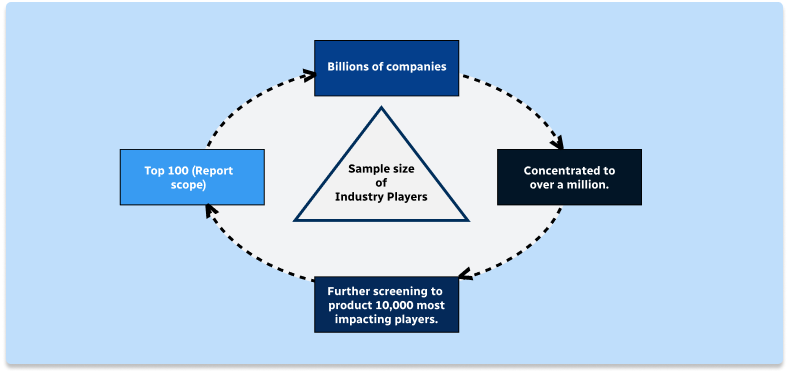
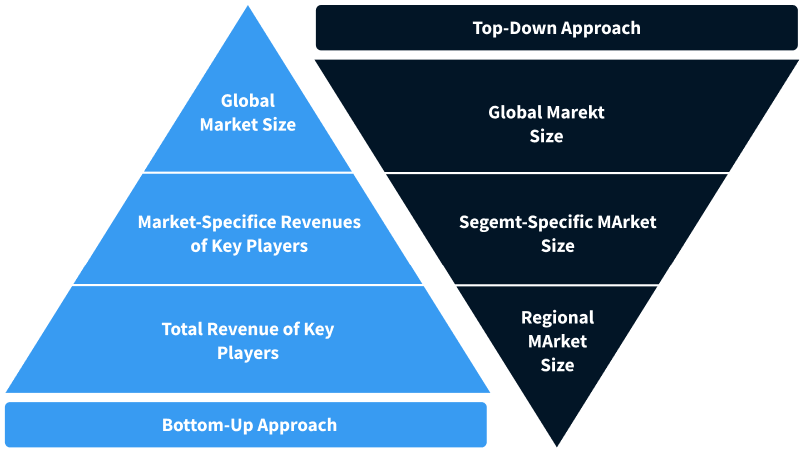
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
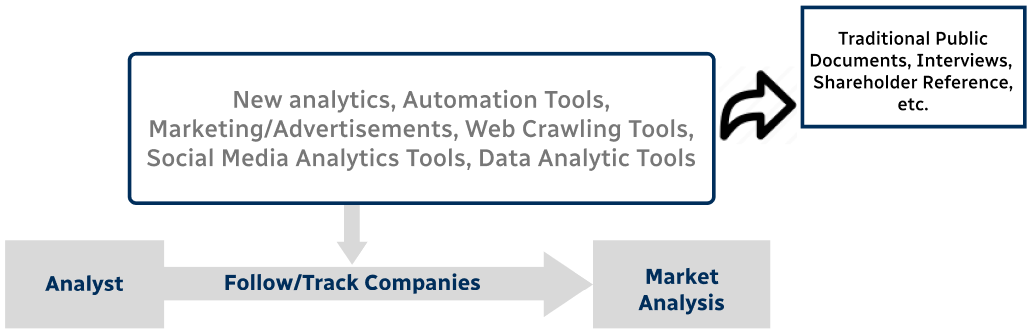
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


