Key Insights
The global market for targeted therapies for Non-Small Cell Lung Cancer (NSCLC) is poised for substantial growth, projected to reach $85.69 billion in 2025 with a robust Compound Annual Growth Rate (CAGR) of 6.87% through 2033. This expansion is primarily fueled by advancements in precision medicine and a deeper understanding of the molecular underpinnings of NSCLC. The market is segmented by application, encompassing squamous cell carcinoma, adenocarcinoma, and large cell carcinoma of NSCLC, with a significant focus on adenocarcinoma due to its higher prevalence and susceptibility to targeted interventions. The diverse array of targeted drug types, including those targeting EGFR, ALK, HER2, ROS1, BRAF, MEK, VEGFR2, VEGF, and MET, reflects the increasing sophistication of treatment strategies. These therapies offer a paradigm shift from traditional chemotherapy, providing more personalized and effective treatment options with potentially fewer side effects, thereby driving adoption and market value.
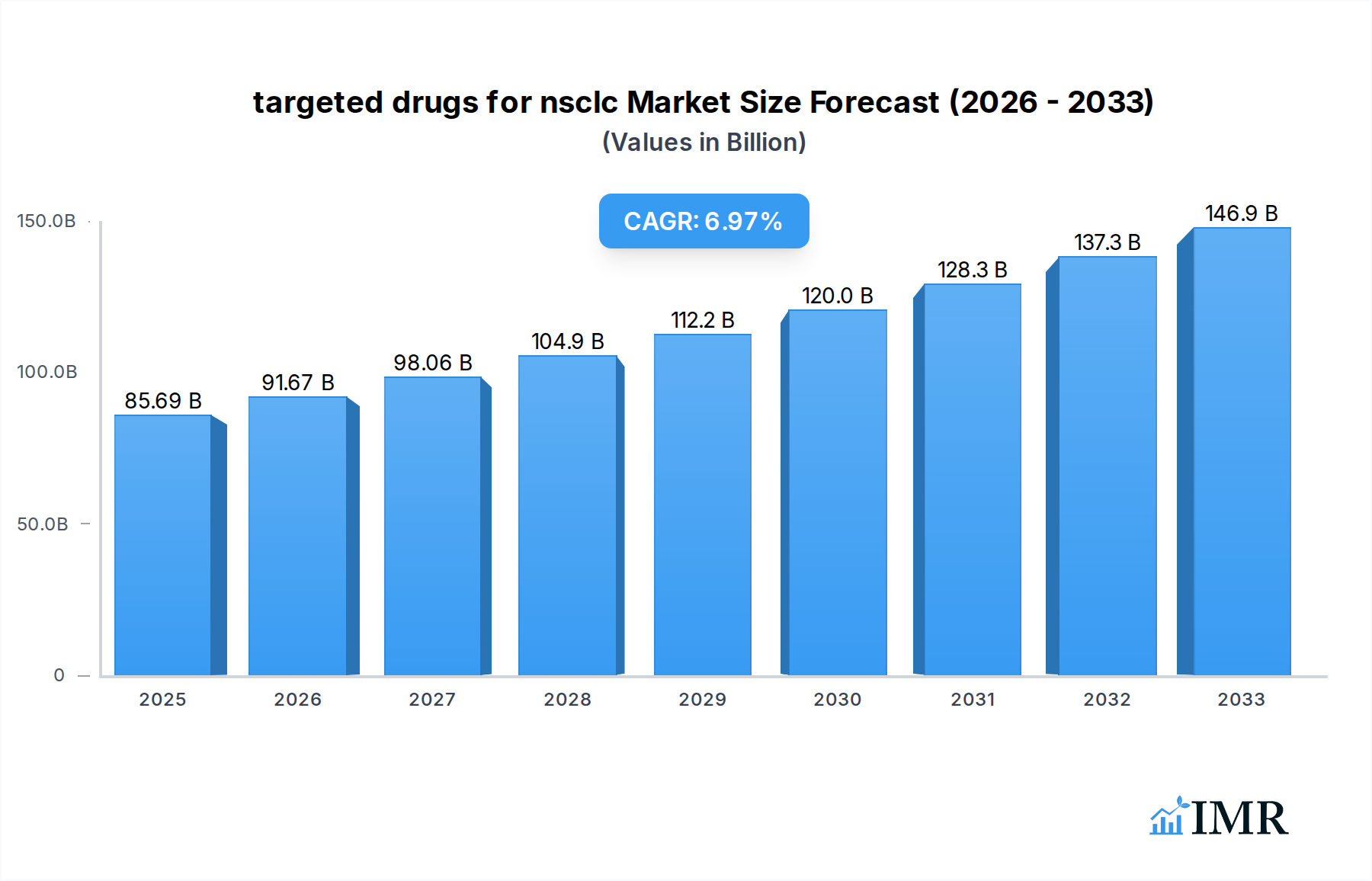
targeted drugs for nsclc Market Size (In Billion)

Key drivers for this market's trajectory include the escalating incidence of lung cancer globally, coupled with a growing demand for personalized medicine that targets specific genetic mutations driving tumor growth. Continuous research and development efforts by major pharmaceutical and biotechnology companies are leading to the discovery and approval of novel targeted drugs, expanding the treatment landscape. Furthermore, favorable reimbursement policies and increasing healthcare expenditure in developed and emerging economies are supporting market expansion. While the market demonstrates strong growth potential, certain restraints may include the high cost of targeted therapies, potential for drug resistance development, and stringent regulatory approval processes for new molecular entities. However, ongoing clinical trials and the exploration of combination therapies are expected to mitigate these challenges and sustain the market's upward trend.
targeted drugs for nsclc Company Market Share

Targeted Drugs for NSCLC: Market Dynamics, Growth Trends, and Future Outlook (2019-2033)
This comprehensive report offers an in-depth analysis of the targeted drugs for NSCLC market, providing critical insights for pharmaceutical companies, research institutions, and investors. Our study meticulously examines the market's evolution, competitive landscape, and future potential, covering the period from 2019 to 2033, with a base year of 2025. The report integrates high-traffic keywords for maximum SEO visibility and presents all values in billion units for industry professionals.
Targeted Drugs for NSCLC Market Dynamics & Structure
The targeted therapies for non-small cell lung cancer (NSCLC) market is characterized by a dynamic and evolving landscape, driven by continuous technological advancements and a growing understanding of cancer genetics. Market concentration is moderately fragmented, with key players investing heavily in research and development to identify novel targets and develop highly specific drugs. Technological innovation is the primary driver, with a strong emphasis on precision medicine and molecular profiling to identify patient populations that will benefit most from targeted treatments. Regulatory frameworks, including FDA and EMA approvals, play a crucial role in shaping market access and drug development timelines. Competitive product substitutes are emerging as research uncovers new genetic alterations and potential therapeutic targets, leading to a diverse pipeline of investigational drugs. End-user demographics are shifting towards older populations and those with specific genetic mutations amenable to targeted therapies. Mergers and acquisitions (M&A) trends are observed as larger pharmaceutical companies seek to strengthen their oncology portfolios and acquire innovative pipeline assets.
- Market Concentration: Moderately fragmented with increasing consolidation through strategic partnerships and M&A.
- Technological Innovation Drivers: Genetic sequencing advancements, liquid biopsy development, and predictive biomarker discovery.
- Regulatory Frameworks: Stringent approval processes by major health authorities like the FDA and EMA, with a growing emphasis on real-world evidence.
- Competitive Product Substitutes: Rapid development of next-generation inhibitors and combination therapies.
- End-User Demographics: Aging global population, increasing prevalence of specific NSCLC genetic mutations (EGFR, ALK, etc.).
- M&A Trends: Strategic acquisitions of small biotech firms with promising targeted therapy pipelines.
Targeted Drugs for NSCLC Growth Trends & Insights
The global market for targeted drugs for NSCLC is projected to experience significant growth, driven by an increasing understanding of NSCLC subtypes and their underlying genetic mutations. The market size evolution is a testament to the efficacy of these therapies in improving patient outcomes and survival rates compared to traditional chemotherapy. Adoption rates are steadily increasing as diagnostic tools become more accessible and affordable, allowing for precise patient stratification. Technological disruptions, such as the development of novel drug delivery systems and advancements in companion diagnostics, are further accelerating market penetration. Consumer behavior shifts are evident, with patients and oncologists increasingly favoring targeted therapies due to their improved safety profiles and efficacy in specific patient populations. The child market encompassing specific genetic targets like EGFR and ALK inhibitors is particularly robust, contributing substantially to the overall parent market of targeted NSCLC therapies. This growth is further propelled by ongoing clinical trials and the potential for label expansions for existing drugs.
The parent market for targeted drugs for NSCLC is anticipated to grow at a Compound Annual Growth Rate (CAGR) of approximately 12.5% from 2025 to 2033, reaching an estimated $XX billion by 2033. The child market for EGFR inhibitors alone is expected to contribute significantly, projected to reach $XX billion by the end of the forecast period, with ALK inhibitors following closely at $XX billion.
Market penetration for targeted therapies is rapidly expanding, moving beyond early-stage approvals to encompass adjuvant and neoadjuvant settings. The increasing availability of these therapies in both developed and emerging economies, coupled with rising healthcare expenditures, is a key growth indicator. Technological breakthroughs in identifying novel driver mutations, such as ROS1 and MET alterations, are opening up new avenues for treatment and expanding the addressable patient population. Furthermore, the growing emphasis on personalized medicine is driving demand for therapies that offer a more tailored approach to cancer treatment, thereby boosting the adoption of targeted drugs. The collaborative efforts between pharmaceutical companies, research institutions, and regulatory bodies are crucial in expediting the drug development and approval process, further contributing to the market's upward trajectory. The evolving landscape of clinical trial designs, incorporating adaptive trials and real-world data, is also instrumental in demonstrating the value and efficacy of targeted therapies, leading to wider clinical acceptance and prescribing patterns.
Dominant Regions, Countries, or Segments in Targeted Drugs for NSCLC
The North America region is currently the dominant force in the targeted drugs for NSCLC market, driven by a confluence of factors that foster innovation, access, and adoption. The region boasts a well-established healthcare infrastructure, high patient awareness regarding advanced treatment options, and a strong presence of leading pharmaceutical companies actively engaged in research and development. Within North America, the United States stands out as the leading country, fueled by robust government funding for cancer research, favorable reimbursement policies for innovative therapies, and a significant patient pool with a high prevalence of targetable NSCLC mutations.
The Adenocarcinoma of NSCLC segment represents a particularly strong driver of market growth. This subtype is more frequently associated with targetable genetic mutations like EGFR, ALK, and ROS1, making it a prime area for the application of targeted therapies. The Target EGFR segment, specifically, holds a dominant position within the types of targeted drugs. This is due to the well-established efficacy of EGFR inhibitors in a significant proportion of NSCLC patients, leading to high prescription rates and substantial market share. The ongoing development of next-generation EGFR inhibitors that can overcome resistance mechanisms further solidifies its dominance.
Dominant Region: North America
- Key Drivers: Advanced healthcare infrastructure, high R&D investment, strong pharmaceutical presence, favorable reimbursement policies, high patient awareness.
- Market Share: Estimated at over 40% of the global market.
- Growth Potential: Continued innovation and expansion of treatment indications.
Dominant Country: United States
- Key Drivers: Significant patient population, extensive clinical trial activity, early adoption of new therapies, comprehensive diagnostic capabilities.
- Market Share: Accounts for the majority of the North American market share.
- Growth Potential: Driven by an aging population and increasing diagnosis of actionable mutations.
Dominant Application Segment: Adenocarcinoma of NSCLC
- Key Drivers: Higher prevalence of targetable mutations (EGFR, ALK, ROS1) compared to other NSCLC subtypes.
- Market Share: Represents a substantial portion of patients receiving targeted therapies.
- Growth Potential: Ongoing research into new targets within this subtype.
Dominant Type Segment: Target EGFR
- Key Drivers: High efficacy of approved EGFR inhibitors, development of next-generation inhibitors, widespread patient testing for EGFR mutations.
- Market Share: Leading segment within targeted drug types.
- Growth Potential: Overcoming resistance mechanisms and potential for broader application.
The rapid adoption of companion diagnostics in the United States is instrumental in identifying patients with specific genetic markers, thereby facilitating the precise use of targeted drugs. Furthermore, the presence of academic medical centers and robust clinical trial networks ensures that novel therapies are rigorously tested and quickly integrated into clinical practice. Economic policies that incentivize pharmaceutical innovation and market access, coupled with a high disposable income for a significant portion of the population, further bolster the growth of the targeted NSCLC drug market in this region. The increasing prevalence of Adenocarcinoma of NSCLC due to improved diagnostic capabilities and a better understanding of its molecular underpinnings directly translates into a larger patient pool eligible for therapies targeting specific mutations. Similarly, the mature market for EGFR inhibitors is sustained by continuous research into overcoming resistance and expanding their use in earlier lines of therapy.
Targeted Drugs for NSCLC Product Landscape
The targeted drugs for NSCLC market is characterized by a pipeline of innovative molecules designed to inhibit specific oncogenic drivers. These therapies, including small molecule inhibitors and antibody-drug conjugates, offer improved efficacy and reduced toxicity compared to traditional chemotherapy. Key product innovations focus on overcoming resistance mechanisms, expanding target coverage, and developing synergistic combination therapies. For instance, next-generation EGFR inhibitors have demonstrated superior activity against common resistance mutations, while novel ALK inhibitors continue to improve patient outcomes. The performance metrics of these drugs are typically measured by objective response rates (ORR), progression-free survival (PFS), and overall survival (OS), with leading products consistently demonstrating significant improvements in these endpoints. The unique selling propositions lie in their molecular specificity, leading to a more personalized approach to cancer treatment.
Key Drivers, Barriers & Challenges in Targeted Drugs for NSCLC
Key Drivers:
- Technological Advancements: Breakthroughs in genomic sequencing, liquid biopsy, and biomarker discovery enable precise patient stratification and identification of actionable mutations.
- Rising Incidence of NSCLC: Increasing global prevalence of lung cancer, particularly adenocarcinoma, drives demand for effective treatment options.
- Improved Patient Outcomes: Targeted therapies offer significantly better efficacy and safety profiles compared to traditional chemotherapy for selected patient groups.
- Growing Investment in R&D: Pharmaceutical companies are heavily investing in developing novel targeted agents and exploring new molecular targets.
- Favorable Regulatory Pathways: Expedited review and approval processes for oncology drugs accelerate market entry.
Barriers & Challenges:
- High Cost of Therapies: The significant price of targeted drugs poses a substantial financial burden on healthcare systems and patients, impacting accessibility.
- Development of Drug Resistance: Tumor cells can develop resistance to targeted therapies over time, necessitating the development of next-generation drugs and combination strategies.
- Complex Diagnostic Pathways: The need for accurate and timely molecular profiling can be a bottleneck, especially in resource-limited settings.
- Regulatory Hurdles for Combination Therapies: Navigating approval processes for novel drug combinations can be complex and time-consuming.
- Supply Chain Disruptions: Global supply chain vulnerabilities can impact the availability of essential drugs and raw materials.
Emerging Opportunities in Targeted Drugs for NSCLC
Emerging opportunities in the targeted drugs for NSCLC market lie in the exploration of novel molecular targets beyond the established ones like EGFR and ALK. The identification of rare mutations and the development of therapies for previously "undruggable" targets present significant untapped potential. Furthermore, the expansion of targeted therapies into earlier stages of NSCLC, including adjuvant and neoadjuvant settings, offers a substantial growth avenue. The increasing integration of artificial intelligence (AI) and machine learning in drug discovery and development is poised to accelerate the identification of new targets and optimize treatment strategies. The growing focus on overcoming treatment resistance through innovative combination therapies and the development of novel drug delivery systems also represents a key opportunity.
Growth Accelerators in the Targeted Drugs for NSCLC Industry
Catalysts driving long-term growth in the targeted drugs for NSCLC industry include the continuous evolution of precision medicine, leading to a deeper understanding of tumor heterogeneity and the identification of new resistance mechanisms. Strategic partnerships and collaborations between pharmaceutical giants and smaller biotechnology firms are accelerating pipeline development and fostering innovation. Market expansion strategies, particularly in emerging economies where access to advanced cancer treatments is growing, are crucial for widening the reach of these therapies. The ongoing advancements in liquid biopsy technologies are enabling less invasive and more frequent monitoring of treatment response and resistance, which will further enhance the utility of targeted drugs.
Key Players Shaping the Targeted Drugs for NSCLC Market
- Allergan
- Amgen
- ARIAD Pharmaceuticals (Takeda)
- Beacon Pharma Limited
- BeiGene
- Biocon
- Boehringer-Ingelheim
- Celgene Corporation
- Drug International Limited
- Fujifilm Kyowa Kirin Biologics
- Genvio Pharma Limited
- Hetero Drugs
- ImClone Systems (Eli Lilly)
- Incepta Pharmaceuticals
- Mylan
- Novartis
- Pfizer
- Reliance Lifesciences
- Roche
- AstraZeneca
- Cipla
- Teva
- OSI Pharmaceuticals
- Glenmark Pharmaceuticals
- Natco Pharma
Notable Milestones in Targeted Drugs for NSCLC Sector
- 2019: Approval of osimertinib for adjuvant treatment of EGFR-mutated NSCLC.
- 2020: Breakthrough designation for novel ALK inhibitors showing efficacy in resistant disease.
- 2021: Significant progress in liquid biopsy for early detection of resistance mutations.
- 2022: Approval of first-in-class MET inhibitor for METex14 skipping NSCLC.
- 2023: Emergence of combination therapies showing synergistic effects in advanced NSCLC.
- 2024: Increased focus on targeting KRAS mutations in NSCLC with novel small molecules.
In-Depth Targeted Drugs for NSCLC Market Outlook
The future market for targeted drugs for NSCLC is exceptionally promising, driven by accelerating growth catalysts such as the continuous refinement of precision medicine approaches and the identification of novel therapeutic targets. Strategic alliances and collaborations are vital in expediting drug development and commercialization. Furthermore, the expanding market reach into emerging economies, coupled with advancements in liquid biopsy and the development of novel combination therapies, will significantly shape market dynamics. The anticipated market size for targeted therapies in NSCLC is projected to reach $XX billion by 2033, representing a significant compound annual growth rate.
Report Scope:
- Study Period: 2019–2033
- Base Year: 2025
- Estimated Year: 2025
- Forecast Period: 2025–2033
- Historical Period: 2019–2024
Segments Covered:
- Application: Squamous Cell Carcinoma of NSCLC, Adenocarcinoma of NSCLC, Large Cell Carcinoma of NSCLC
- Types: Target EGFR, Target ALK, Target HER2, Target ROS1, Target BRAF, Target MEK, Target VEGFR2, Target VEGF, Target MET
Company Coverage: Allergan, Amgen, ARIAD Pharmaceuticals (Takeda), Beacon Pharma Limited, BeiGene, Biocon, Boehringer-Ingelheim, Celgene Corporation, Drug International Limited, Fujifilm Kyowa Kirin Biologics, Genvio Pharma Limited, Hetero Drugs, ImClone Systems (Eli Lilly), Incepta Pharmaceuticals, Mylan, Novartis, Pfizer, Reliance Lifesciences, Roche, AstraZeneca, Cipla, Teva, OSI Pharmaceuticals, Glenmark Pharmaceuticals, Natco Pharma.
targeted drugs for nsclc Segmentation
-
1. Application
- 1.1. Squamous Cell Carcinoma of NSCLC
- 1.2. Adenocarcinoma of NSCLC
- 1.3. Large Cell Carcinoma of NSCLC
-
2. Types
- 2.1. Target EGFR
- 2.2. Target ALK
- 2.3. Target HER2
- 2.4. Target ROS1
- 2.5. Target BRAF
- 2.6. Target MEK
- 2.7. Target VEGFR2
- 2.8. Target VEGF
- 2.9. Target MET
targeted drugs for nsclc Segmentation By Geography
- 1. CA
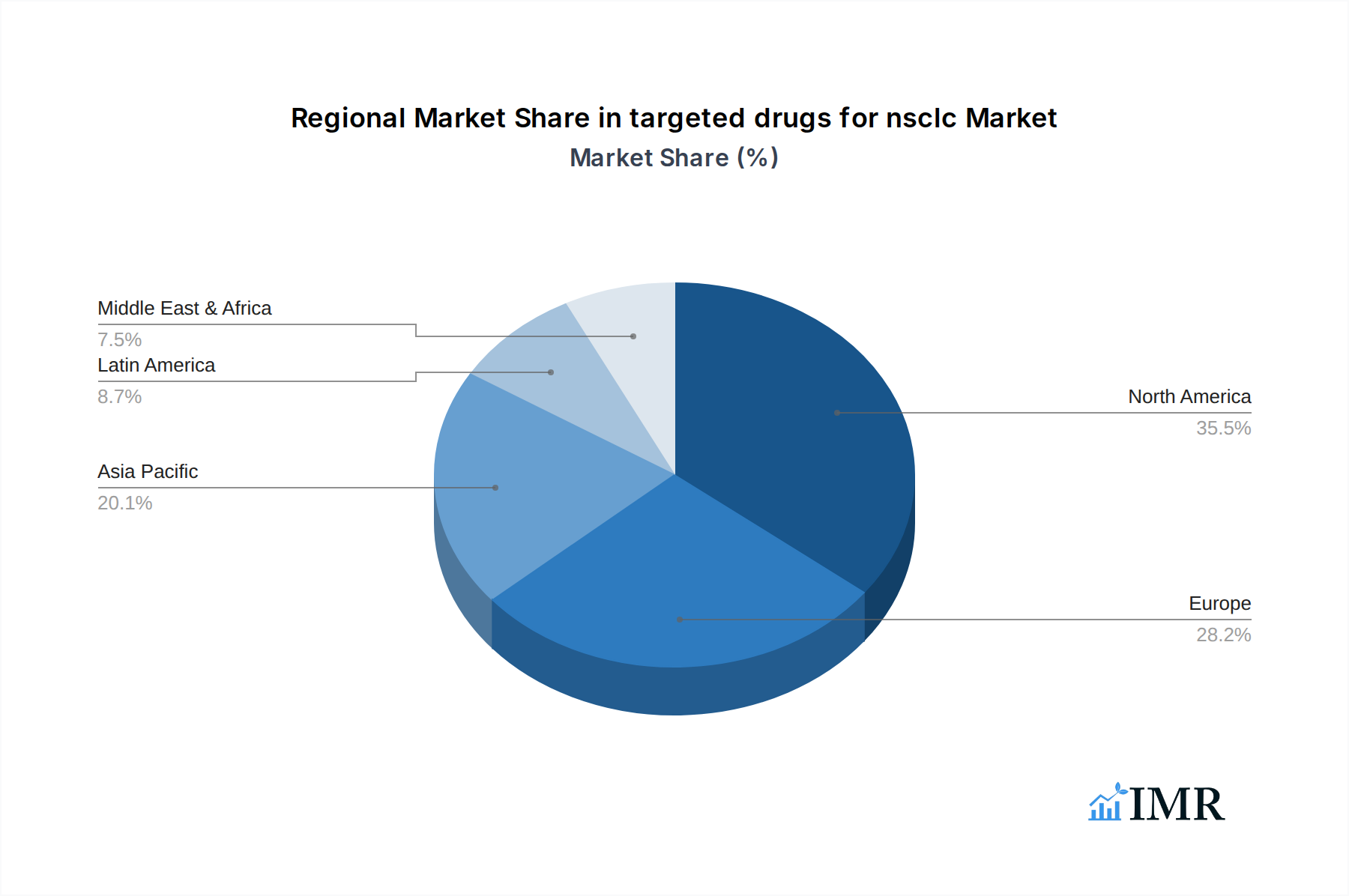
targeted drugs for nsclc Regional Market Share

Geographic Coverage of targeted drugs for nsclc
targeted drugs for nsclc REPORT HIGHLIGHTS
| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.87% from 2020-2034 |
| Segmentation |
|
Table of Contents
- 1. Introduction
- 1.1. Research Scope
- 1.2. Market Segmentation
- 1.3. Research Methodology
- 1.4. Definitions and Assumptions
- 2. Executive Summary
- 2.1. Introduction
- 3. Market Dynamics
- 3.1. Introduction
- 3.2. Market Drivers
- 3.3. Market Restrains
- 3.4. Market Trends
- 4. Market Factor Analysis
- 4.1. Porters Five Forces
- 4.2. Supply/Value Chain
- 4.3. PESTEL analysis
- 4.4. Market Entropy
- 4.5. Patent/Trademark Analysis
- 5. targeted drugs for nsclc Analysis, Insights and Forecast, 2020-2032
- 5.1. Market Analysis, Insights and Forecast - by Application
- 5.1.1. Squamous Cell Carcinoma of NSCLC
- 5.1.2. Adenocarcinoma of NSCLC
- 5.1.3. Large Cell Carcinoma of NSCLC
- 5.2. Market Analysis, Insights and Forecast - by Types
- 5.2.1. Target EGFR
- 5.2.2. Target ALK
- 5.2.3. Target HER2
- 5.2.4. Target ROS1
- 5.2.5. Target BRAF
- 5.2.6. Target MEK
- 5.2.7. Target VEGFR2
- 5.2.8. Target VEGF
- 5.2.9. Target MET
- 5.3. Market Analysis, Insights and Forecast - by Region
- 5.3.1. CA
- 5.1. Market Analysis, Insights and Forecast - by Application
- 6. Competitive Analysis
- 6.1. Market Share Analysis 2025
- 6.2. Company Profiles
- 6.2.1 Allergan
- 6.2.1.1. Overview
- 6.2.1.2. Products
- 6.2.1.3. SWOT Analysis
- 6.2.1.4. Recent Developments
- 6.2.1.5. Financials (Based on Availability)
- 6.2.2 Amgen
- 6.2.2.1. Overview
- 6.2.2.2. Products
- 6.2.2.3. SWOT Analysis
- 6.2.2.4. Recent Developments
- 6.2.2.5. Financials (Based on Availability)
- 6.2.3 ARIAD Pharmaceuticals (Takeda)
- 6.2.3.1. Overview
- 6.2.3.2. Products
- 6.2.3.3. SWOT Analysis
- 6.2.3.4. Recent Developments
- 6.2.3.5. Financials (Based on Availability)
- 6.2.4 Beacon Pharma Limited
- 6.2.4.1. Overview
- 6.2.4.2. Products
- 6.2.4.3. SWOT Analysis
- 6.2.4.4. Recent Developments
- 6.2.4.5. Financials (Based on Availability)
- 6.2.5 BeiGene
- 6.2.5.1. Overview
- 6.2.5.2. Products
- 6.2.5.3. SWOT Analysis
- 6.2.5.4. Recent Developments
- 6.2.5.5. Financials (Based on Availability)
- 6.2.6 Biocon
- 6.2.6.1. Overview
- 6.2.6.2. Products
- 6.2.6.3. SWOT Analysis
- 6.2.6.4. Recent Developments
- 6.2.6.5. Financials (Based on Availability)
- 6.2.7 Boehringer-Ingelheim
- 6.2.7.1. Overview
- 6.2.7.2. Products
- 6.2.7.3. SWOT Analysis
- 6.2.7.4. Recent Developments
- 6.2.7.5. Financials (Based on Availability)
- 6.2.8 Celgene Corporation
- 6.2.8.1. Overview
- 6.2.8.2. Products
- 6.2.8.3. SWOT Analysis
- 6.2.8.4. Recent Developments
- 6.2.8.5. Financials (Based on Availability)
- 6.2.9 Drug International Limted
- 6.2.9.1. Overview
- 6.2.9.2. Products
- 6.2.9.3. SWOT Analysis
- 6.2.9.4. Recent Developments
- 6.2.9.5. Financials (Based on Availability)
- 6.2.10 Fujifilm Kyowa Kirin Biologics
- 6.2.10.1. Overview
- 6.2.10.2. Products
- 6.2.10.3. SWOT Analysis
- 6.2.10.4. Recent Developments
- 6.2.10.5. Financials (Based on Availability)
- 6.2.11 Genvio Pharma Limited
- 6.2.11.1. Overview
- 6.2.11.2. Products
- 6.2.11.3. SWOT Analysis
- 6.2.11.4. Recent Developments
- 6.2.11.5. Financials (Based on Availability)
- 6.2.12 Hetero Drugs
- 6.2.12.1. Overview
- 6.2.12.2. Products
- 6.2.12.3. SWOT Analysis
- 6.2.12.4. Recent Developments
- 6.2.12.5. Financials (Based on Availability)
- 6.2.13 ImClone Systems (Eli Lilly)
- 6.2.13.1. Overview
- 6.2.13.2. Products
- 6.2.13.3. SWOT Analysis
- 6.2.13.4. Recent Developments
- 6.2.13.5. Financials (Based on Availability)
- 6.2.14 Incepta Pharmaceuticals
- 6.2.14.1. Overview
- 6.2.14.2. Products
- 6.2.14.3. SWOT Analysis
- 6.2.14.4. Recent Developments
- 6.2.14.5. Financials (Based on Availability)
- 6.2.15 Mylan
- 6.2.15.1. Overview
- 6.2.15.2. Products
- 6.2.15.3. SWOT Analysis
- 6.2.15.4. Recent Developments
- 6.2.15.5. Financials (Based on Availability)
- 6.2.16 Novartis
- 6.2.16.1. Overview
- 6.2.16.2. Products
- 6.2.16.3. SWOT Analysis
- 6.2.16.4. Recent Developments
- 6.2.16.5. Financials (Based on Availability)
- 6.2.17 Pfizer
- 6.2.17.1. Overview
- 6.2.17.2. Products
- 6.2.17.3. SWOT Analysis
- 6.2.17.4. Recent Developments
- 6.2.17.5. Financials (Based on Availability)
- 6.2.18 Reliance Lifesciences
- 6.2.18.1. Overview
- 6.2.18.2. Products
- 6.2.18.3. SWOT Analysis
- 6.2.18.4. Recent Developments
- 6.2.18.5. Financials (Based on Availability)
- 6.2.19 Roche
- 6.2.19.1. Overview
- 6.2.19.2. Products
- 6.2.19.3. SWOT Analysis
- 6.2.19.4. Recent Developments
- 6.2.19.5. Financials (Based on Availability)
- 6.2.20 AstraZeneca
- 6.2.20.1. Overview
- 6.2.20.2. Products
- 6.2.20.3. SWOT Analysis
- 6.2.20.4. Recent Developments
- 6.2.20.5. Financials (Based on Availability)
- 6.2.21 Cipla
- 6.2.21.1. Overview
- 6.2.21.2. Products
- 6.2.21.3. SWOT Analysis
- 6.2.21.4. Recent Developments
- 6.2.21.5. Financials (Based on Availability)
- 6.2.22 Teva
- 6.2.22.1. Overview
- 6.2.22.2. Products
- 6.2.22.3. SWOT Analysis
- 6.2.22.4. Recent Developments
- 6.2.22.5. Financials (Based on Availability)
- 6.2.23 OSI Pharmaceuticals
- 6.2.23.1. Overview
- 6.2.23.2. Products
- 6.2.23.3. SWOT Analysis
- 6.2.23.4. Recent Developments
- 6.2.23.5. Financials (Based on Availability)
- 6.2.24 Glenmark Pharmaceuticals
- 6.2.24.1. Overview
- 6.2.24.2. Products
- 6.2.24.3. SWOT Analysis
- 6.2.24.4. Recent Developments
- 6.2.24.5. Financials (Based on Availability)
- 6.2.25 Natco Pharma
- 6.2.25.1. Overview
- 6.2.25.2. Products
- 6.2.25.3. SWOT Analysis
- 6.2.25.4. Recent Developments
- 6.2.25.5. Financials (Based on Availability)
- 6.2.1 Allergan
List of Figures
- Figure 1: targeted drugs for nsclc Revenue Breakdown (billion, %) by Product 2025 & 2033
- Figure 2: targeted drugs for nsclc Share (%) by Company 2025
List of Tables
- Table 1: targeted drugs for nsclc Revenue billion Forecast, by Application 2020 & 2033
- Table 2: targeted drugs for nsclc Revenue billion Forecast, by Types 2020 & 2033
- Table 3: targeted drugs for nsclc Revenue billion Forecast, by Region 2020 & 2033
- Table 4: targeted drugs for nsclc Revenue billion Forecast, by Application 2020 & 2033
- Table 5: targeted drugs for nsclc Revenue billion Forecast, by Types 2020 & 2033
- Table 6: targeted drugs for nsclc Revenue billion Forecast, by Country 2020 & 2033
Frequently Asked Questions
1. What is the projected Compound Annual Growth Rate (CAGR) of the targeted drugs for nsclc?
The projected CAGR is approximately 6.87%.
2. Which companies are prominent players in the targeted drugs for nsclc?
Key companies in the market include Allergan, Amgen, ARIAD Pharmaceuticals (Takeda), Beacon Pharma Limited, BeiGene, Biocon, Boehringer-Ingelheim, Celgene Corporation, Drug International Limted, Fujifilm Kyowa Kirin Biologics, Genvio Pharma Limited, Hetero Drugs, ImClone Systems (Eli Lilly), Incepta Pharmaceuticals, Mylan, Novartis, Pfizer, Reliance Lifesciences, Roche, AstraZeneca, Cipla, Teva, OSI Pharmaceuticals, Glenmark Pharmaceuticals, Natco Pharma.
3. What are the main segments of the targeted drugs for nsclc?
The market segments include Application, Types.
4. Can you provide details about the market size?
The market size is estimated to be USD 85.69 billion as of 2022.
5. What are some drivers contributing to market growth?
N/A
6. What are the notable trends driving market growth?
N/A
7. Are there any restraints impacting market growth?
N/A
8. Can you provide examples of recent developments in the market?
N/A
9. What pricing options are available for accessing the report?
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3400.00, USD 5100.00, and USD 6800.00 respectively.
10. Is the market size provided in terms of value or volume?
The market size is provided in terms of value, measured in billion.
11. Are there any specific market keywords associated with the report?
Yes, the market keyword associated with the report is "targeted drugs for nsclc," which aids in identifying and referencing the specific market segment covered.
12. How do I determine which pricing option suits my needs best?
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
13. Are there any additional resources or data provided in the targeted drugs for nsclc report?
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
14. How can I stay updated on further developments or reports in the targeted drugs for nsclc?
To stay informed about further developments, trends, and reports in the targeted drugs for nsclc, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.
Methodology
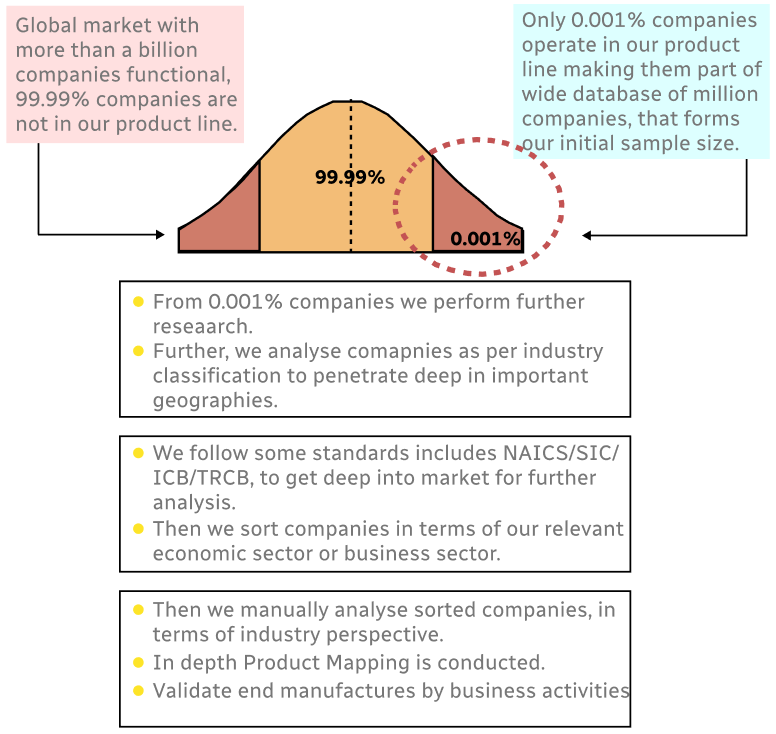
Step 1 - Identification of Relevant Samples Size from Population Database
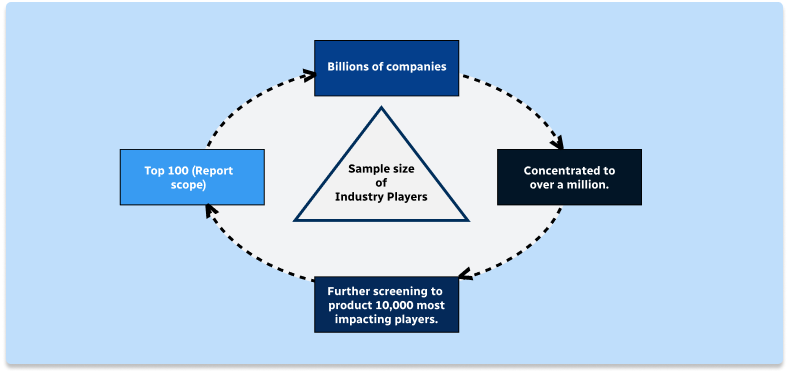
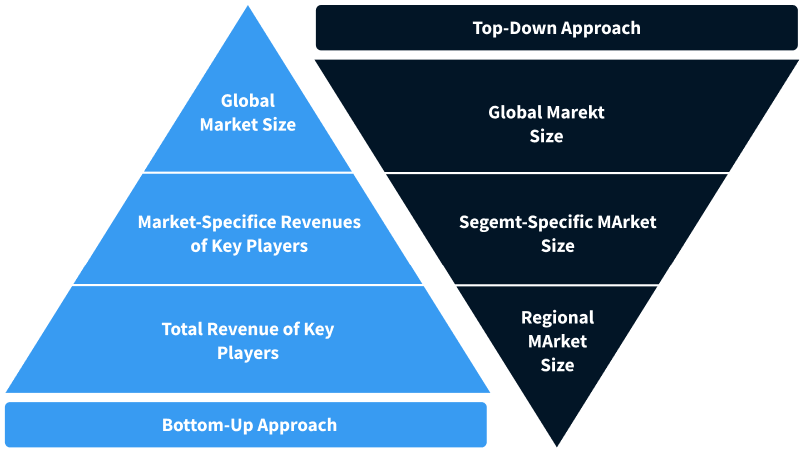
Step 2 - Approaches for Defining Global Market Size (Value, Volume* & Price*)
Note*: In applicable scenarios
Step 3 - Data Sources
Primary Research
- Web Analytics
- Survey Reports
- Research Institute
- Latest Research Reports
- Opinion Leaders
Secondary Research
- Annual Reports
- White Paper
- Latest Press Release
- Industry Association
- Paid Database
- Investor Presentations
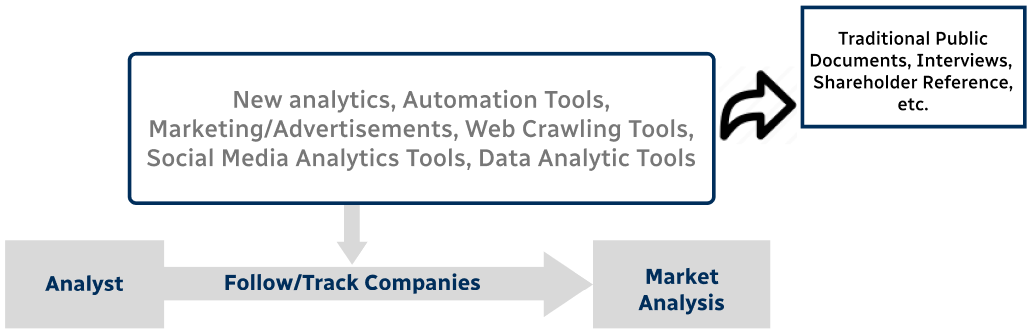
Step 4 - Data Triangulation
Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence


