Key Insights
The Gene Detection for Individualized Cancer Therapy market is poised for remarkable expansion, projected to reach $11.34 billion by 2025, driven by a robust CAGR of 24.01% through 2033. This significant growth is primarily fueled by the increasing prevalence of cancer globally, coupled with a growing understanding of the critical role genetic mutations play in disease development and progression. Personalized medicine, which leverages gene detection to tailor treatment strategies for individual patients, is becoming the cornerstone of effective oncology care. Advances in high-throughput sequencing technologies, improved bioinformatics tools, and a greater emphasis on preventative healthcare are further accelerating market adoption. The rising demand for early cancer detection and risk assessment, particularly through genetic screening and reproductive genetic testing, is also a key catalyst, empowering individuals to make informed decisions about their health and proactive interventions.
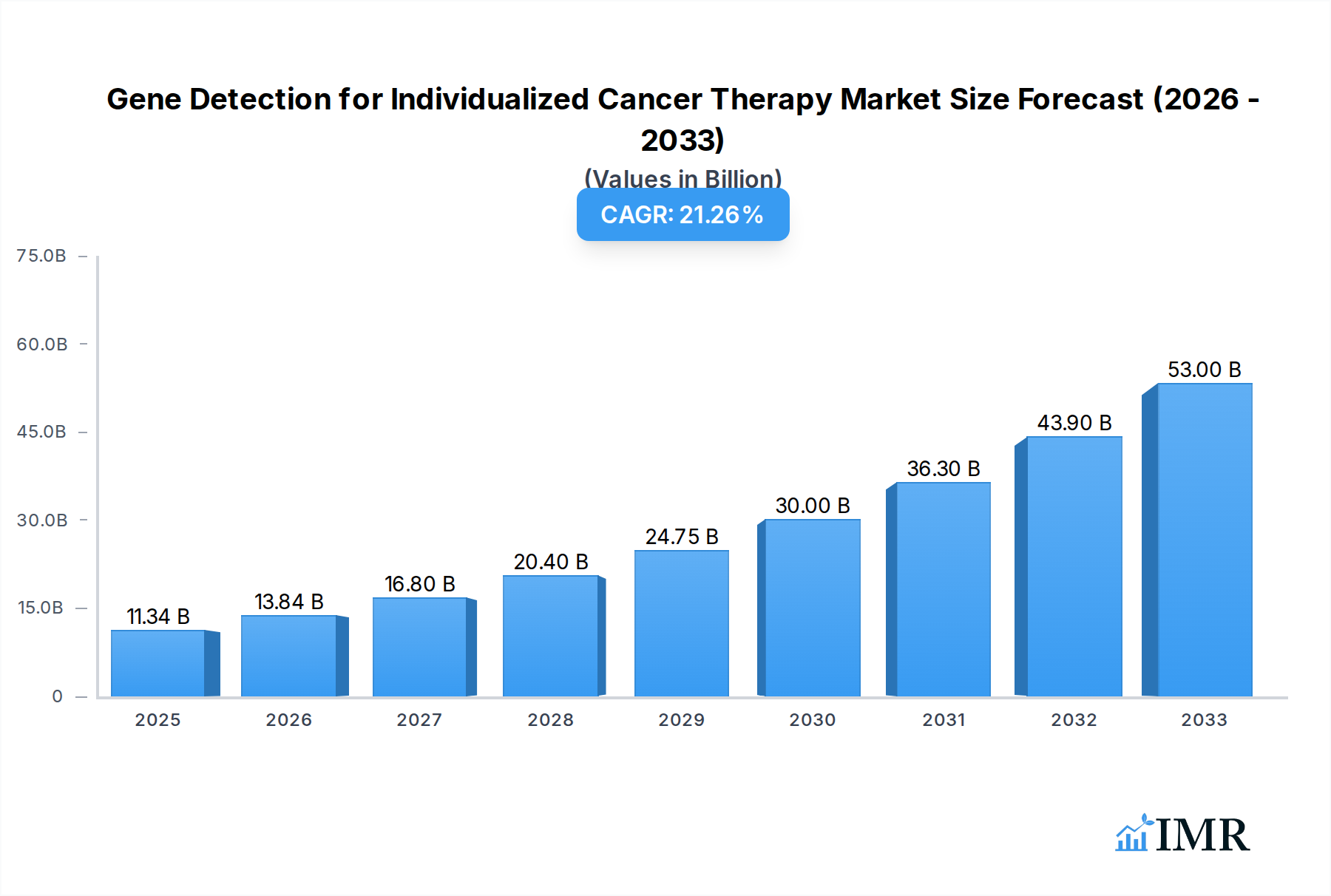
Gene Detection for Individualized Cancer Therapy Market Size (In Billion)

The market's dynamism is further shaped by several emerging trends, including the integration of liquid biopsy techniques, which offer a less invasive method for cancer detection and monitoring. Furthermore, the expanding applications of gene detection in diagnostic tests, gene carrier testing, and predictive testing before symptom onset underscore its versatility. Key market players are actively investing in research and development to enhance the accuracy and accessibility of these tests, fostering collaborations with healthcare providers and diagnostic centers. While the market demonstrates immense potential, potential restraints such as the high cost of advanced genetic testing technologies and the need for stringent regulatory frameworks to ensure data privacy and ethical use of genetic information warrant careful consideration. Nevertheless, the overarching shift towards precision oncology, supported by technological innovation and increasing patient awareness, ensures a promising trajectory for the Gene Detection for Individualized Cancer Therapy market.
Gene Detection for Individualized Cancer Therapy Company Market Share

This comprehensive report delves into the dynamic Gene Detection for Individualized Cancer Therapy Market, providing an in-depth analysis of its evolution, growth trajectory, and future potential. Targeted towards oncology professionals, researchers, pharmaceutical companies, and investment firms, this report offers actionable insights into a rapidly expanding sector critical for precision medicine. We explore the intricate market structure, key growth drivers, dominant regional landscapes, and the innovative product developments shaping the future of cancer treatment. The report covers the Parent Market of Genetic Testing and Diagnostics, with a particular focus on the Child Market of Oncology Gene Profiling.
Gene Detection for Individualized Cancer Therapy Market Dynamics & Structure
The Gene Detection for Individualized Cancer Therapy Market is characterized by a moderately concentrated structure, with key players investing heavily in research and development to gain a competitive edge. Technological innovation is a primary driver, fueled by advancements in next-generation sequencing (NGS) technologies and the increasing understanding of cancer genomics. Regulatory frameworks, while evolving, are crucial for market entry and product approval, ensuring diagnostic accuracy and clinical utility. Competitive product substitutes include traditional diagnostic methods and broad-spectrum cancer treatments, but the superior specificity of gene detection for targeted therapies is a significant differentiator. End-user demographics are shifting towards a greater demand for personalized treatment options from healthcare providers and a growing awareness among patient advocacy groups. Mergers and acquisitions (M&A) are a prominent trend as larger companies seek to integrate specialized gene detection capabilities and expand their oncology portfolios.
- Market Concentration: Moderate, with key players dominating a significant portion of the market.
- Technological Innovation Drivers: Advancements in NGS, liquid biopsy, CRISPR-based diagnostics, and AI-powered data analysis.
- Regulatory Frameworks: FDA, EMA, and national regulatory bodies are crucial for market approval and standardization.
- Competitive Product Substitutes: Broad-spectrum chemotherapy, immunotherapy without genetic profiling.
- End-User Demographics: Oncologists, pathologists, hospital systems, diagnostic laboratories, and increasingly, patients seeking tailored treatments.
- M&A Trends: Strategic acquisitions and partnerships to consolidate market share and acquire innovative technologies. The Parent Market of Genetic Testing and Diagnostics is projected to reach $65.2 billion by 2025, with the Child Market of Oncology Gene Profiling representing a substantial and rapidly growing segment.
Gene Detection for Individualized Cancer Therapy Growth Trends & Insights
The Gene Detection for Individualized Cancer Therapy Market is poised for robust expansion, driven by an escalating demand for precision medicine and a deeper understanding of tumor biology. The market size evolution is characterized by a consistent upward trajectory, with significant growth anticipated over the forecast period. Adoption rates for gene detection technologies are accelerating as clinical evidence supporting their efficacy in guiding treatment decisions grows. Technological disruptions, such as the refinement of liquid biopsy techniques and the integration of artificial intelligence for complex genomic data interpretation, are revolutionizing the diagnostic landscape. Consumer behavior shifts are also playing a vital role, with an increasing number of patients actively seeking personalized treatment plans informed by their genetic makeup. The projected Compound Annual Growth Rate (CAGR) for the Gene Detection for Individualized Cancer Therapy Market is expected to be approximately 18.5% from 2025 to 2033. Market penetration is currently at XX%, with significant room for expansion in both developed and emerging economies. The shift from reactive to proactive cancer management, facilitated by early detection through genetic screening and testing before symptoms appear, is a key trend.
The global market for Gene Detection for Individualized Cancer Therapy is estimated at $18.5 billion in 2025, with projections indicating a substantial increase to $60.1 billion by 2033. This growth is underpinned by several critical factors. Firstly, the increasing global incidence of cancer, coupled with an aging population, necessitates more effective and targeted therapeutic strategies. Gene detection provides the molecular blueprint for identifying specific driver mutations and biomarkers, allowing oncologists to select the most appropriate treatments, thereby improving patient outcomes and survival rates. The declining cost of Next-Generation Sequencing (NGS) technologies, once a significant barrier to widespread adoption, has made comprehensive genomic profiling more accessible to a broader range of healthcare institutions and patients. This cost reduction, combined with improvements in analytical accuracy and turnaround times, is driving higher adoption rates, particularly in Diagnostic Centers and Hospitals.
Furthermore, the growing emphasis on Testing Before Symptoms Appear and Genetic Screening for hereditary cancer predispositions is opening up new avenues for early intervention and preventive measures. This proactive approach not only enhances patient prognosis but also reduces the long-term healthcare burden associated with advanced-stage cancers. The integration of gene detection into routine clinical practice is also being bolstered by supportive reimbursement policies in several key markets, further accelerating market penetration. The market is witnessing a surge in the development of novel assays and platforms, including advanced Diagnostic Tests that can identify a wider range of actionable mutations and resistance mechanisms. The development of Gene Carrier Tests is also gaining traction, empowering individuals with knowledge about their genetic risk and enabling informed reproductive decisions. The Parent Market of Genetic Testing and Diagnostics is projected to reach $65.2 billion by 2025, and the Child Market of Oncology Gene Profiling, a direct component of this report's focus, is a significant contributor to this overall growth.
Dominant Regions, Countries, or Segments in Gene Detection for Individualized Cancer Therapy
North America, particularly the United States, currently holds a dominant position in the Gene Detection for Individualized Cancer Therapy Market. This leadership is attributed to several interconnected factors, including robust healthcare infrastructure, high R&D investment, favorable regulatory environments, and a well-established reimbursement system for genetic testing. The presence of leading pharmaceutical and biotechnology companies, coupled with a strong academic research base, fosters rapid innovation and adoption of advanced diagnostic technologies. Economic policies that encourage technological advancement and a significant patient population with a high awareness of personalized medicine contribute to market supremacy. The widespread adoption of Diagnostic Tests for various cancer types and the increasing prevalence of Genetic Screening programs are key drivers.
The Application segment of Hospitals is a significant contributor to market growth in North America, as these institutions are at the forefront of adopting new diagnostic and therapeutic approaches. Diagnostic Centers also play a crucial role by providing specialized genetic testing services. Within the Types of gene detection, Diagnostic Test represents the largest segment, driven by the need for precise molecular characterization of tumors for treatment selection. However, the growth potential for Testing Before Symptoms Appear and Genetic Screening is immense, as awareness and proactive health management increase. Countries like Germany and the United Kingdom also demonstrate strong growth in Europe, driven by government initiatives to integrate precision medicine into national healthcare systems. Asia-Pacific, led by China and Japan, is emerging as a rapidly growing region, fueled by increasing healthcare expenditure, a burgeoning middle class, and a growing focus on R&D in the life sciences sector. The economic policies in these regions are increasingly supportive of advanced medical technologies.
- Leading Region: North America (primarily the United States).
- Key Drivers in North America:
- High R&D expenditure and strong biopharmaceutical industry.
- Advanced healthcare infrastructure and accessibility to cutting-edge technologies.
- Favorable reimbursement policies for genetic testing.
- High prevalence of cancer and increasing patient demand for personalized treatments.
- Early adoption of Diagnostic Test and Genetic Screening applications.
- Dominant Application Segment: Hospitals and Diagnostic Centers.
- Dominant Type Segment: Diagnostic Test, with significant growth in Genetic Screening and Testing Before Symptoms Appear.
- Growth Potential: High in Asia-Pacific and emerging markets due to increasing healthcare investment and rising awareness.
Gene Detection for Individualized Cancer Therapy Product Landscape
The Gene Detection for Individualized Cancer Therapy Market is characterized by a continuous stream of product innovations focused on enhancing accuracy, speed, and comprehensiveness. Technologies like Next-Generation Sequencing (NGS) are central, enabling the simultaneous analysis of multiple genes and genomic alterations. Innovations include multiplex PCR-based panels for targeted gene detection and the refinement of liquid biopsy techniques for non-invasive tumor profiling from blood or other bodily fluids. Performance metrics such as sensitivity, specificity, and turnaround time are critical selling points. Unique selling propositions often revolve around the breadth of genes covered, the ability to detect rare mutations, and the integration of bioinformatics tools for sophisticated data analysis and interpretation, aiding in the selection of targeted therapies and immunotherapies.
Key Drivers, Barriers & Challenges in Gene Detection for Individualized Cancer Therapy
The Gene Detection for Individualized Cancer Therapy Market is propelled by several key drivers. The increasing global cancer burden and the growing demand for personalized medicine are paramount. Advancements in genomic technologies, such as NGS, have significantly improved the feasibility and affordability of gene detection. Growing clinical evidence demonstrating improved patient outcomes with targeted therapies also fuels market growth. Furthermore, supportive government initiatives and increasing healthcare expenditure in many regions are accelerating adoption.
- Key Drivers:
- Rising cancer incidence.
- Demand for precision oncology.
- Technological advancements in NGS and liquid biopsy.
- Improved efficacy of targeted therapies.
- Favorable reimbursement policies and government support.
Conversely, the market faces several barriers and challenges. High upfront costs for advanced sequencing equipment and bioinformatics infrastructure can be a restraint, particularly for smaller healthcare facilities. Stringent regulatory approval processes for new diagnostic tests can also delay market entry. Lack of standardized protocols for sample handling, assay validation, and data interpretation can lead to variability in results. The shortage of skilled personnel, including genetic counselors and bioinformaticians, presents a significant operational challenge. Limited awareness among some healthcare professionals and patients regarding the benefits of gene detection also hinders widespread adoption.
- Key Barriers & Challenges:
- High cost of technology and infrastructure.
- Complex and lengthy regulatory approval processes.
- Need for standardization in protocols and data analysis.
- Shortage of skilled professionals.
- Limited awareness and understanding among some stakeholders.
- Ethical considerations and data privacy concerns.
Emerging Opportunities in Gene Detection for Individualized Cancer Therapy
Emerging opportunities in the Gene Detection for Individualized Cancer Therapy Market lie in the expansion of liquid biopsy applications for early cancer detection and monitoring of treatment response. The development of advanced AI-powered platforms for comprehensive genomic data analysis and predictive modeling offers significant potential for identifying novel therapeutic targets and patient stratification strategies. Untapped markets in developing economies, with increasing healthcare investments, present substantial growth prospects. Furthermore, the integration of gene detection with companion diagnostics for a wider range of therapeutic agents, including novel immunotherapies, is a rapidly evolving area. The growing interest in germline testing for hereditary cancer predispositions, enabling proactive risk management and preventative strategies, also represents a significant opportunity.
Growth Accelerators in the Gene Detection for Individualized Cancer Therapy Industry
Key growth accelerators in the Gene Detection for Individualized Cancer Therapy Industry include continued technological breakthroughs in sequencing accuracy, speed, and cost-effectiveness. Strategic partnerships between diagnostic companies, pharmaceutical firms, and academic institutions are fostering innovation and accelerating the translation of research into clinical applications. Market expansion strategies, focusing on emerging economies and underserved patient populations, will be critical for long-term growth. The increasing regulatory support for precision medicine and the development of clear reimbursement pathways for gene detection services will further catalyze market expansion.
Key Players Shaping the Gene Detection for Individualized Cancer Therapy Market
- Premed
- Yin Feng Gene
- United Gene Group
- Geneis
- Topgen
- Annoroad
- Gene+
- Sanvalley
- Sansure
Notable Milestones in Gene Detection for Individualized Cancer Therapy Sector
- 2019: Launch of advanced AI-driven genomic analysis platforms to accelerate variant interpretation.
- 2020: Significant advancements in liquid biopsy technology, enabling detection of circulating tumor DNA (ctDNA) with enhanced sensitivity.
- 2021: Increased regulatory approvals for comprehensive genomic profiling (CGP) panels for various cancer types.
- 2022: Strategic mergers and acquisitions to consolidate market share and expand technological capabilities.
- 2023: Growing emphasis on real-world evidence generation to support the clinical utility of gene detection in diverse patient populations.
- 2024: Development of novel assay designs for detecting low-frequency mutations and resistance mechanisms.
In-Depth Gene Detection for Individualized Cancer Therapy Market Outlook
The future outlook for the Gene Detection for Individualized Cancer Therapy Market is exceptionally bright, driven by ongoing technological innovation and a fundamental shift towards personalized healthcare. Growth accelerators such as advancements in liquid biopsy for early detection and minimal residual disease monitoring, coupled with sophisticated AI-driven data interpretation, will continue to shape the market. Strategic collaborations and market expansion into emerging economies will unlock new avenues for revenue generation. The increasing integration of gene detection with immunotherapies and the growing focus on hereditary cancer predisposition testing will further solidify its indispensable role in modern oncology. The market is set for sustained high growth, promising enhanced patient outcomes and a more precise approach to cancer management.
Gene Detection for Individualized Cancer Therapy Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Clinic
- 1.3. Diagnostic Center
-
2. Types
- 2.1. Genetic Screening
- 2.2. Reproductive Genetic Testing
- 2.3. Diagnostic Test
- 2.4. Gene carrier Test
- 2.5. Testing Before Symptoms Appear
Gene Detection for Individualized Cancer Therapy Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
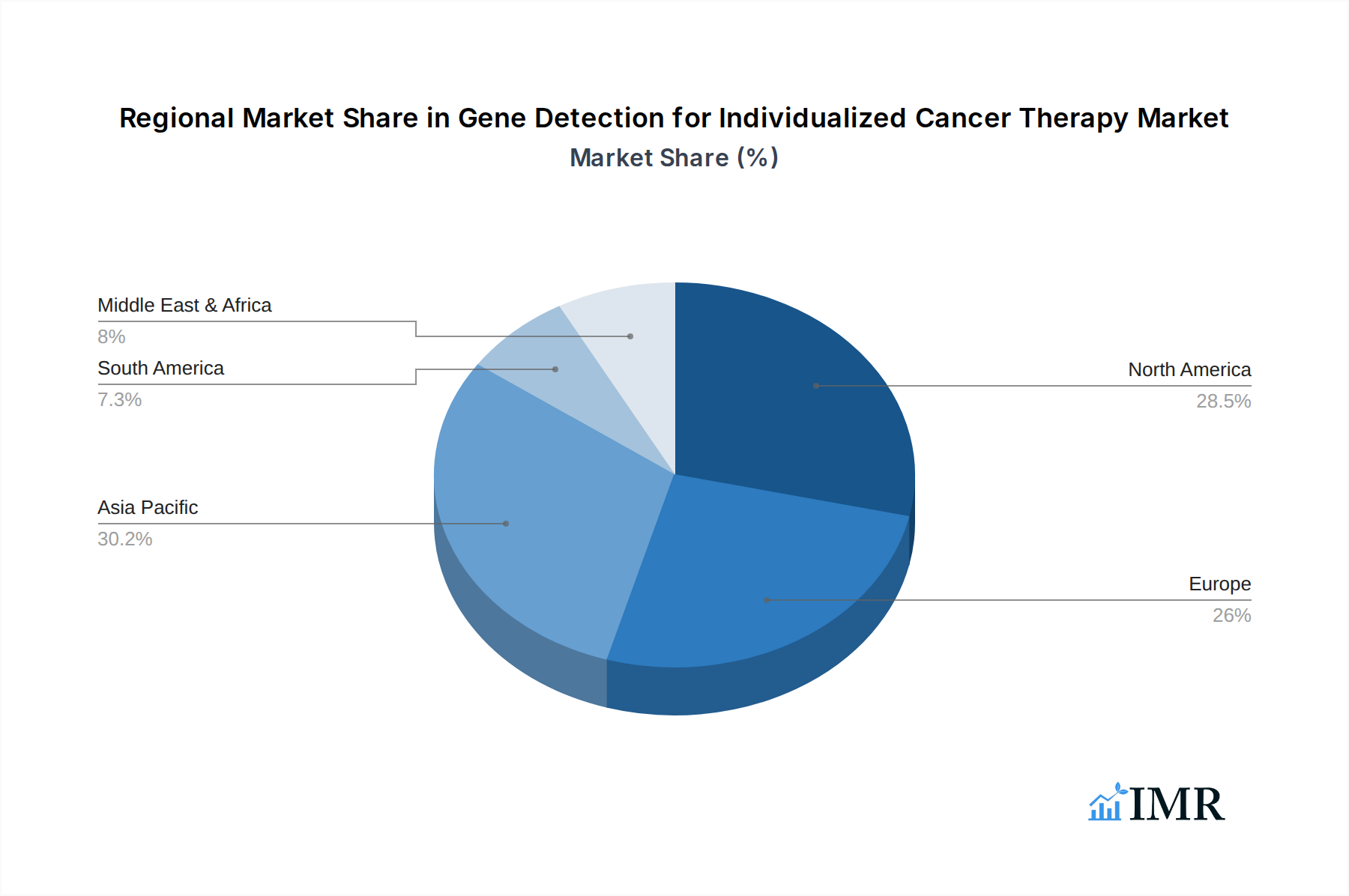
Gene Detection for Individualized Cancer Therapy Regional Market Share

Geographic Coverage of Gene Detection for Individualized Cancer Therapy
Gene Detection for Individualized Cancer Therapy REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 24.01% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global Gene Detection for Individualized Cancer Therapy Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Clinic
- 5.1.3. Diagnostic Center
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Genetic Screening
- 5.2.2. Reproductive Genetic Testing
- 5.2.3. Diagnostic Test
- 5.2.4. Gene carrier Test
- 5.2.5. Testing Before Symptoms Appear
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America Gene Detection for Individualized Cancer Therapy Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Clinic
- 6.1.3. Diagnostic Center
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Genetic Screening
- 6.2.2. Reproductive Genetic Testing
- 6.2.3. Diagnostic Test
- 6.2.4. Gene carrier Test
- 6.2.5. Testing Before Symptoms Appear
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America Gene Detection for Individualized Cancer Therapy Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Clinic
- 7.1.3. Diagnostic Center
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Genetic Screening
- 7.2.2. Reproductive Genetic Testing
- 7.2.3. Diagnostic Test
- 7.2.4. Gene carrier Test
- 7.2.5. Testing Before Symptoms Appear
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe Gene Detection for Individualized Cancer Therapy Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Clinic
- 8.1.3. Diagnostic Center
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Genetic Screening
- 8.2.2. Reproductive Genetic Testing
- 8.2.3. Diagnostic Test
- 8.2.4. Gene carrier Test
- 8.2.5. Testing Before Symptoms Appear
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa Gene Detection for Individualized Cancer Therapy Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Clinic
- 9.1.3. Diagnostic Center
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Genetic Screening
- 9.2.2. Reproductive Genetic Testing
- 9.2.3. Diagnostic Test
- 9.2.4. Gene carrier Test
- 9.2.5. Testing Before Symptoms Appear
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific Gene Detection for Individualized Cancer Therapy Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Clinic
- 10.1.3. Diagnostic Center
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Genetic Screening
- 10.2.2. Reproductive Genetic Testing
- 10.2.3. Diagnostic Test
- 10.2.4. Gene carrier Test
- 10.2.5. Testing Before Symptoms Appear
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 Premed
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Yin Feng Gene
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 United Gene Group
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Geneis
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Topgen
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.6 Annoroad
- 11.2.6.1. Overview
- 11.2.6.2. Products
- 11.2.6.3. SWOT Analysis
- 11.2.6.4. Recent Developments
- 11.2.6.5. Financials (Based on Availability)
- 11.2.7 Gene+
- 11.2.7.1. Overview
- 11.2.7.2. Products
- 11.2.7.3. SWOT Analysis
- 11.2.7.4. Recent Developments
- 11.2.7.5. Financials (Based on Availability)
- 11.2.8 Sanvalley
- 11.2.8.1. Overview
- 11.2.8.2. Products
- 11.2.8.3. SWOT Analysis
- 11.2.8.4. Recent Developments
- 11.2.8.5. Financials (Based on Availability)
- 11.2.9 Sansure
- 11.2.9.1. Overview
- 11.2.9.2. Products
- 11.2.9.3. SWOT Analysis
- 11.2.9.4. Recent Developments
- 11.2.9.5. Financials (Based on Availability)
- 11.2.1 Premed
List of Figures
- Figure 1: Global Gene Detection for Individualized Cancer Therapy Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific Gene Detection for Individualized Cancer Therapy Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific Gene Detection for Individualized Cancer Therapy Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global Gene Detection for Individualized Cancer Therapy Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific Gene Detection for Individualized Cancer Therapy Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the Gene Detection for Individualized Cancer Therapy?
The projected CAGR is approximately 24.01%.
2. Which companies are prominent players in the Gene Detection for Individualized Cancer Therapy?
Key companies in the market include Premed, Yin Feng Gene, United Gene Group, Geneis, Topgen, Annoroad, Gene+, Sanvalley, Sansure.
3. What are the main segments of the Gene Detection for Individualized Cancer Therapy?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 5600.00, USD 8400.00, and USD 11200.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "Gene Detection for Individualized Cancer Therapy," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the Gene Detection for Individualized Cancer Therapy report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the Gene Detection for Individualized Cancer Therapy?
To stay informed about further developments, trends, and reports in the Gene Detection for Individualized Cancer Therapy, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
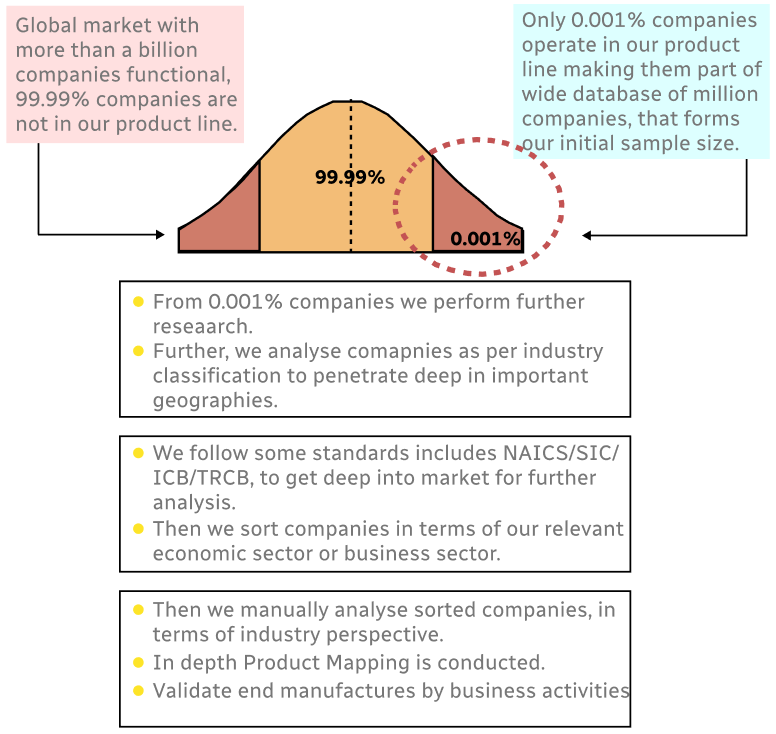
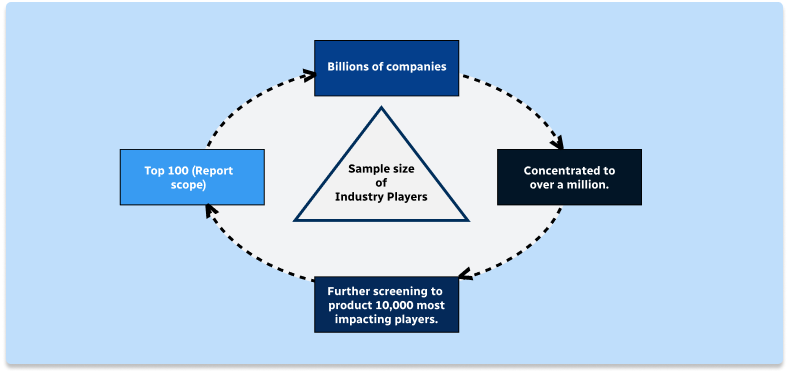
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
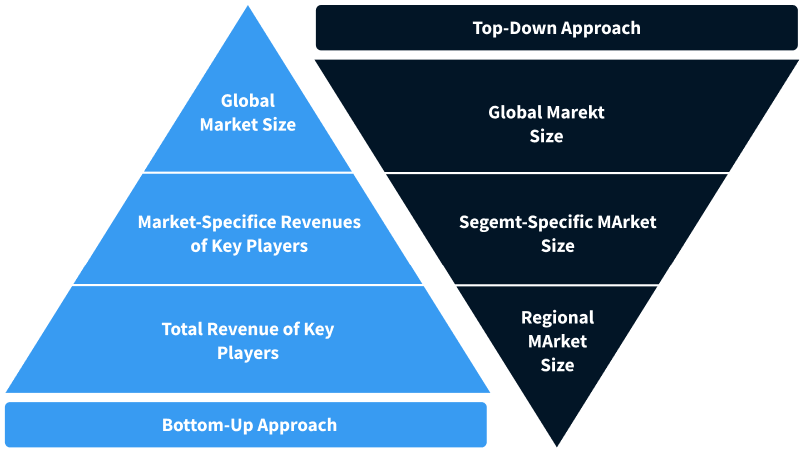
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
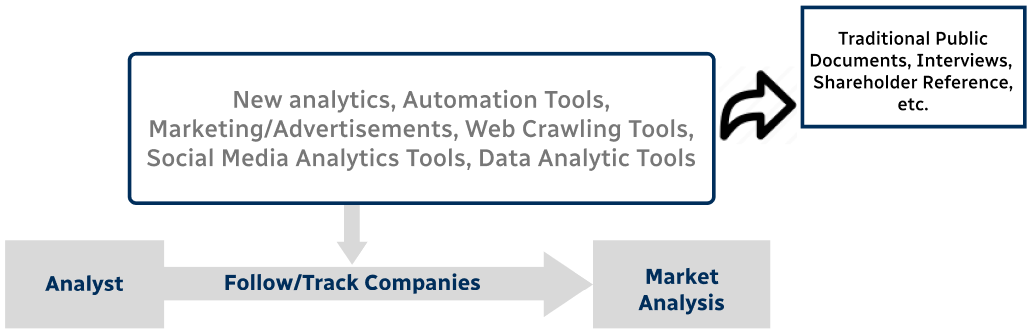
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


