Key Insights
The global Spine Biologics market is poised for significant expansion, with an estimated market size of $8.98 billion in 2025, driven by a robust CAGR of 15.24% projected through 2033. This remarkable growth trajectory is underpinned by several key factors. A rapidly aging global population is a primary driver, leading to an increased incidence of degenerative spine conditions and consequently, a higher demand for effective spinal fusion and regenerative treatments. Advancements in biomaterials and regenerative medicine are also fueling market expansion. The development of innovative bone graft substitutes, including advanced bone morphogenetic proteins (BMPs) and sophisticated synthetic bone grafts, offers superior efficacy and reduced risks compared to traditional methods. Furthermore, the increasing prevalence of spinal deformities and trauma cases, coupled with a growing awareness among patients and healthcare professionals regarding the benefits of spine biologics in achieving successful fusion outcomes and improving patient mobility, are significant growth catalysts. The expanding healthcare infrastructure, particularly in emerging economies, and the continuous investment in research and development by leading market players are further bolstering the market's positive outlook.
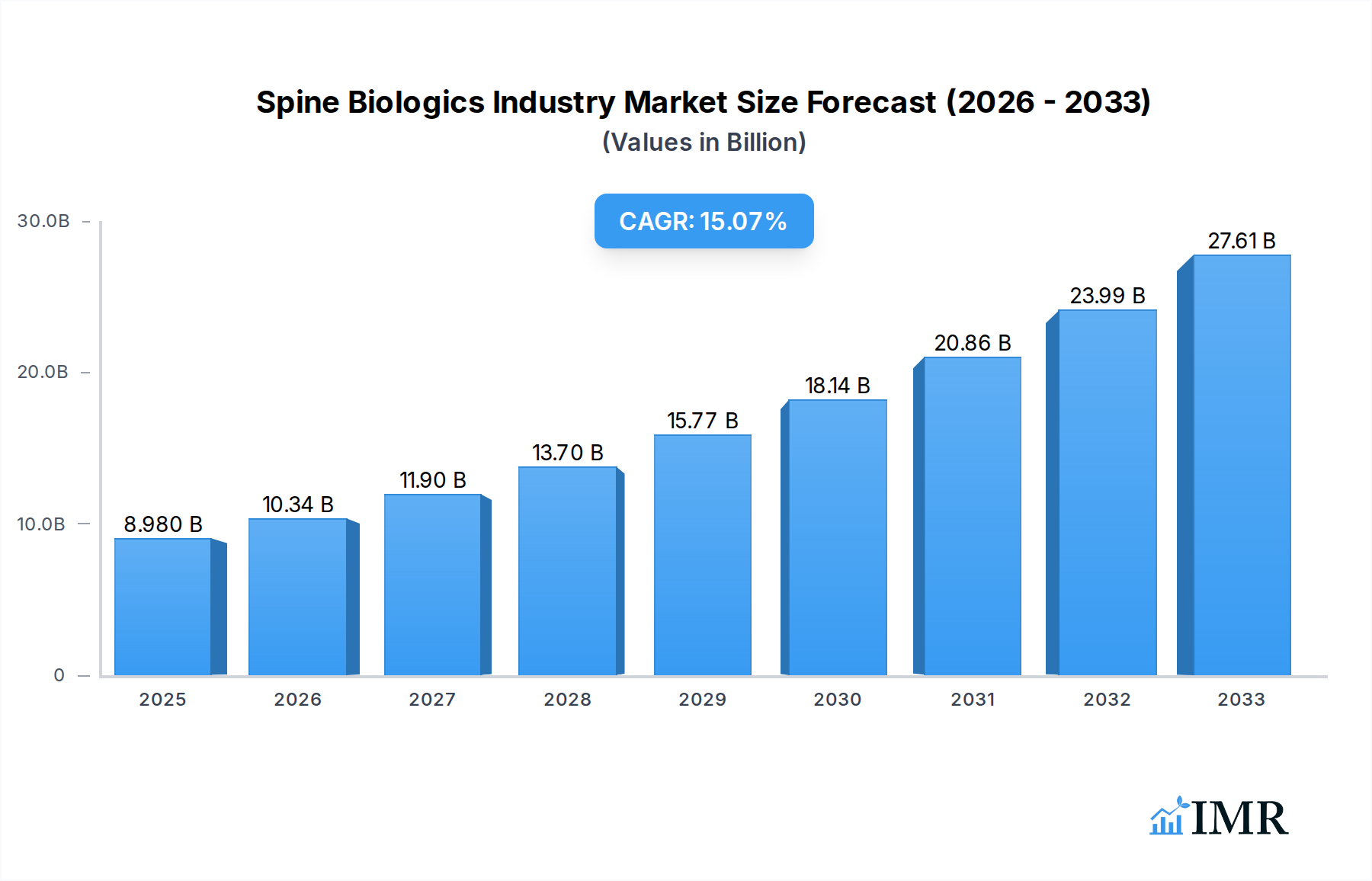
Spine Biologics Industry Market Size (In Billion)

The market's segmentation reveals a dynamic landscape. Within product segments, Bone Graft Substitutes, encompassing Bone Morphogenetic Proteins and Synthetic Bone Grafts, are expected to witness substantial demand due to their enhanced fusion rates and reduced donor site morbidity. Spinal Allografts, including Machined Bone Allograft and Demineralized Bone Matrix, also hold a significant market share, leveraging their proven efficacy in spinal fusion procedures. The 'Others' segment, likely encompassing tissue-engineered products and novel regenerative therapies, is anticipated to exhibit strong growth as innovation continues. End-user analysis indicates that Hospitals will remain the largest segment, owing to the complexity of spinal surgeries performed within these institutions and the availability of advanced surgical equipment. Ambulatory Surgical Centers are also expected to see considerable growth as minimally invasive spine procedures become more prevalent, allowing for outpatient treatments. Geographically, North America, particularly the United States, is anticipated to lead the market due to high healthcare expenditure, advanced technological adoption, and a high prevalence of spinal disorders. Europe and the Asia Pacific region are also poised for substantial growth, driven by an increasing patient pool, improving healthcare access, and growing investments in orthopedic research.
Spine Biologics Industry Company Market Share

This in-depth report provides a comprehensive analysis of the global Spine Biologics Market, exploring its dynamics, growth trajectories, and future potential. Covering the study period from 2019 to 2033, with a base year of 2025 and a forecast period of 2025–2033, this report is an essential resource for stakeholders seeking to understand the intricate workings and expansion avenues within the bone graft substitutes, spinal allografts, and other segments of the spine biologics landscape. The market size is projected to reach $XX billion in 2025, exhibiting a robust CAGR of XX% throughout the forecast period.
Spine Biologics Industry Market Dynamics & Structure
The Spine Biologics Industry is characterized by a moderately concentrated market structure, with key players like Medtronic plc, Stryker Corporation, Zimmer Biomet, Johnson And Johnson (Depuy Synthes), NuVasive Inc., Arthrex Inc., Orthofix Medical Inc., Exactech Inc., Spine Wave Inc., and others holding significant market shares. Technological innovation remains a primary driver, fueled by advancements in bone morphogenetic proteins (BMPs) and synthetic bone grafts that offer superior efficacy and reduced donor site morbidity. The regulatory framework, overseen by bodies like the FDA, plays a crucial role in market entry and product approval, sometimes acting as a barrier to rapid innovation. Competitive product substitutes, including advanced fusion techniques and non-biological implants, also influence market dynamics. End-user demographics, primarily hospitals and ambulatory surgical centers, are increasingly demanding cost-effective and minimally invasive solutions. Mergers and acquisitions (M&A) are a notable trend, with companies acquiring innovative technologies and expanding their product portfolios to consolidate market position. For instance, the historical period saw XX M&A deals impacting the market landscape.
- Market Concentration: Moderate, with a few dominant players.
- Key Innovation Drivers: BMPs, synthetic bone graft advancements, minimally invasive techniques.
- Regulatory Impact: Crucial for product approval and market access; can create barriers.
- Competitive Landscape: Driven by product efficacy, cost-effectiveness, and technological differentiation.
- End-User Focus: Hospitals and ASCs demanding advanced, less invasive solutions.
- M&A Activity: Strategic acquisitions to gain market share and technological expertise.
Spine Biologics Industry Growth Trends & Insights
The Spine Biologics Industry is poised for significant expansion, driven by a confluence of factors that are reshaping patient care and surgical practices. The escalating prevalence of spinal disorders, including degenerative disc disease, herniated discs, and spinal deformities, directly fuels demand for effective spine fusion procedures. This demographic shift, coupled with an aging global population, creates a sustained need for advanced bone graft substitutes and spinal allografts. Furthermore, technological disruptions are continuously enhancing product efficacy and patient outcomes. The development of novel bone morphogenetic proteins (BMPs), engineered for targeted osteoinduction, and the refinement of synthetic bone grafts with improved biocompatibility and bioresorbability, are revolutionizing treatment options.
The adoption rates for these advanced biologics are steadily increasing as surgeons gain confidence in their performance and as healthcare reimbursement models become more supportive. This trend is particularly evident in the hospitals and ambulatory surgical centers segments, which are investing in these cutting-edge technologies to offer superior patient care and reduce revision surgery rates. Consumer behavior shifts are also playing a role, with patients becoming more informed and actively seeking out less invasive and more effective treatment modalities. The drive towards minimally invasive spine surgery (MISS) further accelerates the demand for sophisticated biologics that facilitate fusion in challenging anatomical locations and with reduced trauma.
The market penetration of biologics in spine surgery is projected to rise from approximately XX% in 2024 to an estimated XX% by 2033. This growth trajectory is underpinned by a compound annual growth rate (CAGR) estimated at XX% for the forecast period. The industry is witnessing a paradigm shift from traditional autograft procedures to the wider acceptance of allografts and synthetic alternatives, driven by the desire to minimize donor site morbidity and improve surgical efficiency. Continuous research and development are unlocking new applications and enhancing the regenerative potential of these biological materials, promising a future where spinal fusion is more predictable, efficient, and less burdensome for patients.
Dominant Regions, Countries, or Segments in Spine Biologics Industry
The global Spine Biologics Industry is experiencing robust growth, with North America currently leading the market, closely followed by Europe. The United States, in particular, stands out as a dominant country, driven by a combination of high healthcare expenditure, a large aging population susceptible to spinal conditions, and the early adoption of advanced medical technologies. The strong presence of leading spine companies, coupled with significant investment in research and development, further solidifies its leading position. The region's advanced healthcare infrastructure, including a high density of hospitals and ambulatory surgical centers, ensures widespread access to innovative spine biologics and surgical procedures.
Within the Product segment, Bone Graft Substitutes are currently the largest and fastest-growing category, with Bone Morphogenetic Proteins (BMPs) and Synthetic Bone Grafts leading the sub-segments. The demand for BMPs is propelled by their proven osteoinductive properties, enabling faster and more reliable bone fusion. Simultaneously, the continuous innovation in synthetic bone grafts, offering customizable properties and enhanced biocompatibility, is capturing a significant market share. Spinal Allografts, comprising Machined Bones Allograft and Demineralized Bone Matrix (DBM), also hold a substantial market share, benefiting from their established track record and cost-effectiveness. However, concerns regarding potential disease transmission, albeit low, are being addressed by stringent processing and screening protocols.
The End User segment is dominated by Hospitals, which perform the majority of complex spinal surgeries requiring biologics. However, Ambulatory Surgical Centers (ASCs) are emerging as a significant growth driver, as they increasingly offer specialized spinal procedures, particularly for less complex cases, benefiting from their cost efficiencies and patient convenience. This trend is fostering a more decentralized approach to spinal care and expanding the market reach for spine biologics.
- Leading Region: North America, driven by the United States.
- Dominant Country: United States, due to high healthcare spending and technological adoption.
- Key Product Segments:
- Bone Graft Substitutes: Leading growth, with BMPs and Synthetic Bone Grafts at the forefront.
- Spinal Allografts: Significant market share, with DBM and Machined Bone Allografts being prominent.
- Primary End Users: Hospitals remain dominant, with Ambulatory Surgical Centers showing strong growth potential.
- Growth Drivers: Aging population, rising prevalence of spinal disorders, technological advancements, favorable reimbursement policies, and the shift towards minimally invasive surgery.
Spine Biologics Industry Product Landscape
The Spine Biologics Industry product landscape is defined by continuous innovation aimed at enhancing osteoconduction, osteoinduction, and osteogenesis. Bone graft substitutes, particularly Bone Morphogenetic Proteins (BMPs) and advanced Synthetic Bone Grafts, are at the forefront, offering predictable fusion rates and reduced donor site morbidity compared to autografts. BMPs, such as recombinant human bone morphogenetic protein-2 (rhBMP-2), stimulate new bone formation, while synthetic materials, including calcium phosphates and bioactive glasses, provide biocompatible scaffolds with tailored resorption rates. Spinal Allografts, including Demineralized Bone Matrix (DBM) and Machined Bones Allograft, remain crucial, leveraging the body's natural bone-healing processes. Innovations focus on improving processing techniques to enhance cellular viability and antigenicity reduction. The unique selling proposition lies in the ability of these products to accelerate fusion, improve surgical outcomes, and minimize patient discomfort, paving the way for more efficient and effective spinal fusion procedures.
Key Drivers, Barriers & Challenges in Spine Biologics Industry
The Spine Biologics Industry is propelled by several key drivers. Technological advancements in bone morphogenetic proteins (BMPs) and synthetic bone grafts are improving efficacy and patient outcomes. The increasing global prevalence of spinal disorders, fueled by an aging population and sedentary lifestyles, creates a sustained demand for effective treatment solutions. Furthermore, the shift towards minimally invasive spine surgery (MISS) favors biologics that facilitate fusion with reduced trauma. Economic factors, such as growing healthcare expenditure and improving reimbursement policies for advanced biologics, also contribute to market growth.
However, the industry faces significant barriers and challenges. The high cost of innovative biologics, particularly BMPs, can limit their adoption, especially in cost-sensitive healthcare systems. Stringent regulatory hurdles for product approval, requiring extensive clinical trials, can prolong time-to-market and increase development costs. Supply chain complexities and the risk of donor-site morbidity associated with allografts, though diminishing with advanced processing, can also pose challenges. Intense competitive pressure from established players and emerging technologies, including non-biological implants, necessitates continuous innovation and cost management.
Emerging Opportunities in Spine Biologics Industry
Emerging opportunities in the Spine Biologics Industry lie in several key areas. The development of novel biomaterials with enhanced regenerative capabilities, such as those incorporating growth factors or stem cells, presents a significant avenue for innovation. Expansion into emerging markets with growing healthcare infrastructure and increasing patient awareness of advanced treatment options offers substantial untapped potential. Furthermore, the application of 3D printing technology in creating patient-specific bone graft substitutes tailored for complex spinal deformities represents a groundbreaking frontier. Evolving consumer preferences for less invasive and faster-healing procedures will continue to drive demand for advanced biologic solutions.
Growth Accelerators in the Spine Biologics Industry Industry
The Spine Biologics Industry is set to accelerate its growth through several catalysts. Continued technological breakthroughs in developing more potent and targeted osteoinductive agents, alongside the refinement of synthetic bone grafts with improved bioactivity, will be pivotal. Strategic partnerships between biologic manufacturers, implant companies, and research institutions will foster collaboration and accelerate product development and market penetration. Furthermore, market expansion strategies focused on underserved regions and the increasing adoption of biologics in ambulatory surgical centers will broaden the market reach and drive volume growth. The growing evidence base from clinical studies demonstrating the long-term efficacy and safety of spine biologics will further bolster surgeon confidence and patient acceptance.
Key Players Shaping the Spine Biologics Industry Market
- Arthrex Inc.
- Exactech Inc.
- Spine Wave Inc.
- Johnson And Johnson (Depuy Synthes)
- Orthofix Medical Inc.
- NuVasive Inc.
- Stryker Corporation
- Zimmer Biomet
- Medtronic plc
Notable Milestones in Spine Biologics Industry Sector
- 2019: Launch of new synthetic bone graft formulations offering enhanced handling and fusion properties.
- 2020: FDA approval for a novel bone morphogenetic protein (BMP) delivery system for spinal fusion.
- 2021: Strategic acquisition of a key player in the demineralized bone matrix (DBM) market to expand product portfolio.
- 2022: Significant increase in clinical trial publications demonstrating superior fusion rates with advanced biologics.
- 2023: Introduction of bioresorbable synthetic scaffolds designed for osteopromotion and controlled drug delivery.
- 2024: Growing trend of biologics integration into minimally invasive spine surgery (MISS) techniques.
In-Depth Spine Biologics Industry Market Outlook
The Spine Biologics Industry is projected for sustained and robust growth, driven by a confluence of factors including an aging global population, the increasing incidence of spinal pathologies, and continuous technological innovation. The market outlook is highly positive, with a strong emphasis on regenerative medicine and the development of advanced biomaterials that mimic natural bone-healing processes. Key growth accelerators include the ongoing refinement of bone morphogenetic proteins (BMPs) and synthetic bone grafts, offering enhanced osteoinductivity and osteoconductivity. The expanding application of these biologics in minimally invasive spine surgeries further amplifies their market potential. Strategic collaborations and the increasing focus on patient-centric solutions will continue to shape the competitive landscape, ensuring a dynamic and expanding future for the spine biologics industry.
Spine Biologics Industry Segmentation
-
1. Product
-
1.1. Bone Graft Substitutes
- 1.1.1. Bone Morphogenetic Proteins
- 1.1.2. Synthetic Bone Grafts
-
1.2. Spinal Allografts
- 1.2.1. Machined Bones Allograft
- 1.2.2. Demineralized Bone Matrix
- 1.3. Others
-
1.1. Bone Graft Substitutes
-
2. End User
- 2.1. Hospitals
- 2.2. Ambulatory Surgical Centers
- 2.3. Others
Spine Biologics Industry Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. Europe
- 2.1. Germany
- 2.2. United Kingdom
- 2.3. France
- 2.4. Italy
- 2.5. Spain
- 2.6. Rest of Europe
-
3. Asia Pacific
- 3.1. China
- 3.2. Japan
- 3.3. India
- 3.4. Australia
- 3.5. South Korea
- 3.6. Rest of Asia Pacific
-
4. Middle East and Africa
- 4.1. GCC
- 4.2. South Africa
- 4.3. Rest of Middle East and Africa
-
5. South America
- 5.1. Brazil
- 5.2. Argentina
- 5.3. Rest of South America
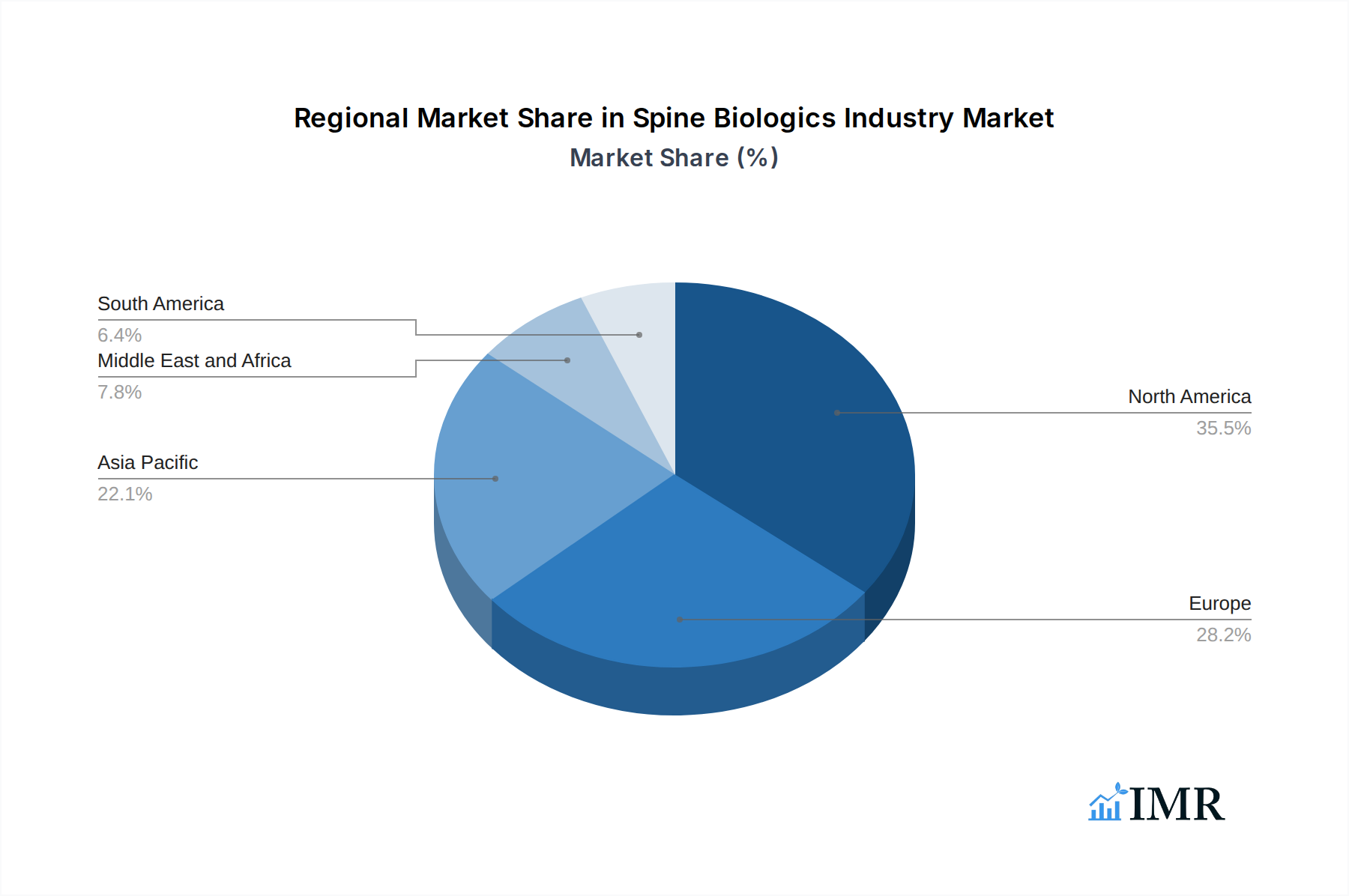
Spine Biologics Industry Regional Market Share

Geographic Coverage of Spine Biologics Industry
Spine Biologics Industry REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 15.24% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Objective
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Market Snapshot
- 3. Market Dynamics
- 3.1. Market Drivers
- 3.2. Market Restrains
- 3.3. Market Trends
- 3.4. Market Opportunities
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.1.1. Bargaining Power of Suppliers
- 4.1.2. Bargaining Power of Buyers
- 4.1.3. Threat of New Entrants
- 4.1.4. Threat of Substitutes
- 4.1.5. Competitive Rivalry
- 4.2. PESTEL analysis
- 4.3. BCG Analysis
- 4.3.1. Stars (High Growth, High Market Share)
- 4.3.2. Cash Cows (Low Growth, High Market Share)
- 4.3.3. Question Mark (High Growth, Low Market Share)
- 4.3.4. Dogs (Low Growth, Low Market Share)
- 4.4. Ansoff Matrix Analysis
- 4.5. Supply Chain Analysis
- 4.6. Regulatory Landscape
- 4.7. Current Market Potential and Opportunity Assessment (TAM–SAM–SOM Framework)
- 4.8. IMR Analyst Note
- 4.1. Porters Five Forces
- 5. Market Analysis, Insights and Forecast 2021-2033
- 5.1. Market Analysis, Insights and Forecast - by Product
- 5.1.1. Bone Graft Substitutes
- 5.1.1.1. Bone Morphogenetic Proteins
- 5.1.1.2. Synthetic Bone Grafts
- 5.1.2. Spinal Allografts
- 5.1.2.1. Machined Bones Allograft
- 5.1.2.2. Demineralized Bone Matrix
- 5.1.3. Others
- 5.1.1. Bone Graft Substitutes
- 5.2. Market Analysis, Insights and Forecast - by End User
- 5.2.1. Hospitals
- 5.2.2. Ambulatory Surgical Centers
- 5.2.3. Others
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. Europe
- 5.3.3. Asia Pacific
- 5.3.4. Middle East and Africa
- 5.3.5. South America
- 5.1. Market Analysis, Insights and Forecast - by Product
- 6. Global Spine Biologics Industry Analysis, Insights and Forecast, 2021-2033
- 6.1. Market Analysis, Insights and Forecast - by Product
- 6.1.1. Bone Graft Substitutes
- 6.1.1.1. Bone Morphogenetic Proteins
- 6.1.1.2. Synthetic Bone Grafts
- 6.1.2. Spinal Allografts
- 6.1.2.1. Machined Bones Allograft
- 6.1.2.2. Demineralized Bone Matrix
- 6.1.3. Others
- 6.1.1. Bone Graft Substitutes
- 6.2. Market Analysis, Insights and Forecast - by End User
- 6.2.1. Hospitals
- 6.2.2. Ambulatory Surgical Centers
- 6.2.3. Others
- 6.1. Market Analysis, Insights and Forecast - by Product
- 7. North America Spine Biologics Industry Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Product
- 7.1.1. Bone Graft Substitutes
- 7.1.1.1. Bone Morphogenetic Proteins
- 7.1.1.2. Synthetic Bone Grafts
- 7.1.2. Spinal Allografts
- 7.1.2.1. Machined Bones Allograft
- 7.1.2.2. Demineralized Bone Matrix
- 7.1.3. Others
- 7.1.1. Bone Graft Substitutes
- 7.2. Market Analysis, Insights and Forecast - by End User
- 7.2.1. Hospitals
- 7.2.2. Ambulatory Surgical Centers
- 7.2.3. Others
- 7.1. Market Analysis, Insights and Forecast - by Product
- 8. Europe Spine Biologics Industry Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Product
- 8.1.1. Bone Graft Substitutes
- 8.1.1.1. Bone Morphogenetic Proteins
- 8.1.1.2. Synthetic Bone Grafts
- 8.1.2. Spinal Allografts
- 8.1.2.1. Machined Bones Allograft
- 8.1.2.2. Demineralized Bone Matrix
- 8.1.3. Others
- 8.1.1. Bone Graft Substitutes
- 8.2. Market Analysis, Insights and Forecast - by End User
- 8.2.1. Hospitals
- 8.2.2. Ambulatory Surgical Centers
- 8.2.3. Others
- 8.1. Market Analysis, Insights and Forecast - by Product
- 9. Asia Pacific Spine Biologics Industry Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Product
- 9.1.1. Bone Graft Substitutes
- 9.1.1.1. Bone Morphogenetic Proteins
- 9.1.1.2. Synthetic Bone Grafts
- 9.1.2. Spinal Allografts
- 9.1.2.1. Machined Bones Allograft
- 9.1.2.2. Demineralized Bone Matrix
- 9.1.3. Others
- 9.1.1. Bone Graft Substitutes
- 9.2. Market Analysis, Insights and Forecast - by End User
- 9.2.1. Hospitals
- 9.2.2. Ambulatory Surgical Centers
- 9.2.3. Others
- 9.1. Market Analysis, Insights and Forecast - by Product
- 10. Middle East and Africa Spine Biologics Industry Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Product
- 10.1.1. Bone Graft Substitutes
- 10.1.1.1. Bone Morphogenetic Proteins
- 10.1.1.2. Synthetic Bone Grafts
- 10.1.2. Spinal Allografts
- 10.1.2.1. Machined Bones Allograft
- 10.1.2.2. Demineralized Bone Matrix
- 10.1.3. Others
- 10.1.1. Bone Graft Substitutes
- 10.2. Market Analysis, Insights and Forecast - by End User
- 10.2.1. Hospitals
- 10.2.2. Ambulatory Surgical Centers
- 10.2.3. Others
- 10.1. Market Analysis, Insights and Forecast - by Product
- 11. South America Spine Biologics Industry Analysis, Insights and Forecast, 2020-2032
- 11.1. Market Analysis, Insights and Forecast - by Product
- 11.1.1. Bone Graft Substitutes
- 11.1.1.1. Bone Morphogenetic Proteins
- 11.1.1.2. Synthetic Bone Grafts
- 11.1.2. Spinal Allografts
- 11.1.2.1. Machined Bones Allograft
- 11.1.2.2. Demineralized Bone Matrix
- 11.1.3. Others
- 11.1.1. Bone Graft Substitutes
- 11.2. Market Analysis, Insights and Forecast - by End User
- 11.2.1. Hospitals
- 11.2.2. Ambulatory Surgical Centers
- 11.2.3. Others
- 11.1. Market Analysis, Insights and Forecast - by Product
- 12. Competitive Analysis
- 12.1. Company Profiles
- 12.1.1 Arthrex Inc
- 12.1.1.1. Company Overview
- 12.1.1.2. Products
- 12.1.1.3. Company Financials
- 12.1.1.4. SWOT Analysis
- 12.1.2 Exactech Inc
- 12.1.2.1. Company Overview
- 12.1.2.2. Products
- 12.1.2.3. Company Financials
- 12.1.2.4. SWOT Analysis
- 12.1.3 Spine Wave Inc
- 12.1.3.1. Company Overview
- 12.1.3.2. Products
- 12.1.3.3. Company Financials
- 12.1.3.4. SWOT Analysis
- 12.1.4 Johnson And Johnson (Depuy Synthes)
- 12.1.4.1. Company Overview
- 12.1.4.2. Products
- 12.1.4.3. Company Financials
- 12.1.4.4. SWOT Analysis
- 12.1.5 Orthofix Medical Inc
- 12.1.5.1. Company Overview
- 12.1.5.2. Products
- 12.1.5.3. Company Financials
- 12.1.5.4. SWOT Analysis
- 12.1.6 NuVasive Inc
- 12.1.6.1. Company Overview
- 12.1.6.2. Products
- 12.1.6.3. Company Financials
- 12.1.6.4. SWOT Analysis
- 12.1.7 Stryker Corporation
- 12.1.7.1. Company Overview
- 12.1.7.2. Products
- 12.1.7.3. Company Financials
- 12.1.7.4. SWOT Analysis
- 12.1.8 Zimmer Biomet
- 12.1.8.1. Company Overview
- 12.1.8.2. Products
- 12.1.8.3. Company Financials
- 12.1.8.4. SWOT Analysis
- 12.1.9 Medtronic plc
- 12.1.9.1. Company Overview
- 12.1.9.2. Products
- 12.1.9.3. Company Financials
- 12.1.9.4. SWOT Analysis
- 12.1.1 Arthrex Inc
- 12.2. Market Entropy
- 12.2.1 Company's Key Areas Served
- 12.2.2 Recent Developments
- 12.3. Company Market Share Analysis 2025
- 12.3.1 Top 5 Companies Market Share Analysis
- 12.3.2 Top 3 Companies Market Share Analysis
- 12.4. List of Potential Customers
- 13. Research Methodology
List of Figures
- Figure 1: Global Spine Biologics Industry Revenue Breakdown (billion, %) by Region 2025 & 2033
- Figure 2: North America Spine Biologics Industry Revenue (billion), by Product 2025 & 2033
- Figure 3: North America Spine Biologics Industry Revenue Share (%), by Product 2025 & 2033
- Figure 4: North America Spine Biologics Industry Revenue (billion), by End User 2025 & 2033
- Figure 5: North America Spine Biologics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 6: North America Spine Biologics Industry Revenue (billion), by Country 2025 & 2033
- Figure 7: North America Spine Biologics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 8: Europe Spine Biologics Industry Revenue (billion), by Product 2025 & 2033
- Figure 9: Europe Spine Biologics Industry Revenue Share (%), by Product 2025 & 2033
- Figure 10: Europe Spine Biologics Industry Revenue (billion), by End User 2025 & 2033
- Figure 11: Europe Spine Biologics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 12: Europe Spine Biologics Industry Revenue (billion), by Country 2025 & 2033
- Figure 13: Europe Spine Biologics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 14: Asia Pacific Spine Biologics Industry Revenue (billion), by Product 2025 & 2033
- Figure 15: Asia Pacific Spine Biologics Industry Revenue Share (%), by Product 2025 & 2033
- Figure 16: Asia Pacific Spine Biologics Industry Revenue (billion), by End User 2025 & 2033
- Figure 17: Asia Pacific Spine Biologics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 18: Asia Pacific Spine Biologics Industry Revenue (billion), by Country 2025 & 2033
- Figure 19: Asia Pacific Spine Biologics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East and Africa Spine Biologics Industry Revenue (billion), by Product 2025 & 2033
- Figure 21: Middle East and Africa Spine Biologics Industry Revenue Share (%), by Product 2025 & 2033
- Figure 22: Middle East and Africa Spine Biologics Industry Revenue (billion), by End User 2025 & 2033
- Figure 23: Middle East and Africa Spine Biologics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 24: Middle East and Africa Spine Biologics Industry Revenue (billion), by Country 2025 & 2033
- Figure 25: Middle East and Africa Spine Biologics Industry Revenue Share (%), by Country 2025 & 2033
- Figure 26: South America Spine Biologics Industry Revenue (billion), by Product 2025 & 2033
- Figure 27: South America Spine Biologics Industry Revenue Share (%), by Product 2025 & 2033
- Figure 28: South America Spine Biologics Industry Revenue (billion), by End User 2025 & 2033
- Figure 29: South America Spine Biologics Industry Revenue Share (%), by End User 2025 & 2033
- Figure 30: South America Spine Biologics Industry Revenue (billion), by Country 2025 & 2033
- Figure 31: South America Spine Biologics Industry Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Spine Biologics Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 2: Global Spine Biologics Industry Revenue billion Forecast, by End User 2020 & 2033
- Table 3: Global Spine Biologics Industry Revenue billion Forecast, by Region 2020 & 2033
- Table 4: Global Spine Biologics Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 5: Global Spine Biologics Industry Revenue billion Forecast, by End User 2020 & 2033
- Table 6: Global Spine Biologics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 7: United States Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 8: Canada Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 9: Mexico Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 10: Global Spine Biologics Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 11: Global Spine Biologics Industry Revenue billion Forecast, by End User 2020 & 2033
- Table 12: Global Spine Biologics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 13: Germany Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 14: United Kingdom Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 15: France Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 16: Italy Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 17: Spain Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 18: Rest of Europe Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 19: Global Spine Biologics Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 20: Global Spine Biologics Industry Revenue billion Forecast, by End User 2020 & 2033
- Table 21: Global Spine Biologics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 22: China Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 23: Japan Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 24: India Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 25: Australia Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 26: South Korea Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 27: Rest of Asia Pacific Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 28: Global Spine Biologics Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 29: Global Spine Biologics Industry Revenue billion Forecast, by End User 2020 & 2033
- Table 30: Global Spine Biologics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 31: GCC Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 32: South Africa Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 33: Rest of Middle East and Africa Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 34: Global Spine Biologics Industry Revenue billion Forecast, by Product 2020 & 2033
- Table 35: Global Spine Biologics Industry Revenue billion Forecast, by End User 2020 & 2033
- Table 36: Global Spine Biologics Industry Revenue billion Forecast, by Country 2020 & 2033
- Table 37: Brazil Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 38: Argentina Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
- Table 39: Rest of South America Spine Biologics Industry Revenue (billion) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Spine Biologics Industry?
The projected CAGR is approximately 15.24%.
2. Which companies are prominent players in the Spine Biologics Industry?
Key companies in the market include Arthrex Inc, Exactech Inc, Spine Wave Inc, Johnson And Johnson (Depuy Synthes), Orthofix Medical Inc, NuVasive Inc , Stryker Corporation, Zimmer Biomet, Medtronic plc.
3. What are the main segments of the Spine Biologics Industry?
The market segments include Product, End User.
4. Can you provide details about the market size?
The market size is estimated to be USD 8.98 billion as of 2022.
5. What are some drivers contributing to market growth?
; Growing Geriatric Population; Growing Prevalence of Spine Deformities; Technological Advancements.
6. What are the notable trends driving market growth?
Spinal Allografts in Spinal Biologics is Estimated to Witness a Healthy Growth in Future.
7. Are there any restraints impacting market growth?
; Reimbursement Policies.
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4750, USD 5250, and USD 8750 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Spine Biologics Industry," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Spine Biologics Industry report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Spine Biologics Industry?
To stay informed about further developments, trends, and reports in the Spine Biologics Industry, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
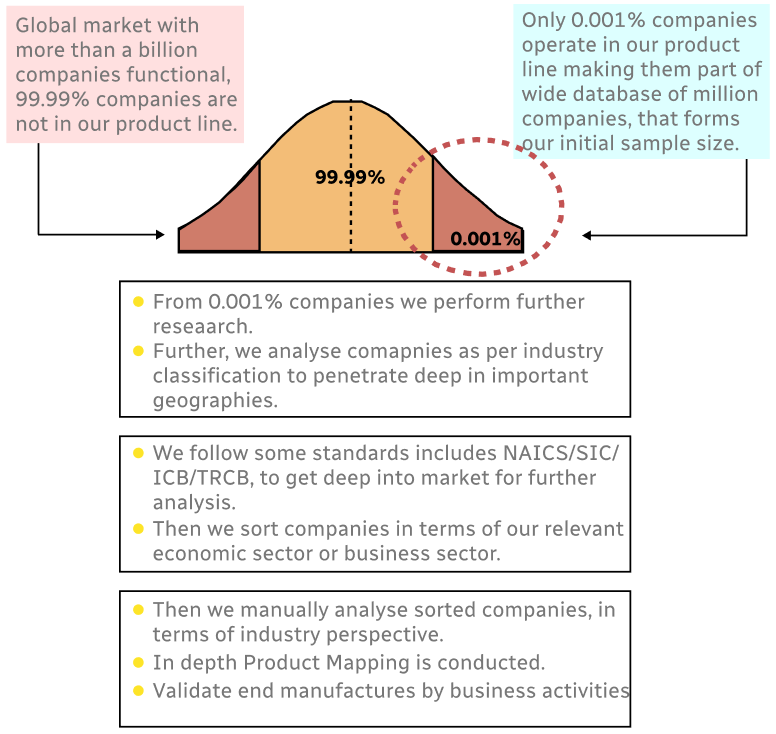
Step 1 - Identification of Relevant Samples Size from Population Database
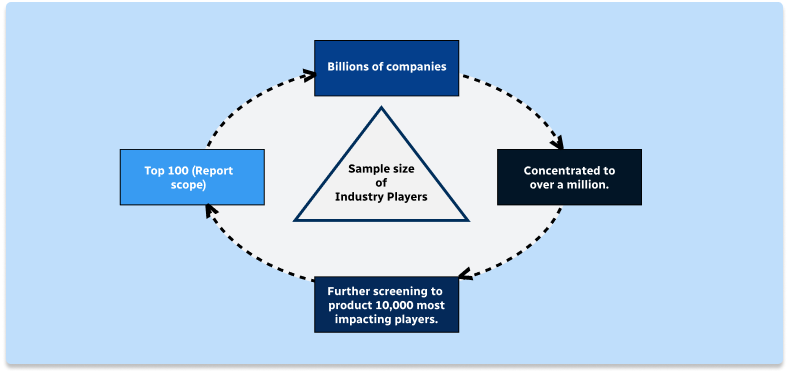
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
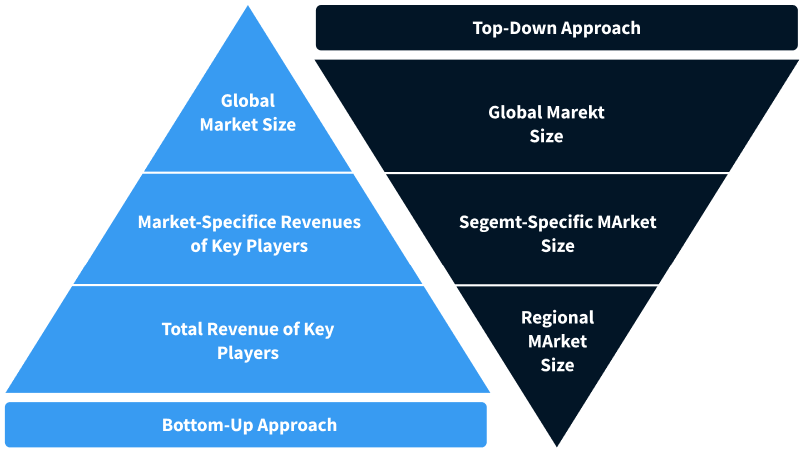
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


