Key Insights
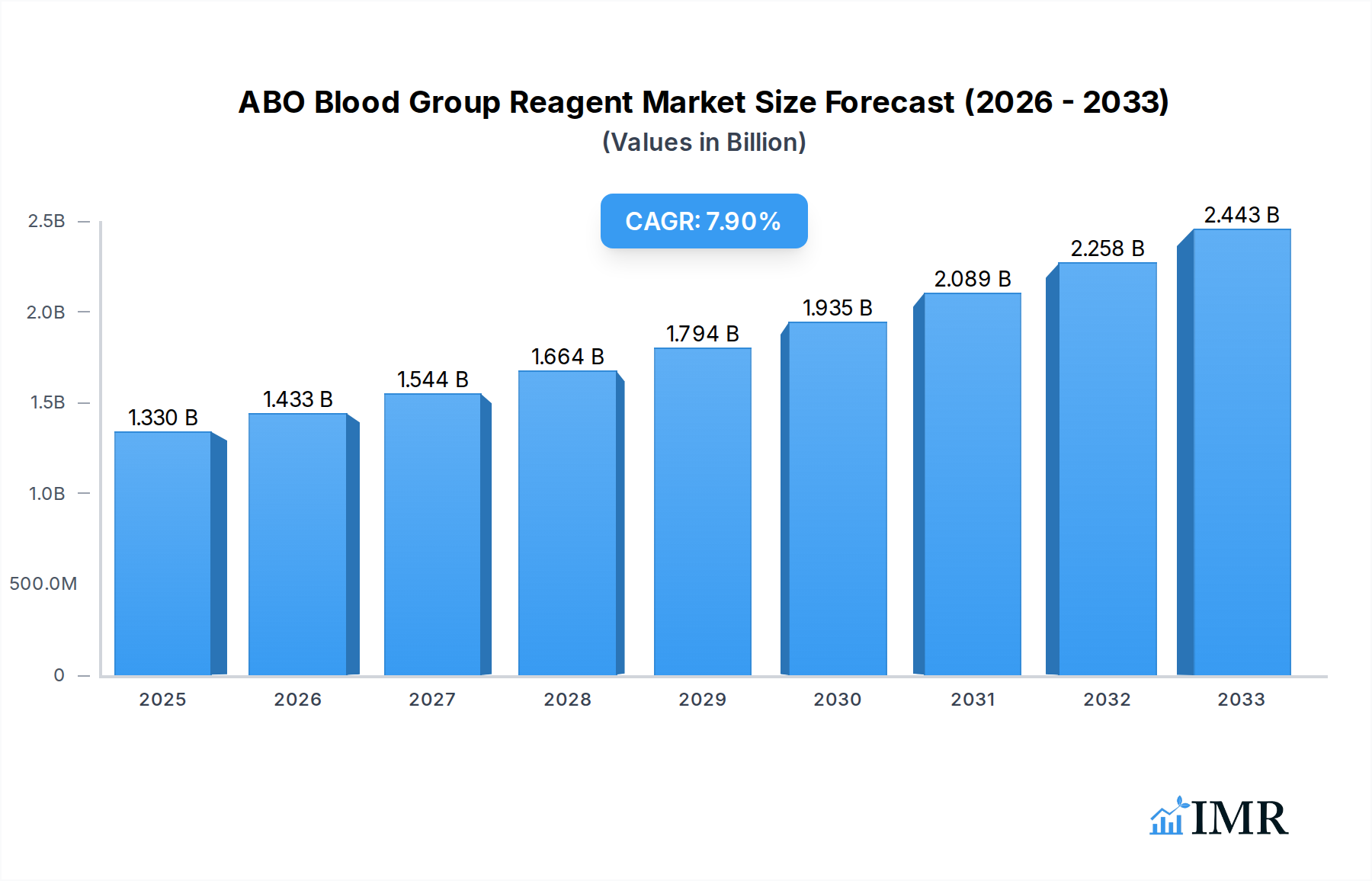
The ABO Blood Group Reagent market is poised for significant expansion, estimated to reach $1.33 billion in 2025. This robust growth is fueled by an anticipated Compound Annual Growth Rate (CAGR) of 7.8%, projecting a dynamic trajectory over the forecast period. Key drivers propelling this market forward include the increasing demand for accurate and efficient blood typing in clinical diagnostics, particularly for hospital transfusions and blood bank operations. The rising prevalence of blood-related disorders and the continuous need for blood donations further underscore the critical role of ABO blood group reagents. Advancements in reagent technology, leading to improved sensitivity and specificity, are also contributing to market expansion. Furthermore, growing awareness about the importance of proper blood group identification in preventing transfusion reactions is a crucial factor driving adoption across healthcare settings.
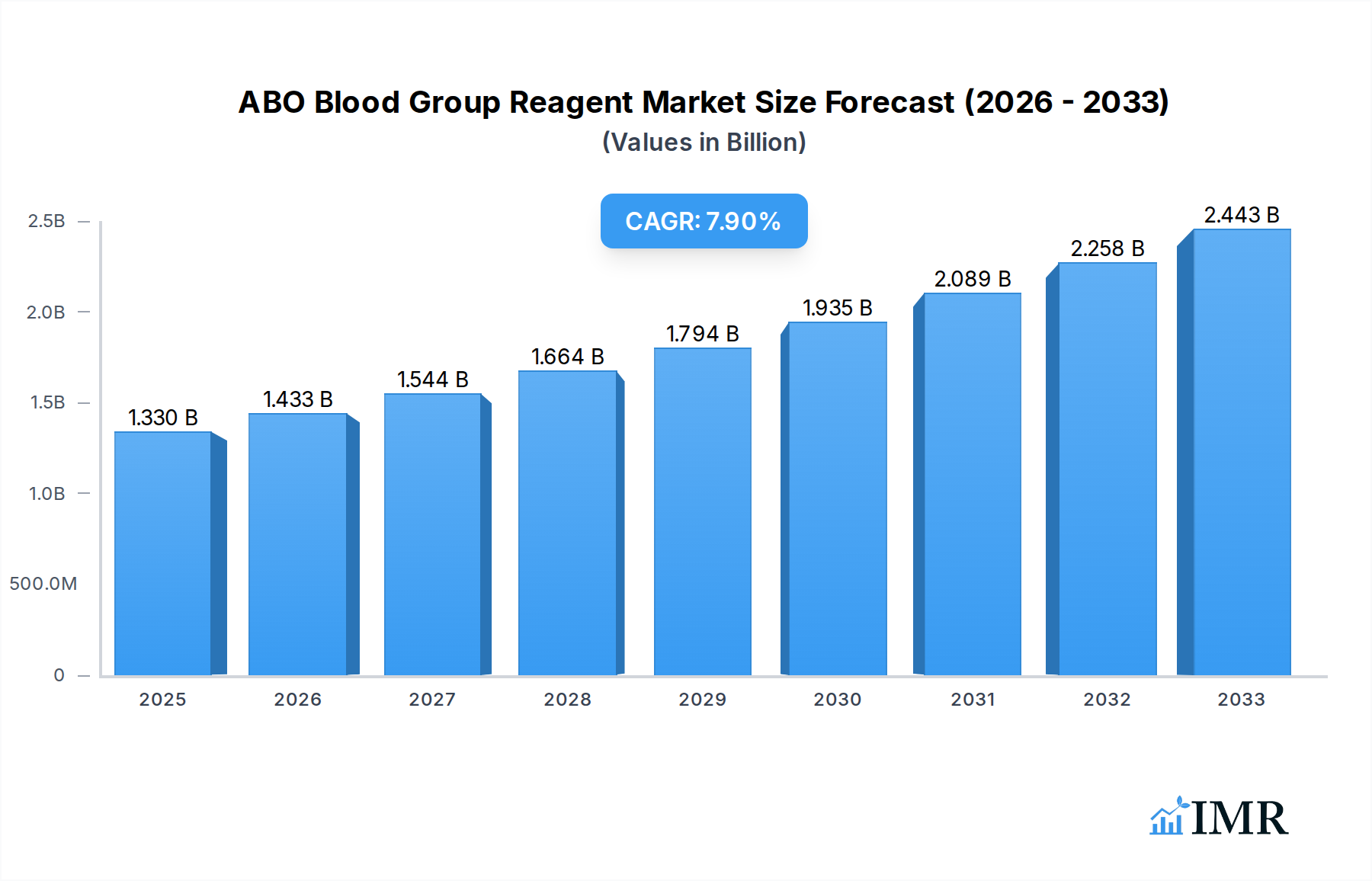
ABO Blood Group Reagent Market Size (In Billion)

The market is segmented by application into Hospitals, Blood Banks, Research, and Other. Hospitals and Blood Banks represent the largest segments due to their direct involvement in patient care and blood supply management. The "Positive Identification" and "Reverse Identification" types of ABO blood group reagents cater to distinct diagnostic needs, with both experiencing consistent demand. Geographically, North America and Europe are expected to lead the market, owing to well-established healthcare infrastructures and high adoption rates of advanced diagnostic technologies. However, the Asia Pacific region is projected to witness the fastest growth, driven by expanding healthcare access, increasing investments in diagnostic laboratories, and a growing awareness of blood safety protocols. Key players like CE-Immundiagnostika GmbH, Dominion Biologicals Limited, and SHPBC are actively innovating to capture market share through product development and strategic partnerships.
ABO Blood Group Reagent Company Market Share

ABO Blood Group Reagent Market: Comprehensive Analysis and Future Outlook (2019-2033)
This report offers an in-depth analysis of the global ABO Blood Group Reagent market, providing critical insights for industry stakeholders. Covering historical trends, current dynamics, and future projections, it details market size, growth drivers, regional dominance, product landscape, and competitive intelligence. The study utilizes a robust methodology, encompassing a study period from 2019 to 2033, with 2025 as the base and estimated year, and a forecast period from 2025 to 2033, supported by historical data from 2019 to 2024.
ABO Blood Group Reagent Market Dynamics & Structure
The ABO Blood Group Reagent market is characterized by a moderately concentrated structure, with key players strategically positioning themselves through innovation and targeted acquisitions. Technological advancements in reagent formulation, particularly enhanced sensitivity and specificity, act as significant drivers of market growth. Regulatory frameworks, such as those governed by the FDA and EMA, play a crucial role in ensuring product safety and efficacy, influencing market entry and product development strategies. Competitive product substitutes, while limited in core ABO typing, exist in ancillary diagnostic tools. End-user demographics, primarily healthcare professionals in hospitals, blood banks, and research institutions, dictate demand patterns. Mergers and acquisitions (M&A) activity, while not overtly high, is strategic, aimed at consolidating market share and expanding product portfolios. For instance, the past three years have seen an estimated XX M&A deals, with an average deal value of $XX billion, indicating consolidation and synergy-seeking among larger entities. Barriers to innovation include the stringent validation processes required for diagnostic reagents and the need for substantial R&D investment.
- Market Concentration: Moderately concentrated with a few dominant players.
- Technological Drivers: Enhanced reagent sensitivity, specificity, and automation compatibility.
- Regulatory Frameworks: FDA, EMA, and national health authorities ensuring quality and safety standards.
- Competitive Substitutes: Ancillary diagnostics for broader blood component analysis.
- End-User Demographics: Hospitals, blood banks, research facilities, diagnostic laboratories.
- M&A Trends: Strategic acquisitions for market expansion and portfolio diversification.
- Innovation Barriers: Rigorous validation processes, high R&D costs.
ABO Blood Group Reagent Growth Trends & Insights
The global ABO Blood Group Reagent market is poised for significant expansion, driven by increasing blood transfusion demands, advancements in diagnostic technologies, and a growing emphasis on patient safety in healthcare settings. The market size, projected to reach approximately $XX billion by 2025, is anticipated to grow at a Compound Annual Growth Rate (CAGR) of X.X% during the forecast period of 2025-2033. This growth is fueled by rising prevalence of chronic diseases requiring blood transfusions and the continuous need for accurate blood typing in organ transplantation procedures. Adoption rates for automated blood typing systems, which enhance efficiency and reduce human error, are steadily increasing, particularly in large hospitals and blood banks. Technological disruptions are emerging in the form of microfluidic-based reagents and advanced serological techniques, offering faster and more precise results. Consumer behavior shifts are observed towards demand for integrated diagnostic solutions and user-friendly reagent kits. The historical period from 2019 to 2024 saw a steady market growth of approximately X.X% CAGR, with increased adoption of rapid diagnostic tests and a surge in demand for blood components due to the global pandemic. The market penetration of advanced ABO blood group reagents is estimated to be around XX% in developed regions, with significant growth potential in emerging economies. The increasing incidence of road accidents and surgical procedures further bolsters the demand for ABO blood group reagents, as these are critical for emergency transfusions and pre-operative compatibility testing. Furthermore, the expanding pediatric population and the rising number of complex surgeries contribute to a sustained demand for reliable blood typing solutions. The integration of artificial intelligence (AI) in laboratory diagnostics is also beginning to influence reagent development, aiming for smarter, self-optimizing testing platforms.
Dominant Regions, Countries, or Segments in ABO Blood Group Reagent
North America currently holds a dominant position in the ABO Blood Group Reagent market, driven by its advanced healthcare infrastructure, high per capita healthcare expenditure, and strong regulatory oversight ensuring the quality and reliability of diagnostic reagents. The United States, in particular, is a significant contributor due to its extensive network of hospitals, blood banks, and research institutions, coupled with a proactive approach towards adopting new diagnostic technologies. The Application: Hospital segment is a primary growth engine within North America, accounting for an estimated XX% of the regional market share, owing to the high volume of blood transfusions required for various medical procedures. The Types: Positive Identification reagents are widely utilized due to their direct and conclusive results in determining blood group antigens. The region's robust reimbursement policies for diagnostic tests and government initiatives promoting blood donation further bolster market demand. Economic policies supporting healthcare innovation and infrastructure development, such as advanced laboratory automation, have significantly contributed to the sustained dominance of North America. The market share of North America in the global ABO Blood Group Reagent market is approximately XX billion units, with a projected CAGR of X.X% during the forecast period.
Asia Pacific, however, presents the highest growth potential. Countries like China and India are witnessing a rapid expansion of their healthcare sectors, driven by increasing populations, rising disposable incomes, and government investments in improving healthcare access. The Application: Blood Bank segment in these regions is experiencing substantial growth, as blood donation drives and blood storage infrastructure are being significantly enhanced. The adoption of Types: Reverse Identification reagents is also on the rise, complementing positive identification methods for comprehensive blood typing. Economic policies focused on public health and the increasing incidence of transfusion-dependent diseases are key drivers. Infrastructure development, including the establishment of new diagnostic laboratories and advanced blood processing centers, further accelerates market penetration. The market share of Asia Pacific is estimated at XX billion units, with a projected CAGR of X.X%, indicating a significant shift in regional dominance over the coming years.
- North America Dominance: Advanced healthcare, high expenditure, stringent regulations.
- United States Contribution: Extensive network of healthcare facilities, early technology adoption.
- Dominant Application (North America): Hospitals.
- Dominant Type (North America): Positive Identification.
- Asia Pacific Growth Potential: Rapid healthcare expansion, growing economies, increasing population.
- Dominant Application (Asia Pacific): Blood Banks.
- Dominant Type (Asia Pacific): Reverse Identification.
- Key Drivers in Asia Pacific: Economic policies, infrastructure development, disease prevalence.
ABO Blood Group Reagent Product Landscape
The ABO Blood Group Reagent product landscape is defined by continuous innovation focused on improving accuracy, speed, and ease of use. Manufacturers are developing highly specific monoclonal antibody-based reagents that minimize cross-reactivity and deliver unambiguous ABO and Rh blood typing results. Advanced formulations offer extended shelf lives and improved stability under varying storage conditions, crucial for global supply chains. Applications span routine pre-transfusion testing in hospitals and blood banks to specialized research in immunohematology and serology. Performance metrics such as sensitivity, specificity, and reaction time are continuously being optimized. Unique selling propositions include the development of ready-to-use liquid reagents and kits compatible with automated immunoassay analyzers, significantly reducing turnaround times and laboratory workload. Technological advancements are also exploring novel detection methods for even rarer blood group antigens.
Key Drivers, Barriers & Challenges in ABO Blood Group Reagent
The ABO Blood Group Reagent market is propelled by several key drivers. Foremost among these is the increasing global demand for blood transfusions, driven by aging populations, rising chronic disease prevalence, and advancements in surgical procedures. Technological innovation, leading to more sensitive, specific, and automated reagent systems, enhances diagnostic accuracy and efficiency. The growing emphasis on patient safety and accurate compatibility testing further fuels market growth. Stringent regulatory approvals, while a barrier to entry, also ensure product quality and reliability, indirectly driving demand for compliant reagents.
However, the market faces significant barriers and challenges. High research and development costs associated with developing and validating new diagnostic reagents represent a substantial financial hurdle for many companies. Stringent regulatory approval processes, requiring extensive clinical trials and documentation, can lead to prolonged market entry timelines. Competitive pressures from established players and the threat of alternative diagnostic methodologies, though less prevalent for core ABO typing, can impact market share. Supply chain disruptions, as seen during global health crises, can affect the availability of raw materials and finished products, leading to stockouts and increased costs.
Key Drivers:
- Increasing demand for blood transfusions.
- Technological advancements in reagent formulation.
- Emphasis on patient safety and accurate compatibility testing.
- Stringent regulatory standards ensuring quality.
Barriers & Challenges:
- High R&D investment and validation costs.
- Lengthy and complex regulatory approval processes.
- Intense competition among market players.
- Potential for supply chain disruptions.
- Price sensitivity in certain markets.
Emerging Opportunities in ABO Blood Group Reagent
Emerging opportunities in the ABO Blood Group Reagent market lie in the development of integrated diagnostic platforms that combine ABO typing with other essential blood grouping tests, offering a more comprehensive and efficient solution for laboratories. The untapped potential in emerging economies, particularly in Africa and parts of Asia, presents significant growth prospects as healthcare infrastructure expands and access to diagnostic services improves. Innovations in point-of-care testing (POCT) for ABO blood grouping, enabling rapid testing at the bedside or in remote settings, is another promising avenue. Evolving consumer preferences for user-friendly, automated systems that minimize manual intervention and reduce the risk of human error also create opportunities for novel reagent kits and analyzers. Furthermore, the increasing focus on personalized medicine and the need for precise blood matching in complex transplant procedures will drive demand for highly specialized and accurate ABO blood group reagents.
Growth Accelerators in the ABO Blood Group Reagent Industry
Several catalysts are driving long-term growth in the ABO Blood Group Reagent industry. Technological breakthroughs in molecular diagnostics and genomics are paving the way for more sophisticated blood typing techniques, which may eventually complement or enhance traditional serological methods, leading to broader market appeal. Strategic partnerships between reagent manufacturers and diagnostic instrument companies are crucial for developing integrated solutions that improve laboratory workflow and efficiency. Market expansion strategies focused on penetrating underserved regions with tailored product offerings and competitive pricing will unlock new revenue streams. The increasing adoption of cloud-based laboratory information management systems (LIMS) will facilitate better data management and traceability for blood group testing, indirectly boosting confidence and demand for reliable reagents. Furthermore, a growing awareness of the importance of accurate blood typing in preventing transfusion reactions and ensuring patient safety will continue to be a significant growth accelerator.
Key Players Shaping the ABO Blood Group Reagent Market
- CE-Immundiagnostika GmbH
- Dominion Biologicals Limited
- SHPBC
- Brother Biotech
- Kinghawk
Notable Milestones in ABO Blood Group Reagent Sector
- 2019: Introduction of advanced liquid stable reagents offering enhanced shelf-life and room temperature storage.
- 2020: Increased demand for rapid ABO typing kits due to global health emergencies.
- 2021: Development of automated ABO blood grouping systems with reduced hands-on time.
- 2022: Focus on eco-friendly packaging and sustainable manufacturing practices by key players.
- 2023: Integration of AI-powered quality control features in some reagent batches.
- 2024: Expansion of product portfolios to include reagents for rare blood group phenotyping alongside ABO.
In-Depth ABO Blood Group Reagent Market Outlook
The future of the ABO Blood Group Reagent market is characterized by sustained growth driven by ongoing technological advancements and expanding global healthcare access. Key growth accelerators include the continued demand for blood transfusions, the development of more sophisticated and automated diagnostic systems, and strategic market expansion into emerging economies. The increasing emphasis on patient safety and the need for accurate blood matching in complex medical procedures will further bolster the market. Opportunities for innovation lie in integrated diagnostic solutions, point-of-care testing, and reagents catering to personalized medicine. Stakeholders are advised to focus on product differentiation through enhanced sensitivity, specificity, and user-friendliness, while also exploring strategic partnerships to leverage technological synergies and expand market reach. The global ABO Blood Group Reagent market is projected to continue its upward trajectory, presenting significant opportunities for players who can adapt to evolving technological landscapes and address the growing global demand for reliable blood typing diagnostics.
ABO Blood Group Reagent Segmentation
-
1. Application
- 1.1. Hospital
- 1.2. Blood Bank
- 1.3. Research
- 1.4. Other
-
2. Types
- 2.1. Positive Identification
- 2.2. Reverse Identification
ABO Blood Group Reagent Segmentation By Geography
-
1. North America
- 1.1. United States
- 1.2. Canada
- 1.3. Mexico
-
2. South America
- 2.1. Brazil
- 2.2. Argentina
- 2.3. Rest of South America
-
3. Europe
- 3.1. United Kingdom
- 3.2. Germany
- 3.3. France
- 3.4. Italy
- 3.5. Spain
- 3.6. Russia
- 3.7. Benelux
- 3.8. Nordics
- 3.9. Rest of Europe
-
4. Middle East & Africa
- 4.1. Turkey
- 4.2. Israel
- 4.3. GCC
- 4.4. North Africa
- 4.5. South Africa
- 4.6. Rest of Middle East & Africa
-
5. Asia Pacific
- 5.1. China
- 5.2. India
- 5.3. Japan
- 5.4. South Korea
- 5.5. ASEAN
- 5.6. Oceania
- 5.7. Rest of Asia Pacific
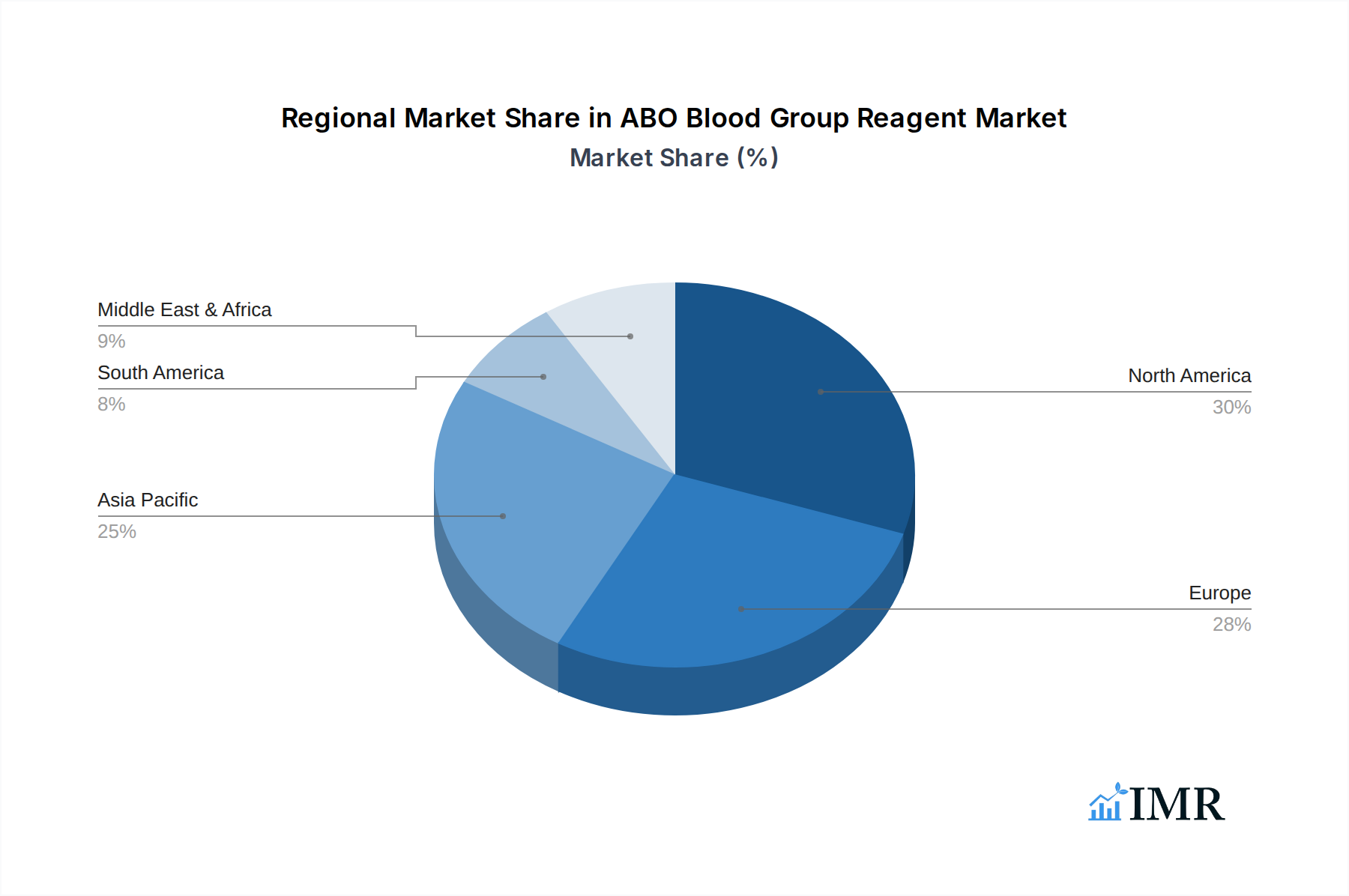
ABO Blood Group Reagent Regional Market Share

Geographic Coverage of ABO Blood Group Reagent
ABO Blood Group Reagent REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.8% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. Global ABO Blood Group Reagent Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Hospital
- 5.1.2. Blood Bank
- 5.1.3. Research
- 5.1.4. Other
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Positive Identification
- 5.2.2. Reverse Identification
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. North America
- 5.3.2. South America
- 5.3.3. Europe
- 5.3.4. Middle East & Africa
- 5.3.5. Asia Pacific
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. North America ABO Blood Group Reagent Analysis, Insights and Forecast, 2020-2032
- 6.1. Market Analysis, Insights and Forecast - by Application
- 6.1.1. Hospital
- 6.1.2. Blood Bank
- 6.1.3. Research
- 6.1.4. Other
- 6.2. Market Analysis, Insights and Forecast - by Types
- 6.2.1. Positive Identification
- 6.2.2. Reverse Identification
- 6.1. Market Analysis, Insights and Forecast - by Application
- 7. South America ABO Blood Group Reagent Analysis, Insights and Forecast, 2020-2032
- 7.1. Market Analysis, Insights and Forecast - by Application
- 7.1.1. Hospital
- 7.1.2. Blood Bank
- 7.1.3. Research
- 7.1.4. Other
- 7.2. Market Analysis, Insights and Forecast - by Types
- 7.2.1. Positive Identification
- 7.2.2. Reverse Identification
- 7.1. Market Analysis, Insights and Forecast - by Application
- 8. Europe ABO Blood Group Reagent Analysis, Insights and Forecast, 2020-2032
- 8.1. Market Analysis, Insights and Forecast - by Application
- 8.1.1. Hospital
- 8.1.2. Blood Bank
- 8.1.3. Research
- 8.1.4. Other
- 8.2. Market Analysis, Insights and Forecast - by Types
- 8.2.1. Positive Identification
- 8.2.2. Reverse Identification
- 8.1. Market Analysis, Insights and Forecast - by Application
- 9. Middle East & Africa ABO Blood Group Reagent Analysis, Insights and Forecast, 2020-2032
- 9.1. Market Analysis, Insights and Forecast - by Application
- 9.1.1. Hospital
- 9.1.2. Blood Bank
- 9.1.3. Research
- 9.1.4. Other
- 9.2. Market Analysis, Insights and Forecast - by Types
- 9.2.1. Positive Identification
- 9.2.2. Reverse Identification
- 9.1. Market Analysis, Insights and Forecast - by Application
- 10. Asia Pacific ABO Blood Group Reagent Analysis, Insights and Forecast, 2020-2032
- 10.1. Market Analysis, Insights and Forecast - by Application
- 10.1.1. Hospital
- 10.1.2. Blood Bank
- 10.1.3. Research
- 10.1.4. Other
- 10.2. Market Analysis, Insights and Forecast - by Types
- 10.2.1. Positive Identification
- 10.2.2. Reverse Identification
- 10.1. Market Analysis, Insights and Forecast - by Application
- 11. Competitive Analysis
- 11.1. Global Market Share Analysis 2025
- 11.2. Company Profiles
- 11.2.1 CE-Immundiagnostika GmbH
- 11.2.1.1. Overview
- 11.2.1.2. Products
- 11.2.1.3. SWOT Analysis
- 11.2.1.4. Recent Developments
- 11.2.1.5. Financials (Based on Availability)
- 11.2.2 Dominion Biologicals Limited
- 11.2.2.1. Overview
- 11.2.2.2. Products
- 11.2.2.3. SWOT Analysis
- 11.2.2.4. Recent Developments
- 11.2.2.5. Financials (Based on Availability)
- 11.2.3 SHPBC
- 11.2.3.1. Overview
- 11.2.3.2. Products
- 11.2.3.3. SWOT Analysis
- 11.2.3.4. Recent Developments
- 11.2.3.5. Financials (Based on Availability)
- 11.2.4 Brother Biotech
- 11.2.4.1. Overview
- 11.2.4.2. Products
- 11.2.4.3. SWOT Analysis
- 11.2.4.4. Recent Developments
- 11.2.4.5. Financials (Based on Availability)
- 11.2.5 Kinghawk
- 11.2.5.1. Overview
- 11.2.5.2. Products
- 11.2.5.3. SWOT Analysis
- 11.2.5.4. Recent Developments
- 11.2.5.5. Financials (Based on Availability)
- 11.2.1 CE-Immundiagnostika GmbH
List of Figures
- Figure 1: Global ABO Blood Group Reagent Revenue Breakdown (undefined, %) by Region 2025 & 2033
- Figure 2: North America ABO Blood Group Reagent Revenue (undefined), by Application 2025 & 2033
- Figure 3: North America ABO Blood Group Reagent Revenue Share (%), by Application 2025 & 2033
- Figure 4: North America ABO Blood Group Reagent Revenue (undefined), by Types 2025 & 2033
- Figure 5: North America ABO Blood Group Reagent Revenue Share (%), by Types 2025 & 2033
- Figure 6: North America ABO Blood Group Reagent Revenue (undefined), by Country 2025 & 2033
- Figure 7: North America ABO Blood Group Reagent Revenue Share (%), by Country 2025 & 2033
- Figure 8: South America ABO Blood Group Reagent Revenue (undefined), by Application 2025 & 2033
- Figure 9: South America ABO Blood Group Reagent Revenue Share (%), by Application 2025 & 2033
- Figure 10: South America ABO Blood Group Reagent Revenue (undefined), by Types 2025 & 2033
- Figure 11: South America ABO Blood Group Reagent Revenue Share (%), by Types 2025 & 2033
- Figure 12: South America ABO Blood Group Reagent Revenue (undefined), by Country 2025 & 2033
- Figure 13: South America ABO Blood Group Reagent Revenue Share (%), by Country 2025 & 2033
- Figure 14: Europe ABO Blood Group Reagent Revenue (undefined), by Application 2025 & 2033
- Figure 15: Europe ABO Blood Group Reagent Revenue Share (%), by Application 2025 & 2033
- Figure 16: Europe ABO Blood Group Reagent Revenue (undefined), by Types 2025 & 2033
- Figure 17: Europe ABO Blood Group Reagent Revenue Share (%), by Types 2025 & 2033
- Figure 18: Europe ABO Blood Group Reagent Revenue (undefined), by Country 2025 & 2033
- Figure 19: Europe ABO Blood Group Reagent Revenue Share (%), by Country 2025 & 2033
- Figure 20: Middle East & Africa ABO Blood Group Reagent Revenue (undefined), by Application 2025 & 2033
- Figure 21: Middle East & Africa ABO Blood Group Reagent Revenue Share (%), by Application 2025 & 2033
- Figure 22: Middle East & Africa ABO Blood Group Reagent Revenue (undefined), by Types 2025 & 2033
- Figure 23: Middle East & Africa ABO Blood Group Reagent Revenue Share (%), by Types 2025 & 2033
- Figure 24: Middle East & Africa ABO Blood Group Reagent Revenue (undefined), by Country 2025 & 2033
- Figure 25: Middle East & Africa ABO Blood Group Reagent Revenue Share (%), by Country 2025 & 2033
- Figure 26: Asia Pacific ABO Blood Group Reagent Revenue (undefined), by Application 2025 & 2033
- Figure 27: Asia Pacific ABO Blood Group Reagent Revenue Share (%), by Application 2025 & 2033
- Figure 28: Asia Pacific ABO Blood Group Reagent Revenue (undefined), by Types 2025 & 2033
- Figure 29: Asia Pacific ABO Blood Group Reagent Revenue Share (%), by Types 2025 & 2033
- Figure 30: Asia Pacific ABO Blood Group Reagent Revenue (undefined), by Country 2025 & 2033
- Figure 31: Asia Pacific ABO Blood Group Reagent Revenue Share (%), by Country 2025 & 2033
List of Tables
- Table 1: Global ABO Blood Group Reagent Revenue undefined Forecast, by Application 2020 & 2033
- Table 2: Global ABO Blood Group Reagent Revenue undefined Forecast, by Types 2020 & 2033
- Table 3: Global ABO Blood Group Reagent Revenue undefined Forecast, by Region 2020 & 2033
- Table 4: Global ABO Blood Group Reagent Revenue undefined Forecast, by Application 2020 & 2033
- Table 5: Global ABO Blood Group Reagent Revenue undefined Forecast, by Types 2020 & 2033
- Table 6: Global ABO Blood Group Reagent Revenue undefined Forecast, by Country 2020 & 2033
- Table 7: United States ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 8: Canada ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 9: Mexico ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 10: Global ABO Blood Group Reagent Revenue undefined Forecast, by Application 2020 & 2033
- Table 11: Global ABO Blood Group Reagent Revenue undefined Forecast, by Types 2020 & 2033
- Table 12: Global ABO Blood Group Reagent Revenue undefined Forecast, by Country 2020 & 2033
- Table 13: Brazil ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 14: Argentina ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 15: Rest of South America ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 16: Global ABO Blood Group Reagent Revenue undefined Forecast, by Application 2020 & 2033
- Table 17: Global ABO Blood Group Reagent Revenue undefined Forecast, by Types 2020 & 2033
- Table 18: Global ABO Blood Group Reagent Revenue undefined Forecast, by Country 2020 & 2033
- Table 19: United Kingdom ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 20: Germany ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 21: France ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 22: Italy ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 23: Spain ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 24: Russia ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 25: Benelux ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 26: Nordics ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 27: Rest of Europe ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 28: Global ABO Blood Group Reagent Revenue undefined Forecast, by Application 2020 & 2033
- Table 29: Global ABO Blood Group Reagent Revenue undefined Forecast, by Types 2020 & 2033
- Table 30: Global ABO Blood Group Reagent Revenue undefined Forecast, by Country 2020 & 2033
- Table 31: Turkey ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 32: Israel ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 33: GCC ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 34: North Africa ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 35: South Africa ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 36: Rest of Middle East & Africa ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 37: Global ABO Blood Group Reagent Revenue undefined Forecast, by Application 2020 & 2033
- Table 38: Global ABO Blood Group Reagent Revenue undefined Forecast, by Types 2020 & 2033
- Table 39: Global ABO Blood Group Reagent Revenue undefined Forecast, by Country 2020 & 2033
- Table 40: China ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 41: India ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 42: Japan ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 43: South Korea ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 44: ASEAN ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 45: Oceania ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
- Table 46: Rest of Asia Pacific ABO Blood Group Reagent Revenue (undefined) Forecast, by Application 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the ABO Blood Group Reagent?
The projected CAGR is approximately 7.8%.
2. Which companies are prominent players in the ABO Blood Group Reagent?
Key companies in the market include CE-Immundiagnostika GmbH, Dominion Biologicals Limited, SHPBC, Brother Biotech, Kinghawk.
3. What are the main segments of the ABO Blood Group Reagent?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD XXX N/A as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 5600.00, USD 8400.00, and USD 11200.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in N/A.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "ABO Blood Group Reagent," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the ABO Blood Group Reagent report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the ABO Blood Group Reagent?
To stay informed about further developments, trends, and reports in the ABO Blood Group Reagent, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
Step 1 - Identification of Relevant Samples Size from Population Database
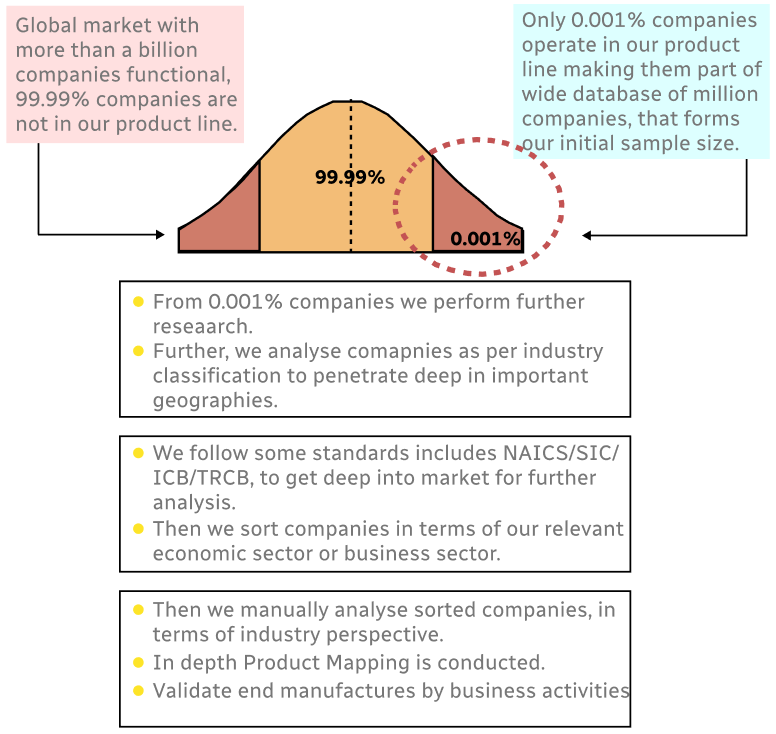
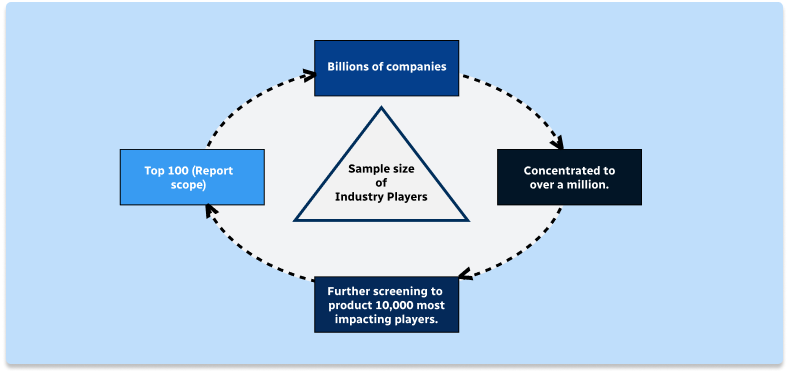
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
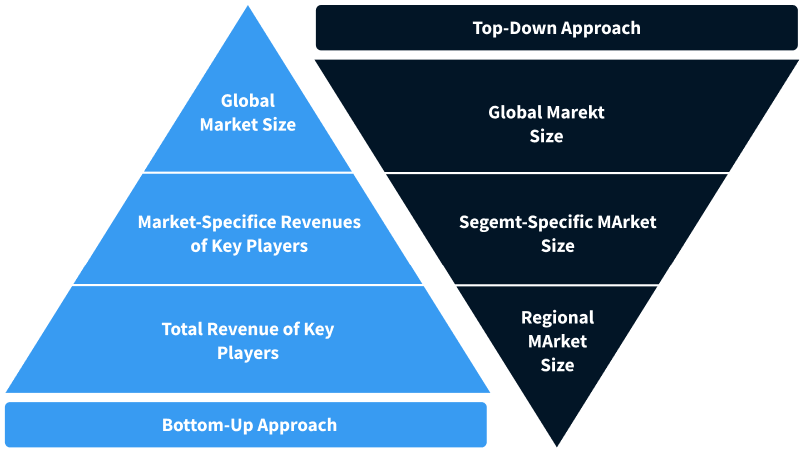
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
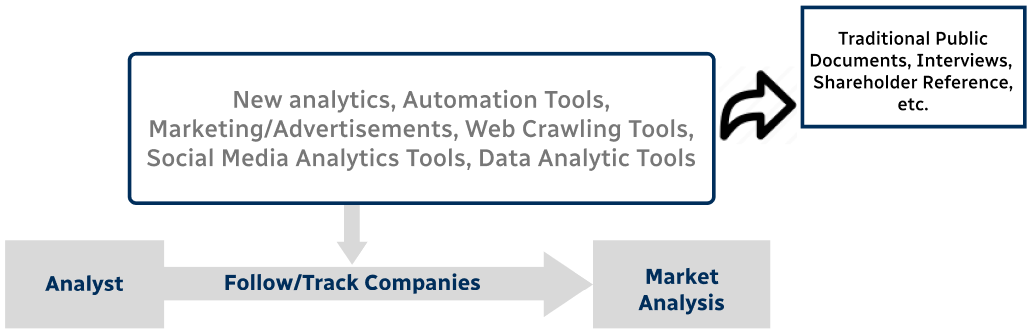
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


